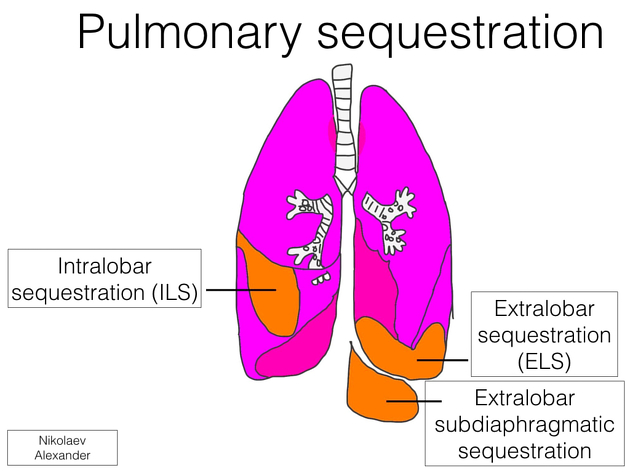
Pulmonary sequestration, also called accessory lung, refers to the aberrant formation of segmental lung tissue that has no connection with the bronchial tree or pulmonary arteries. It is a bronchopulmonary foregut malformation (BPFM).
There are two types:
-
extralobar sequestration (ELS)
extralobar intrathoracic
extralobar subdiaphragmatic
On this page:
Epidemiology
The estimated incidence is 0.1%. Some authors propose a greater male prevalence (this may be the case for the extralobar type) ref. The age of presentation is dependent on the type of sequestration (see below).
Associations
Associated disease is common with the extralobar type (50-60%):
congenital pulmonary airway malformation (CPAM): which is then sometimes termed a hybrid lesion
Clinical presentation
Extralobar sequestration more commonly presents in newborns as respiratory distress, cyanosis, or infection ref. Intralobar sequestration presents in late childhood or adolescence with recurrent pulmonary infections ref, and in adults with cough, chest/back pain, dyspnea, and fever 16.
Pathology
Pulmonary sequestration can be divided into two distinct groups based on the relationship of the aberrant segmental lung tissue to the pleura:
-
accounts for the majority (75-85% of all sequestrations 4,5,7)
present later in childhood with recurrent infections
-
less common (15-25% of all sequestrations 4,5,7)
usually present in the neonatal period with respiratory distress, cyanosis, or infection
recognized male predilection (M:F ratio ~4:1)
can be infradiaphragmatic in ~10% of cases
The two types of sequestration are similar in their relationship to the bronchial tree and arterial supply/venous drainage but differ in their relationship to the pleura.
By definition, there is no communication with the tracheobronchial tree. In the vast majority of cases, the abnormal lung tissue has a systemic arterial supply which is usually a branch of the aorta:
-
intralobar sequestrations
venous drainage commonly occurs via the pulmonary veins but can occur through the azygos-hemiazygos system, portal vein, right atrium or inferior vena cava
closely connected to the adjacent normal lung and do not have a separate pleura
-
extralobar sequestrations
venous drainage most commonly through the systemic veins into the right atrium (but is variable)
separated from any surrounding lung by its own pleura
Location
Overall, sequestration preferentially affects the lower lobes. 60% of intralobar sequestrations affect the left lower lobe, and 40% the right lower lobe. Extralobar sequestrations almost always affect the left lower lobe, however, ~10% of extralobar sequestrations can be subdiaphragmatic 8.
Genetics
Almost all cases occur sporadically.
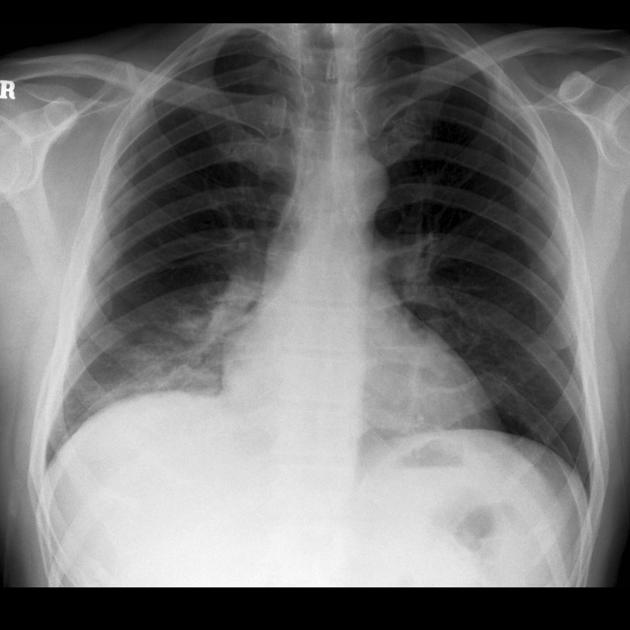
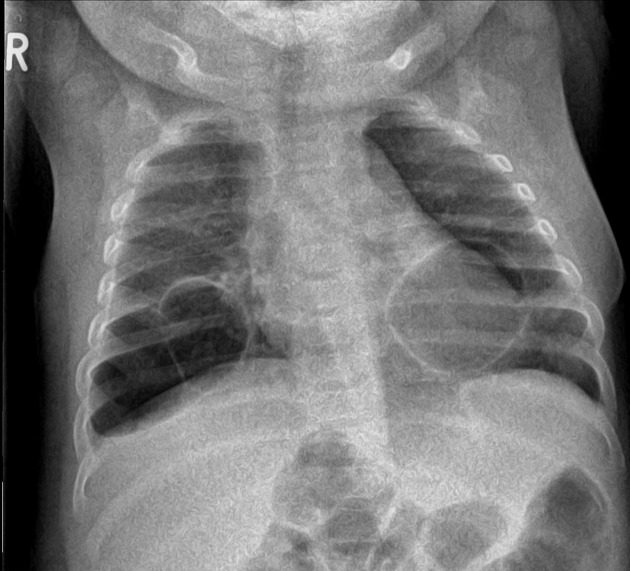
Radiographic features
Plain radiograph
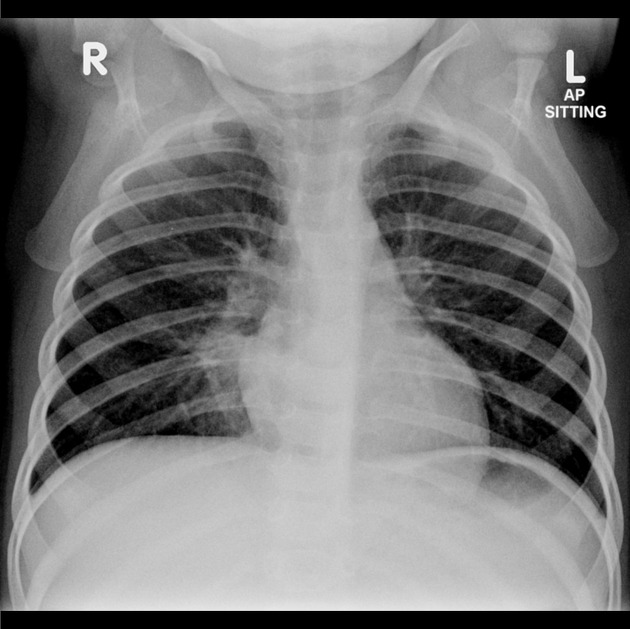
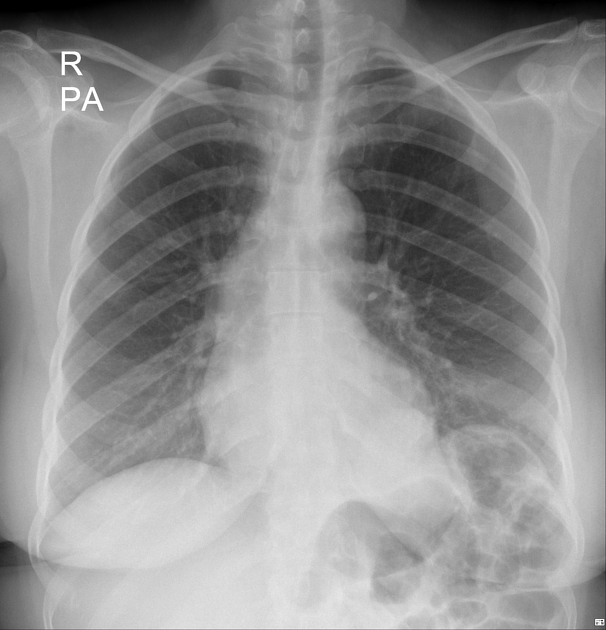
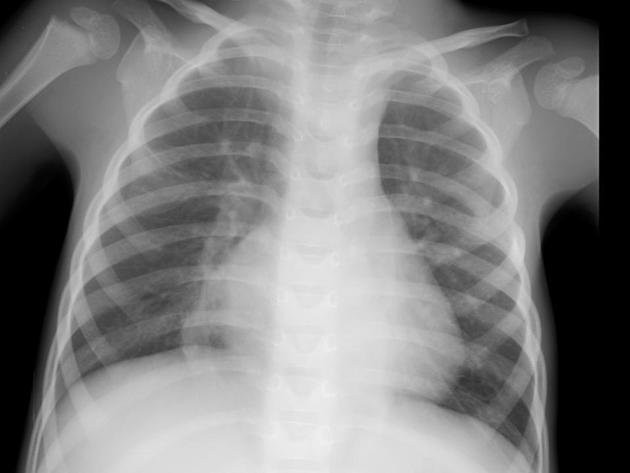
often show a triangular opacity in the affected segment
may show cystic spaces if infected
both intralobar and extra lobar sequestration can rarely have air bronchograms as they may acquire a connection to the bronchial tree due to an infective process or, rarely, they can have foregut communication (esophagus or stomach) as part of hybrid lesion
Ultrasound
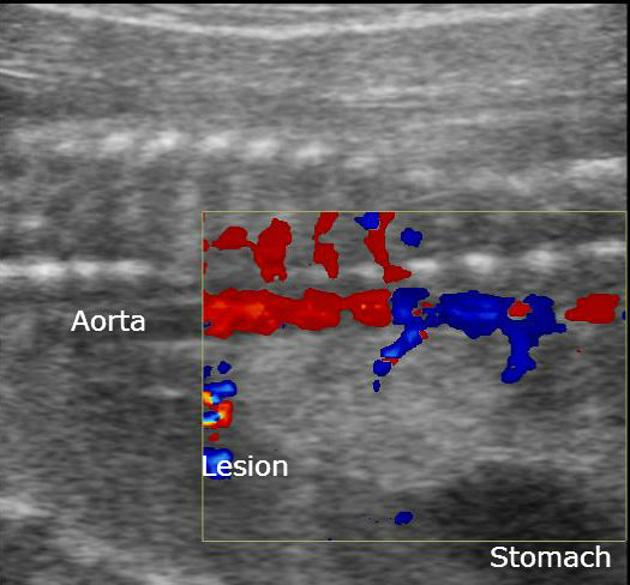
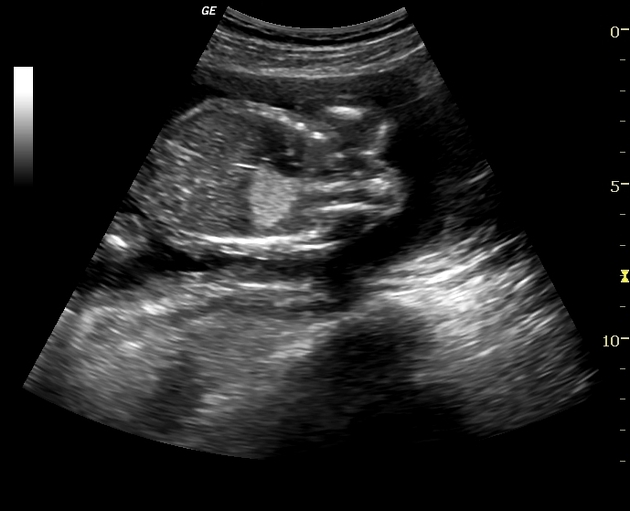
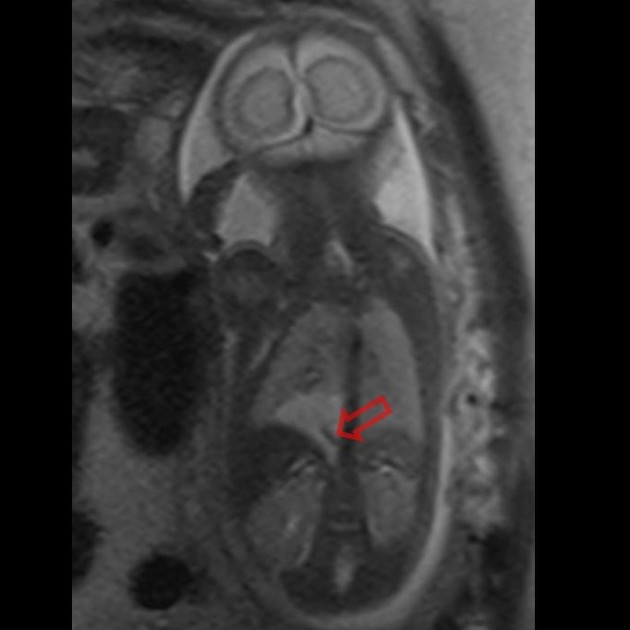
The sequestrated portion of the lung is usually more echogenic than the rest of the lung. On antenatal ultrasound, an extralobar sequestration may be seen as early as 16 weeks gestation and typically appears as a solid well-defined triangular echogenic mass 8. Color Doppler may identify a feeding vessel (in-utero cases) from the aorta. If the sequestration is subdiaphragmatic, it may appear as an echogenic intra-abdominal mass.
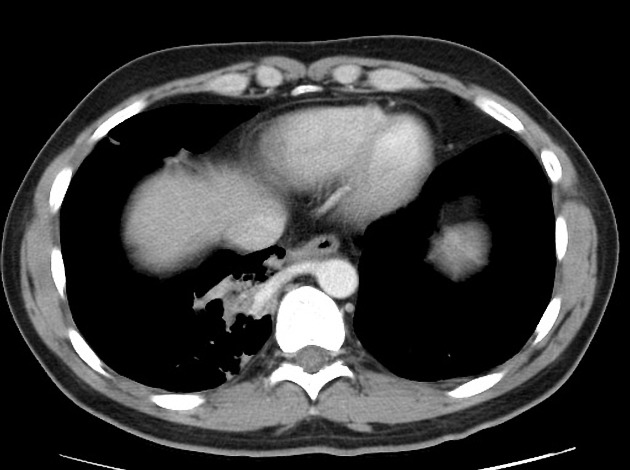
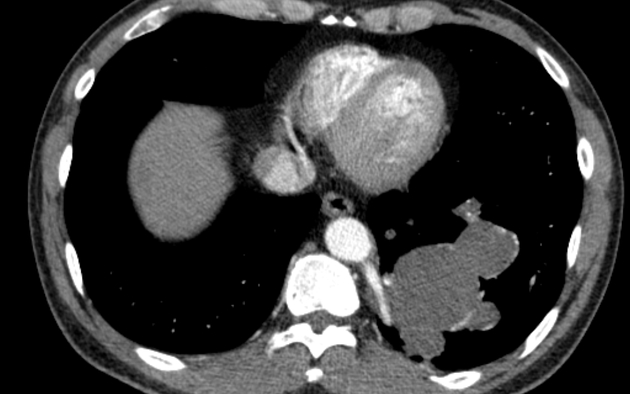
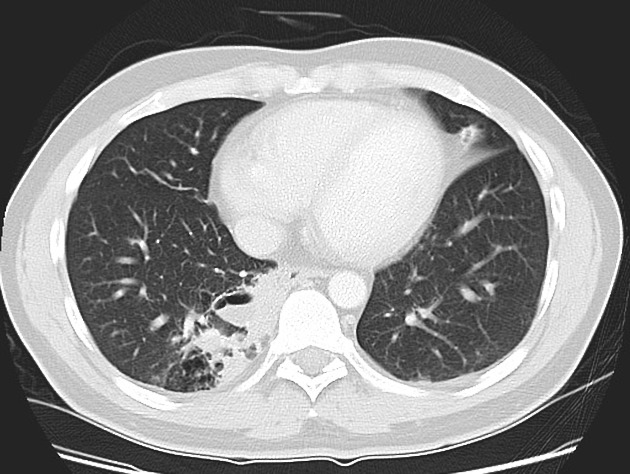
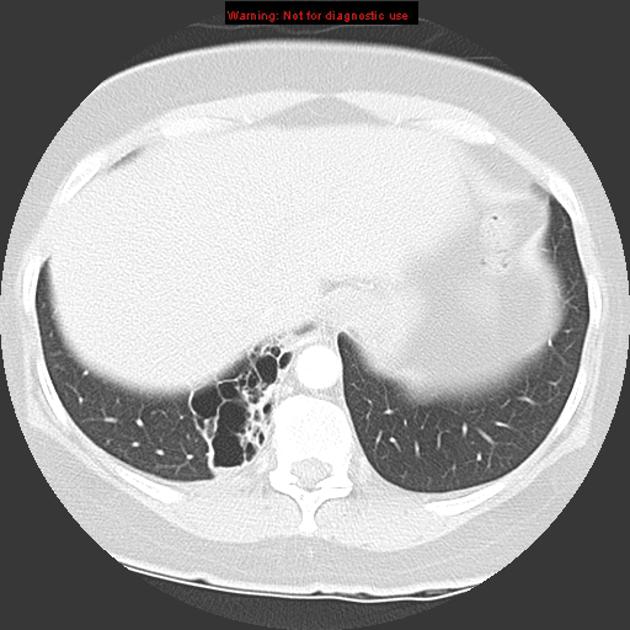
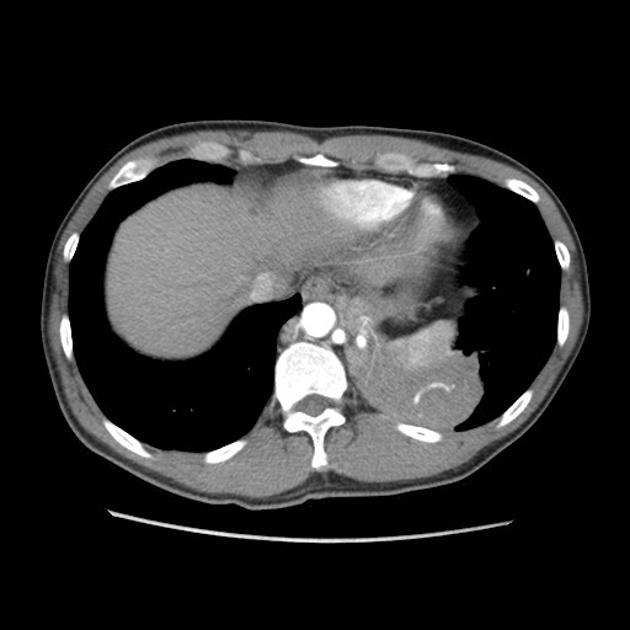
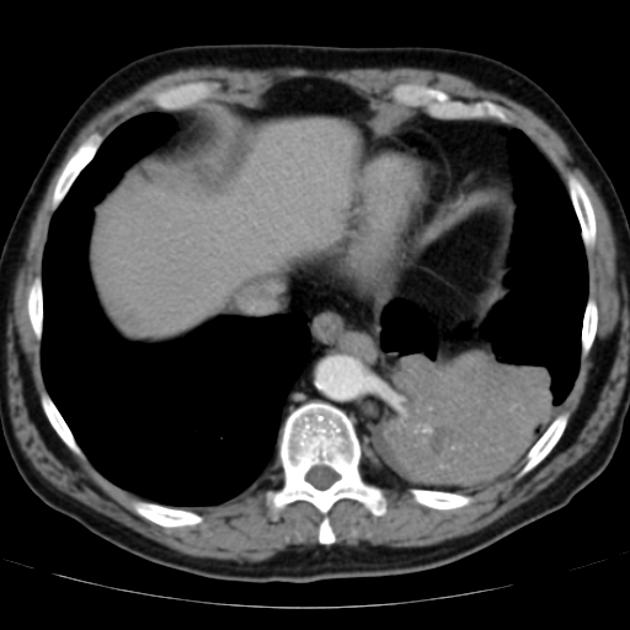
CT
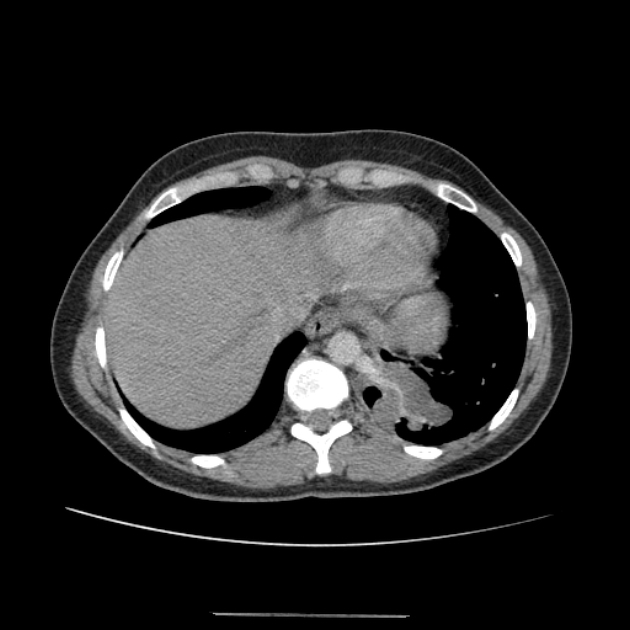
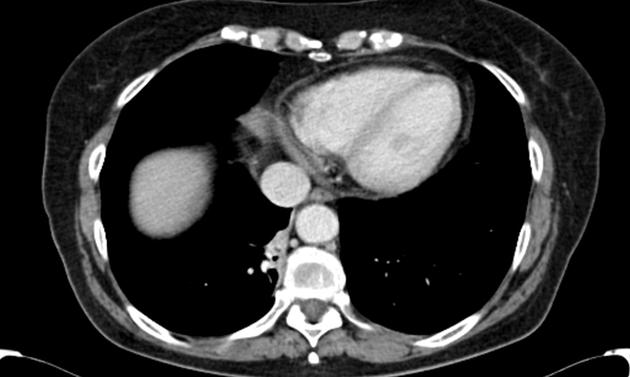
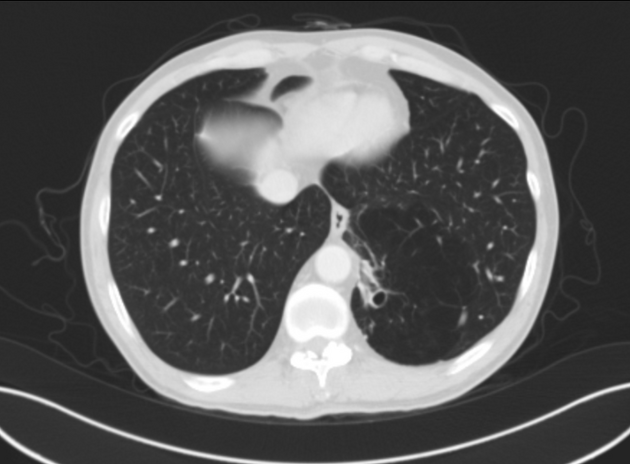
cross-sectional imaging frequently demonstrates the arterial supply by the descending aorta
they may arise below the diaphragm in 20% of patients
usually does not contain air unless infected
-
3D reconstructions can be particularly helpful in detecting 7
anomalous arterial vessels
concurrent anomalous veins
differentiating between intralobar and extralobar sequestrations
Angiography (DSA)
Not part of the routine investigation but is the gold standard in determining arterial supply.
MRI
T1: the sequestrated segment tends to be of comparatively high signal to normal lung tissue 14
T2: also tends to be of comparatively high signal 14
MRA: can be helpful in demonstrating anomalous arterial supply
Treatment and prognosis
Traditionally treatment has been surgical resection. Extralobar sequestrations with their separate pleural investments can usually be removed sparing normal lung tissue, although, with an intralobar type, segmental resection or even lobectomy will be necessary.
Coil embolization has also been successfully trialled in selected cases 4. Spontaneous involution has been reported in occasional cases 10.
Complications
frequent respiratory tract infection
in neonates can be complicated by high-output cardiac failure
History and etymology
in 1946, Pryce used the term pulmonary sequestration for the first time 16.
Differential diagnosis
General imaging differential considerations include:
persistent pneumonia
-
congenital pulmonary airway malformation (CPAM)
can also be an association: bronchopulmonary foregut malformation hybrid lesion
-
can also be an association: bronchopulmonary foregut malformation hybrid lesion
-
small lung with ipsilateral mediastinal shift
tubular structure paralleling the right heart border in the shape of a Turkish sword (“scimitar”)
right heart border may be blurred and may be mistaken for a triangular-shaped sequestration
for anomalous systemic vessel without distinct sequestration consider anomalous systemic arterial supply to normal lung (also called Pryce type 1 sequestration)
if with concurrent lung infection consider pulmonary pseudosequestration




 Unable to process the form. Check for errors and try again.
Unable to process the form. Check for errors and try again.