Presentation
Right upper arm pain and swelling. History of previous curettage of a bony mass lesion in that location.
Patient Data
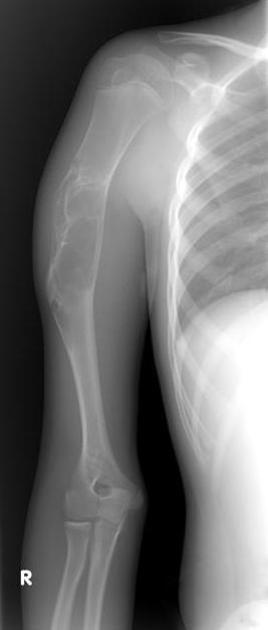
A well-defined right proximal humeral diaphyseal expansile, multilocular lesion with a geographic, sclerotic margin. No matrix calcification and no visible periosteal reaction.

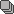

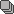

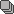

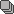

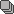

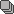

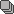
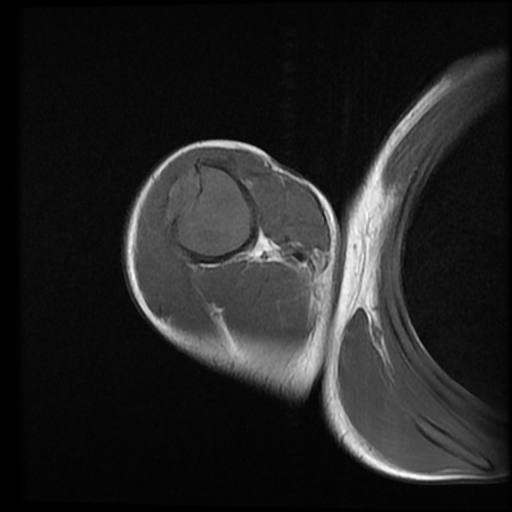
An expansile, mildly lobulated, septated lesion is seen within the humeral diaphysis. The lesion shows low-to-intermediate T1 and high T2/STIR signal intensity with a T1/T2 hypointense sclerotic rim. The lesion measures 11 x 3 x 3 cm in its main CC, TS and AP axes, respectively. There is endosteal scalloping with a sclerotic margin and a narrow zone of transition. Focal cortical destruction is noted with a small extraosseous soft tissue component at the anterolateral aspect of the lesion. No fluid/fluid levels are seen. The lesion shows heterogeneous post-contrast enhancement, with enhancing septa.
Case Discussion
Presumed recurrent chondromyxoid fibroma of the proximal humerus.
The differential diagnosis is:
- Aneurysmal bone cyst (ABC): fluid/fluid level, periosteal reaction, no matrix mineralization. Chondromyxoid fibroma with secondary ABC is not uncommon.
- Fibrous dysplasia: central location, no internal septation or periosteal reaction. Ground glass attenuation is typical.
- Adamantinoma: older patient, vast majority in the upper tibia, cortical destruction with aggressive periosteal reaction.
- Fibrous cortical defect (FCD) and non-ossifying fibroma (NOF): Possible mild cortical expansion, no destruction or periosteal reaction, not painful.




 Unable to process the form. Check for errors and try again.
Unable to process the form. Check for errors and try again.