The injection of dermal fillers refers to the injection of biological or synthetic substances into compartments of the skin to achieve a desirable cosmetic outcome either to restore volume loss or to remove wrinkles or both. They can lead to complications, which might require imaging. Furthermore, they can constitute confounding factors in the workup of orofacial and nasofacial, orbital and cutaneous diseases and malignancies.
On this page:
Terminology
The autologous, biological or synthetic substances injected into the cutaneous compartments are known as dermal fillers or injectable fillers.
Substances/products
Dermal fillers can be classified according to nature, composition or biodegradation time 1-3.
-
fast resorbable dermal fillers
autologous body fat: inconsistent reabsorption rates
collagen (bovine, porcine, human)
hyaluronic acid (bacterial origin)
-
slowly resorbable dermal fillers
polyalkylimide and polyacrylamide hydrogels (PAAG)
poly-L-lactic acid (PLLA)
calcium hydroxyapatite (CHA)
-
non-resorbable/permanent fillers
polymethylmethacrylate (PMMA) microspheres
polytetrafluoroethylene
Indications
Indications of facial dermal injections include the following 1-3:
facial rejuvenation (different degrees)
volume augmentation
soft tissue and static tissue stabilization
volumetric fat loss
facial lipoatrophy
posttraumatic disfiguring
Contraindications
Contraindications include present dermal or subcutaneous infections as well as known allergic reactions to the respective substance (filler) or the anesthetic mixed with it e.g. lidocaine 4.
Procedure
There are different injection techniques, in all patients safety and comfort should be ensured:
-
tunneling technique
suitable for straight lines
aims to ensure the filler does not spread
can be performed at different depths or skin layers
can be performed as retrograde ‘retro-racing’ or anterograde ‘push forward’ injection
the substance is injected as the needle is withdrawn or pushed forward in a straight fashion
-
droplet technique (also known as point technique, serial/ multi-puncture techniques and others)
small droplets of the filling substance are placed at a short distance from each other
usually in the treatment of surface areas
produces a superficial augmentation
-
stretching technique (modification of the droplet technique)
the skin is stretched to influence substance distribution e.g. fine lines on the forehead
Location
Typical locations of facial dermal injections include the following:
perioral region (superior and inferior jowl)
lips
nasolabial folds
marionette lines
medial (malar fat pad) and middle superficial cheek fat
periocular area (superior, inferior and lateral orbital regions)
glabella (central forehead)
Injections can be also classified into superficial, medium, deep and ultra-deep injections in respect to their depth and whether the filler is applied into an intradermal, subdermal, subcutaneous or subperiosteal location. Different depths have different indications and require different fillers. As a rule of thumb, deeper injections may benefit from a filler featuring a higher degree of crosslinking, viscosity and reabsorption time.
Complications
Complications of cosmetic facial intradermal injections can arise from the injection procedure or the substance and include the following 1-4:
hypersensitivity
erythema/bruising
edema
infection/cellulitis
-
vascular complications (rare)
soft tissue necrosis
blindness
overfilling
filler migration (spontaneous displacement of filling material)
abscess formation (e.g. PAAG)
non-inflammatory nodules (more common with non-resorbable fillers)
scarring
The most common complications are erythema, bruising and hypersensitivity do not require a radiological evaluation and also most nodules resolve spontaneously 1. Also, most fillers are accompanied by a mild degree of inflammation, which is usually transient 1,3.
Overfilling is a procedural complication and can create lumps and patient dissatisfaction, but can be detected on imaging and treated with needle aspiration or hyaluronidase if hyaluronic acid is the causative agent 1-3. Filler migration also might lead to cosmetically unsatisfactory results and can be treated similarly with hyaluronidase, corticosteroid injections or filler removal 3.
Occasionally, delayed reactions to injected fillers can occur. These reactions can resemble granulomas, however both the time course and the delayed gadolinium enhancement should suggest a delayed reaction.
Radiographic features
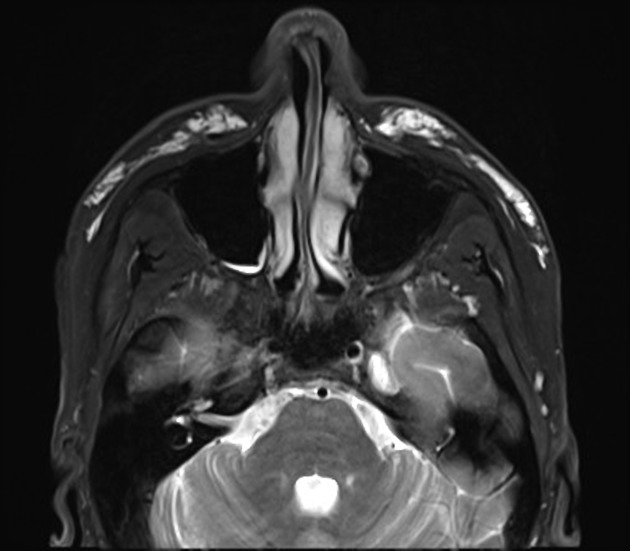
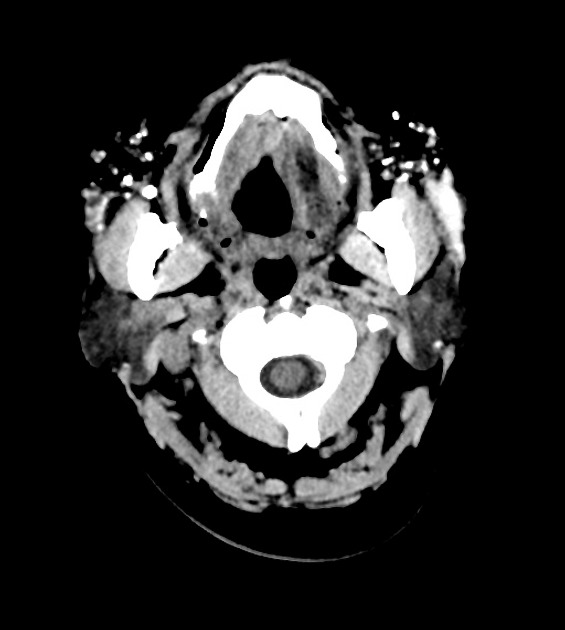
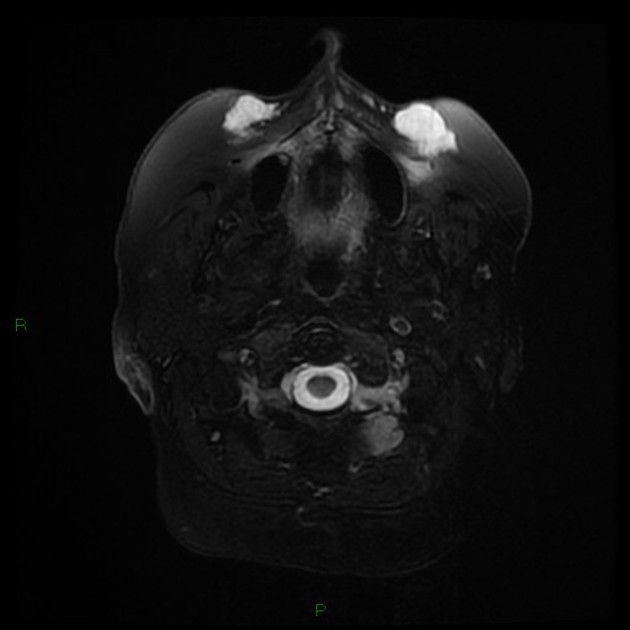
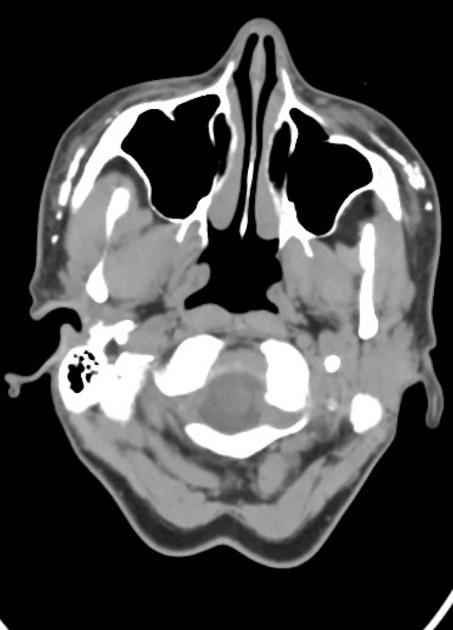
The result of facial dermal injections or injectable facial fillers is seen as incidental findings on imaging, including CT, MRI, and PET-CT. They can usually be recognized by their appearance and distinctive anatomical distribution but can act as confounding factors in the assessment and staging of facial, orbital and cutaneous malignancies 1.
Ultrasound
Ultrasound is a common and widely available method for the evaluation of facial fillers as well as their most common superficial complications 1. Liquid silicone typically exhibits a hyperechoic ‘snowstorm’ appearance.
CT
CT can nicely detect and depict calcifications, which can be seen in certain dermal fillers and certain complications. Imaging findings vary with the type of dermal filler.
Most fillers will appear as fluid density (collagen, PAAG) or soft tissue density (HA, PLLA) areas on CT images with or without fat stranding. Silicone might appear a bit denser than other fillers.
Autologous fat will characteristically exhibit fat density.
Calcium hydroxyapatite features hyperdense, calcified linear streaks or rounded masses. Polytetrafluoroethylene appears as linear hyperdensity.
Paraffinomas are characterized by soft tissue nodules with a calcified rim or simple calcific foci.
MRI
Due to its superior soft tissue discrimination capabilities, MRI is a convenient modality for the evaluation of injectable facial fillers, their complications including displacement.
A basic protocol might consist of T2 weighted images with and without fat saturation, diffusion-weighted imaging as well as pre and postcontrast T1 weighted images 1,3. Alternative T1 and T2 weighted DIXON images could be included 2. In case the injectable facial filler is not known the protocol will certainly benefit from a ‘silicone only’ sequence 1-3.
The imaging findings will depend on the type of dermal filler 1-3:
Autologous fat fillers are characterized by fat-signal intensity on all sequences often accompanied by a thin pseudocapsule.
The biological fillers collagen and hyaluronic acid (HA), as well as the synthetic polyalkylimide and polyacrylamide hydrogels (PAAG), feature a high water content or water-binding capabilities and thus are hypointense on T1 weighted images and hyperintense on T2 weighted images and are difficult to differentiate on MRI 1; however, PAAG are more isointense on T1 and T1 with fat suppression sequences than the very T1 hypointense HA2.
Calcium hydroxyapatite returns an intermediate to hypointense signal on both T1-weighted and T2-weighted images, with mild gadolinium enhancement. Polytetrafluoroethylene features similar signal characteristics but should have a more linear appearance.
Poly-l-lactic acid (PLLA) is hypointense on T2 weighted images.
Silicone can be visualized and differentiated from other substances with a ‘silicone only’ sequence.
Nuclear medicine
PET-CT / PET-MRI
Dermal fillers are associated with increased uptake of fluorine-18 fluorodeoxyglucose (FDG), which is a typical pitfall. Especially calcium hydroxyapatite and poly-L-lactic acid are known for FDG uptake 1.
Additionally, complications of facial fillers like inflammation, abscess formation and foreign body granuloma might show increased uptake of FDG 1.
Radiological report
The radiological report should contain a description of the following:
type and location of the dermal filler
suspected complications
overfilling (contour abnormalities, asymmetry, bulges)
foreign body granulomas (eggshell calcifications on CT)
abscesses (a central fluid signal with peripheral enhancement ± diffusion restriction)
filler migration
differential diagnosis (in unclear situations)
Differential diagnosis
Dermal fillers can usually be recognized by their typical anatomical location. Therefore lesions in atypical locations should raise suspicion especially if they show a restricted diffusion. Caution should be used when ascertaining the type of substance injected, especially if the patient was treated with injections from a less-than-reputable source.
Furthermore, dermal filler cases can be highly litigious, especially in the case of complications. Therefore, it is good practice to review the images with the referring physician/ plastic surgeon before deciding on the wording of the report.
Conditions that may mimic the appearance of dermal fillers or their complications include 1:
orofacial tumors
Outcomes
Autologous fat, collagen fillers and hyaluronic acid belong to the rapidly resorbable fillers and usually last a few months up to a year. Slowly resorbable fillers include calcium hydroxyapatite (CHA), poly-L-lactic acid (PLLA) which show effects up to 2 years.
Liquid silicone and polymethylmethacrylate (PMMA) microspheres are non-resorbable or permanent synthetic fillers; the latter has also been combined with collagen for longer effect 1.




 Unable to process the form. Check for errors and try again.
Unable to process the form. Check for errors and try again.