Myocardial mapping or parametric mapping of the heart is one of various magnetic resonance imaging techniques, which has evolved and been increasingly used in the last decade for non-invasive tissue characterization of the myocardium 1-5. Unlike normal T1-, T2- or T2*- images, parametric mapping allows for both visualization and quantification of focal and diffuse myocardial disease 1,2,4.
On this page:
Clinical applications
There is proven clinical utility in a setting of myocarditis, amyloidosis 1,6, Anderson-Fabry disease 1,6 and iron deposition and potential clinical utility regarding many cardiac diseases such as cardiomyopathy, heart failure, congenital heart disease, myocardial ischemia and infarction, transplant rejection athlete’s heart and cardiac masses 1,3,6.
Detailed recommendations for indications, utility and clinical implementation including imaging protocols, the use of reference ranges and reporting are given in a consensus statement by the Society for Cardiovascular Magnetic Resonance (SCMR) 1.
Technique
The technique comprises the generation of a parametric map from a series of co-registered images with different T1, T2 or T2*relaxation times, depending on the mapping technique used. The T1, T2 or T2* values of the myocardium are tissue properties and are encoded in the respective pixels of the map. With the mapping technique, they can be compared to the spared healthy myocardium in focal disease or quantified and evaluated based on normal reference values in diffuse disease 1,5,22.
The T1-map is generated at different degrees of longitudinal relaxation to receive a signal intensity vs time curve, from where T1 can be calculated. Similarly, T2 or T2*-maps are obtained from a signal intensity vs time curve based on different transverse relaxation times. Different acquisition methods for each T1 and T2 quantification exist 7-9,15-17.
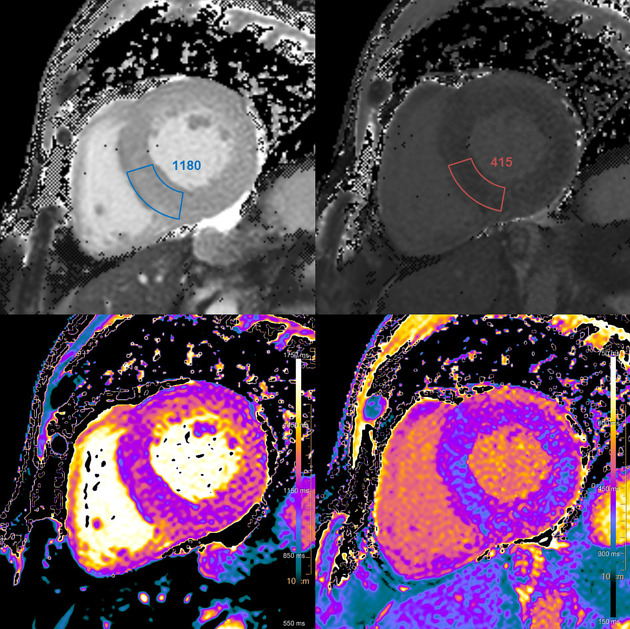
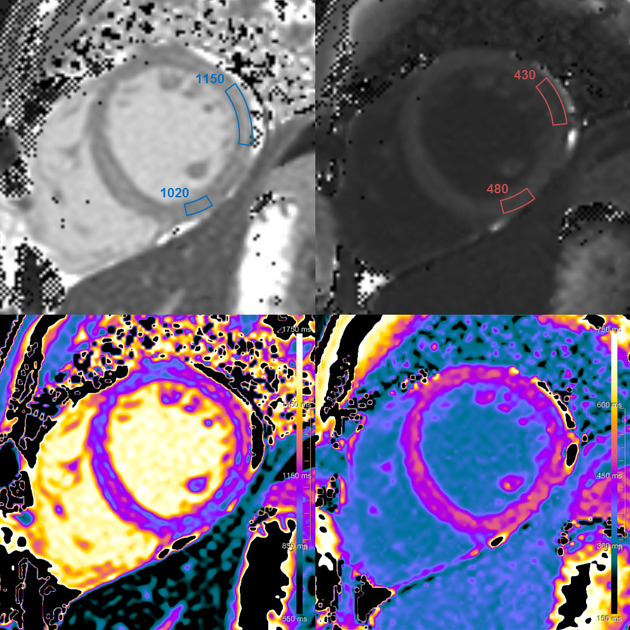
Myocardial T1 mapping
Native T1
The myocardial T1 map depicts the spin-lattice (longitudinal) relaxation of the myocardium and the T1 value [ms] of a voxel represents the respective time constant for the recovery.
Despite extensive research during the last 15 years, histopathological correlates of myocardial T1 values are still not completely elucidated 2. T1 mapping techniques have been studied as a key feature for the assessment of myocardial fibrosis 5,9,10,13,22. However, native T1 reflects changes in intracellular and extracellular compartments and is not only influenced by collagen, but also by protein, water (edema), lipids and iron 1-6,9,10,22.
Myocardial T1 lengthens at higher magnetic field strength3,8 and is also influenced by various physiological factors 4,8.
Native T1 mapping has been applied to investigate most diffuse cardiac disorders as diffuse fibrosis, scar tissue, and inflammation that result in prolonged native myocardial T1 relaxation time1-6,9-14.
Postcontrast T1
Administration of gadolinium leads to a shortening of the T1 time. Postcontrast T1 can be measured and displayed on a map in equal measure. Since most gadolinium agents spread in the intravascular and interstitial compartments and not within the cells, a reduced post-contrast T1 value should either reflect an expansion of the interstitial space due to collagen (myocardial fibrosis), protein (amyloidosis) or access to the intracellular space (in case of myocardial edema or cell membrane disruption e.g. necrosis, myocardial infarction) 1,2,6,9,10,14,22.
Myocardial extracellular volume (ECV)
Extracellular volume (ECV) reflects the volume fraction that is not taken by cells. It combines interstitial and intravascular space and represents approximately 25% of the myocardial volume 6. If native and post-contrast T1 images are co-registered, quantified and adjusted for hematocrit, extracellular volume(ECV) can be calculated for a specific area or specific voxels 1,2. Likewise, an ECV map can be generated. For this purpose postcontrast T1 should be obtained during a slow continuous infusion or at least 15 min after bolus injection 6.
Assessment of extracellular volume (ECV) is considered reasonable in patients getting an extracellular contrast agent1. Myocardial fibrosis can lead to an increase of ECV 1,4,13, as well as disorders affecting the myocardium such as cardiac amyloidosis or myocarditis. On the other hand, athlete's heart for example can lead to a decrease in extracellular volume (ECV) 1.
Myocardial T2 mapping
The myocardial T2 map depicts the spin-spin (transverse) relaxation of the myocardium and T2, encoded in the voxels of the map, is a time constant for the decay of transverse magnetization.
T2 time is related to the water content of the respective tissue, hence the myocardium and thus prolonged T2 reflects myocardial edema, be it due to inflammation or reperfusion 20.
T2 mapping of the myocardium offers the potential for more objective detection and quantification of myocardial edema than standard black-blood T2 and STIR images, which are often of limited value due to susceptibility or slow flow artifacts 20. Myocardial T2 tends to decrease at higher magnetic field strength 17.
Myocardial T2 mapping can detect edematous myocardial territories in diverse cardiac conditions as myocarditis, takotsubo cardiomyopathy or myocardial infarction. Conversely, low T2 values will be seen in iron overload and hemorrhage 1.
Myocardial T2* mapping
T2*-mapping is usually based on gradient echo (GRE) sequences, T2* results principally from variations in the static magnetic field throughout the tissue which will be added to random mechanisms. Thus T2* is a time constant for the decay of transverse magnetization, it is only shorter 26.
T2* relaxation time is sensitive to magnetic field inhomogeneity and has become useful in the evaluation of myocardial iron overload and assessment of chelation therapy 3,23-25 and also shows a decrease in myocardial hemorrhage or stress-induced ischemia 1,3.
Interpretation
Normal values
Normal values of native T1 and T2 times differ depending on magnetic field strength (1,5 and 3 T), acquisition sequence (e.g. MOLLI, shMOLLI, SASHA, SAPPHIRE in case of T1-mapping and T2-SSFP, GraSE in case of T2-mapping). Because of variations between scanners, it is still recommended 1,2,11, that local reference ranges are primarily used, and if a local reference range is not available quantitative results should not be clinically reported 1,2.
Extracellular volume reference ranges from the literature seem to be considered acceptable, if the same MR system and pulse sequences are used 1.
Regarding T2*and iron overload, a 3-tier risk model (low, intermediate and high risk) has been recommended 1,25.
Cardiac magnetic resonance fingerprinting (cMRF)
To overcome the problem that conventional mapping techniques require separate acquisitions for T1 and T2 mapping, of which values are also dependent on different scanner technology, pulse sequences, and various physiological and environmental parameters, cardiac magnetic resonance fingerprinting (cMRF) technique has been introduced for simultaneous measurement of T1 and T2 maps in a single scan 27,28.




 Unable to process the form. Check for errors and try again.
Unable to process the form. Check for errors and try again.