Raynaud phenomenon, also known as Raynaud syndrome, describes a localized vasculopathy whereby there is an exaggerated vascular response to cold temperature or emotional stresses.
On this page:
Terminology
Raynaud phenomenon is classified as being either 'primary' or idiopathic, or 'secondary' to another underlying condition (as discussed below) 1-8. The diagnosis of Raynaud disease often refers to a primary Raynaud phenomenon.
Clinical presentation
Raynaud phenomenon most commonly affects the fingers, but can also affect the toes, knees, nipples, and tips of the ears, nose or tongue 1-3. It is characterized by sudden episodes of vasoconstriction followed by reperfusion, usually in response to cold temperatures or emotional stress, causing a classic change of colors in the extremities involved 1-3.
Clinically, the fingers (or other extremities) suddenly become cold and have pallor ('white'), later develop cyanosis ('blue'), and then become erythematous ('red') 1-3. These episodes tend to last up to 20 minutes in duration and often start with one digit before symmetrically involving other digits 1. Interestingly, involvement of the thumb only commonly occurs in secondary Raynaud phenomenon 4.
In addition to the classic 'white-blue-red' color changes, the vascular changes in Raynaud phenomenon can also cause episodic ischemic pain, episodic peripheral neuropathy, and even digital ulceration and critical tissue ischemia 1-3. These more severe clinical features are more common in secondary Raynaud phenomenon.
Pathology
The pathogenesis of Raynaud phenomenon has not been completely elucidated 1. It is thought that in patients with Raynaud phenomenon, there is a heightened sympathetic response to triggers such as cold temperatures 5. Under normal circumstances, cold temperatures trigger a sympathetic response 5. Extremities such as fingers and toes have a very rich sympathetic supply, but also have a unique vascular structure 5. Such extremities have a high density of arteriovenous anastomoses which play a key role in thermoregulation 5. In Raynaud phenomenon, these arteriovenous anastomoses constrict to a supraphysiological level, due to an exaggerated sympathetic activation, resulting in the aforementioned clinical features 5.
Etiology
There are a large number of conditions potentially underlying secondary Raynaud phenomenon, such as 1,12,13:
connective tissue disorders, vasculitides or other inflammatory disorders: e.g. systemic sclerosis (including CREST syndrome), systemic lupus erythematosus (SLE), Buerger disease, anti-synthetase syndrome
atherosclerosis
arterial trauma
dysautonomia: e.g. multiple system atrophy
drugs: e.g. ergot derivatives
Radiographic features
Although Raynaud phenomenon is primarily a clinical diagnosis, certain imaging modalities may be useful in supporting its diagnosis.
Plain radiograph
Plain radiograph of the fingers may reveal acro-osteolysis 6.
Ultrasound
Doppler ultrasound offers a non-invasive visualization and assessment of digital vasculature 7. Patients with Raynaud phenomenon often demonstrate flow volume and vessel size irregularities 7. This modality is not as sensitive as DSA or MR angiography to vascular changes of Raynaud phenomenon 7.
MRI
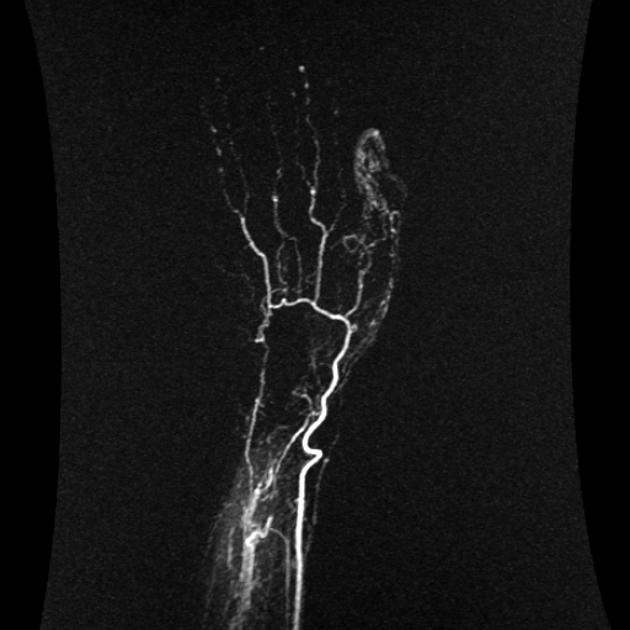
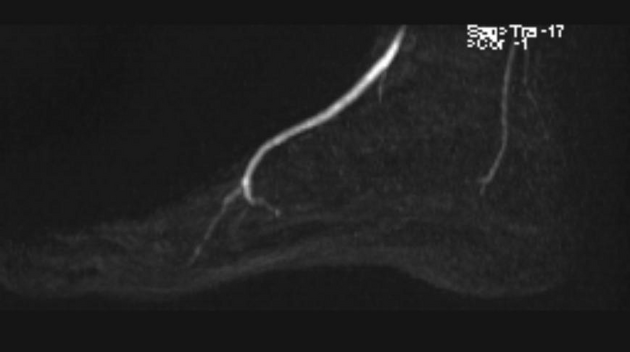
Contrast-enhanced MR angiography may reveal characteristic narrowing and tapering of digital vessels 8. In comparison to DSA, MR angiography has less spatial and temporal resolution but has the advantage of being relatively non-invasive 8.
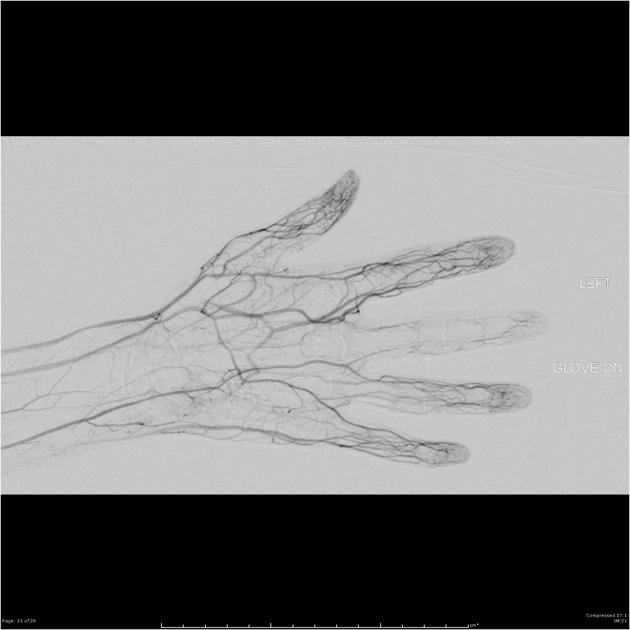
Angiography (DSA)
DSA is considered the gold-standard imaging modality for visualization of extremity vessels in Raynaud phenomenon due to its high spatial and temporal resolution 8,9. Classically, and similarly to MR angiography, DSA will reveal narrowing and tapering of affected digital vessels 8,9.
Treatment and prognosis
There are numerous pharmacological and non-pharmacological options available to patients with Raynaud phenomenon 1-3.
Patient education is the cornerstone of management, with patients being advised to avoid cold exposure, maintain the warmth of the whole body (i.e. not just wearing gloves and socks), cease smoking, avoid sympathomimetic medications, and avoid emotional stress where possible 1-3.
Pharmacologically, dihydropyridine calcium channel blockers are often considered first-line options, followed by phosphodiesterase inhibitors, topical nitrates, and prostacyclin analogs in treatment-resistant cases 1-3.
History and etymology
This condition was first described by French physician Maurice Raynaud (1834-1881) in 1862 1,10, however, the eponym 'Raynaud phenomenon' was first coined by Sir Jonathan Hutchinson (1828-1913), an English physician and surgeon, over thirty years later in 1893 1,11.




 Unable to process the form. Check for errors and try again.
Unable to process the form. Check for errors and try again.