Hypertrophic cardiomyopathy (HCM) is a type of cardiomyopathy defined by left ventricular hypertrophy which cannot otherwise be explained by another cardiac or systemic disease. It is the leading cause of sudden cardiac death in infants, teenagers, and young adults.
On this page:
Terminology
Although hypertrophic cardiomyopathy can generally describe a hypertrophied and non-dilated left ventricle due to any cause, this article focuses on hypertrophic cardiomyopathy in the absence of another systemic or cardiac disease.
Epidemiology
There is a slight male prevalence and is present in 1 out of 500 in the general population, probably remaining undiagnosed and asymptomatic in the majority of affected individuals 15.
Associations
-
LVOT obstruction is present in 70% of cases 12, it is defined as a gradient >30 mmHg
a gradient >50 mm Hg is often considered the threshold of hemodynamic significance 13
-
often 'obstructive hypertrophic cardiomyopathy' is used when LVOT obstruction is present
most commonly occurs at the basal interventricular septum
with systolic anterior motion (SAM) of the chordal apparatus, the dynamic obstruction may occur deeper within the left ventricle
hypertrophic papillary muscles may cause obstruction in the mid-cavity
-
can be associated with systolic anterior motion (SAM) of the anterior mitral leaflet, which can increase LVOT obstruction and decreased coronary and systemic outflow
systolic anterior motion of the posterior leaflet, less commonly, may also occur
-
other secondary signs include:
-
leaflet accessory tissue and elongation may also be observed
left auricle dilation
-
papillary muscle abnormalities
hypertrophy
direct insertion to anterior leaflet of mitral valve
antero-internal displacement
-
Diagnosis
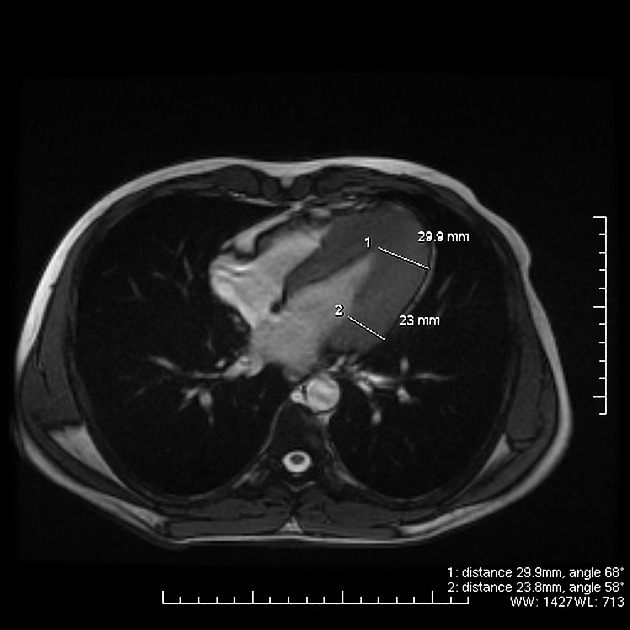
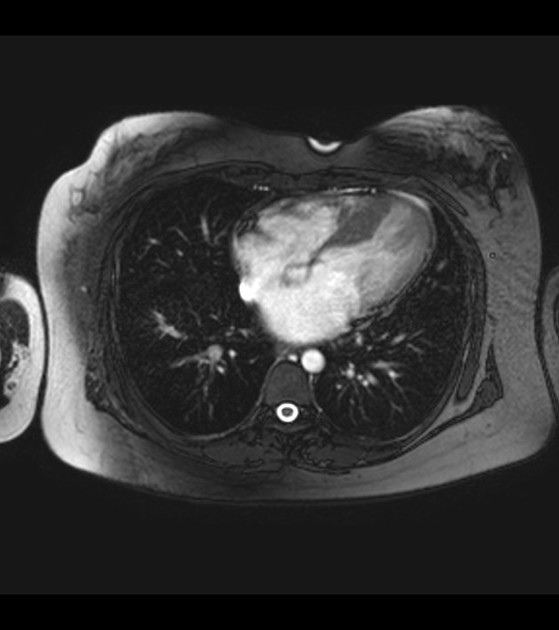
The clinical diagnosis can be made by imaging modalities such as echocardiography or cardiac MRI by the demonstration of a maximal left ventricular wall thickness of ≥15 mm in the absence of any other causes of left ventricular hypertrophy 17.
Clinical presentation
Patients present with symptoms and signs of left-predominant congestive heart failure. Patients with a left ventricular outflow tract (LVOT) obstruction may have additional signs such as an ejection systolic murmur that is classically louder with the Valsalva manoeuver.
The electrocardiogram is abnormal in over 95% of cases; the most common abnormalities include high precordial QRS voltages, secondary repolarization abnormalities (ST-segment depression, T wave inversion), left axis deviation, and deep, narrow so-called "needle-like" q waves, typically in leads I, L, V5 and V6.
Pathology
Hypertrophic cardiomyopathy is characterized by left ventricular hypertrophy (wall thickness >12-15 mm; normal wall thickness is 12 mm or less, measured during diastole) without obvious etiology. Associated right ventricular hypertrophy may be seen in 15-17% of cases.
Genetics
Hypertrophic cardiomyopathy is an autosomal dominant genetic disorder with incomplete penetrance involving the cardiac sarcomere. Mutations in a group of related genes that make up the cardiac sarcomere are found in up to 60% of individuals with a family history of HCM and 30% of those without a family history. Commonly affected genes include 12:
MYBPC3 (myosin binding protein): 30%-40%, chromosome 11
MYH7 (myosin heavy chain): 20%-30%, chromosome 14
TNNT2 (cardiac muscle troponin): ~10%, chromosome 1
TNNI3 (troponin I type 3): ~7%, chromosome 19
MYL2 (myosin light chain 2): ~4%, chromosome 12
MYL3 (myosin light chain 3): ~2%, chromosome 3
TPM1 (tropomyosin 1): ~1%, chromosome 15
Subtypes
Morphologically there are several recognized subtypes or phenotypes of hypertrophic cardiomyopathy. It may be classified as 4,12:
-
asymmetric hypertrophic cardiomyopathy
most common morphologic variant, roughly 60-70% of cases
-
disproportionate septal hypertrophy most frequently involving basal anteroseptum and adjacent basal anterior segments
resultant narrowing of the anatomic left ventricular outflow tract may create substrate for dynamic obstruction
-
suggestive imaging features include:
-
an interventricular septal wall thickness ≥15 mm
greater than two standard deviations above mean (pediatrics)
septal to inferolateral wall thickness ratio exceeding 1.3-1.5
-
-
symmetrical or concentric hypertrophic cardiomyopathy
-
second most common variant; characterized by diffuse left ventricular wall thickening with an associated decrease in left ventricular cavity size
requires exclusion of conditions associated with increased afterload such as hypertension and valvular aortic stenosis
physiologic hypertrophy (i.e. athlete's heart) also must be excluded
-
-
apical hypertrophic cardiomyopathy (Yamaguchi syndrome)
-
hypertrophy predominantly affecting apical segments imparting a characteristic 'ace of spades' appearance
may be "mixed" with concomitant hypertrophy of the septum or "pure" with isolated apical involvement 19
imaging features suggestive of the diagnosis include an apical wall thickness ≥15 mm and a ratio of apical to basal wall thickness > 1.5
-
-
mid-ventricular hypertrophic cardiomyopathy
less common variant (~10% patients)
-
hypertrophy predominantly affects the mid-cavity segments with a resultant "hourglass" or "dumbbell" shape
can lead to mid-cavity obstruction and an aneurysmal morphology to the LV apex
-
mass-like hypertrophic cardiomyopathy 4,12
alternatively tumefactive hypertrophic cardiomyopathy 8
focal thickening of an isolated myocardial segment mimicking a mass
cardiac CT (e.g. absence of calcification) and MRI are useful modalities in diagnosis
-
burned-out phase: end-stage
characterized by systolic dysfunction (LV-EF < 50%)
Another method is a four-pattern model 10:
septal hypertrophy alone ~45%
septal and other segments hypertrophy but sparing the apex ~ 16%
apical segments along with any other segment hypertrophy ~ 27%
apical hypertrophy alone ~ 13%
Radiographic features
Plain radiograph
Chest radiographic findings may be normal or reflect nonspecific features suggesting associated cardiogenic pulmonary edema such as:
Ultrasound
Echocardiography
Echocardiography is the initial and most common imaging modality used in the evaluation of hypertrophic cardiomyopathy due to its practical utility, availability, and high temporal resolution 12. It is used for the evaluation of LVOT obstruction, defining the presence and extent of left ventricular hypertrophy, assessing systolic and diastolic left ventricular function, identification of systolic anterior motion (SAM) of the mitral valve, and assessment of left atrial size. 2D echocardiographic findings supportive of the diagnosis include:
-
septal to inferolateral wall thickness ratio > 1.3
>1.5 in the presence of hypertension
any myocardial segment with an unexplained wall thickness > 15 mm
The temporal resolution of M-mode echocardiography has traditionally been utilized in conjunction with 2D imaging to enhance the detection of SAM of the mitral valve. Findings include:
-
anterior MV leaflet displaced into LVOT toward septum during ventricular systole
contact with the septum may occur in mid-systole when severe 30
-
concomitant "fluttering" or closure in mid-systole of the aortic valve leaflets
with "notching" of the corresponding M-mode tracing 19
Doppler echocardiography
In the presence of SAM of the mitral valve disrupting leaflet coaptation color flow Doppler (CFD) may demonstrate a posteriorly directed jet of mitral regurgitation. Aliased flow may also be noted in the LVOT in the presence of obstruction.
Spectral Doppler modalities are utile in the evaluation of the type, severity and anatomic location of obstruction:
-
pulsed wave Doppler (PWD) may be used to differentiate obstruction occurring at the mid-cavitary level, in the outflow tract, or at the level of the aortic valve by placing the "gate" and obtaining spectral envelopes from the corresponding locations
aliasing from high velocity flow is typical of obstruction
-
continuous wave Doppler (CWD) envelope morphology can suggest the nature (i.e. fixed versus dynamic) of the obstruction and may be used to quantify the peak gradient as a surrogate of hemodynamic significance
dynamic obstruction demonstrates an envelope with a gradual velocity increase in early systole followed by a sharp increase heralding the onset of obstruction with a late peak
a peak gradient (4 x peak velocity 2) of < 30 mmHg is unlikely to be hemodynamically significant
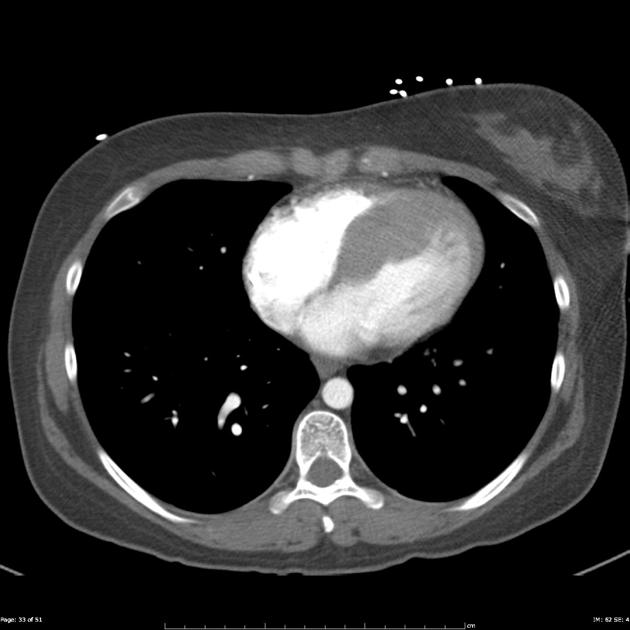
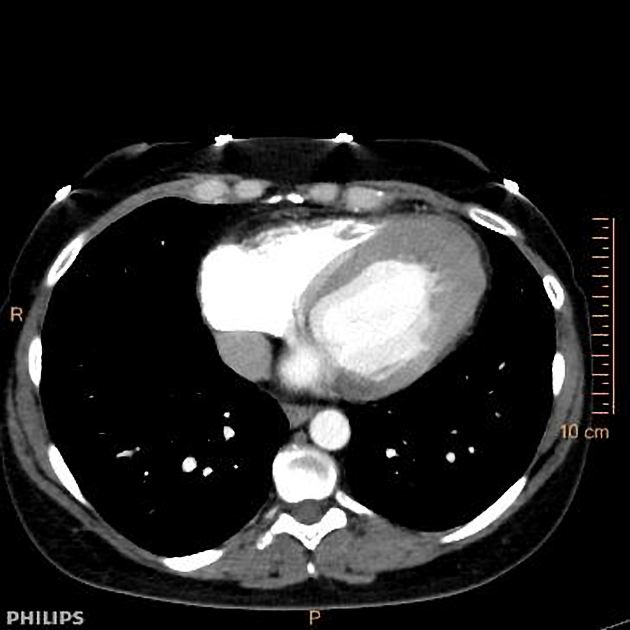
CT
ECG-gated cardiac CT is not routinely performed for hypertrophic cardiomyopathy. It does not provide information on fibrosis and flow dynamics. However, cardiac CT offers excellent spatial resolution and serves as an alternative modality in patients in whom MRI is contraindicated 12.
MRI
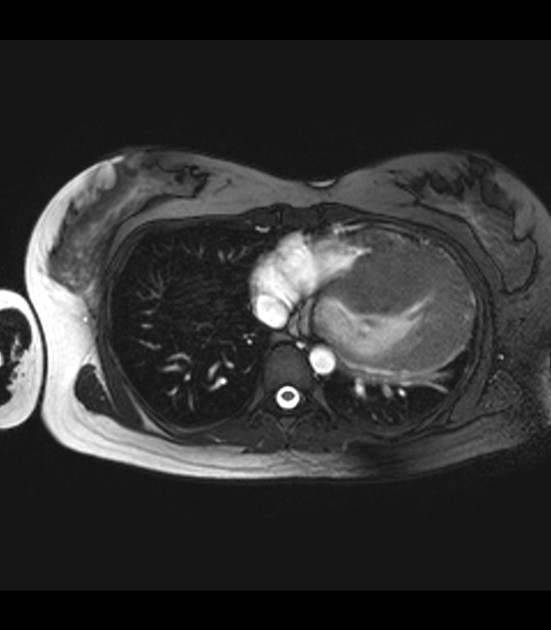
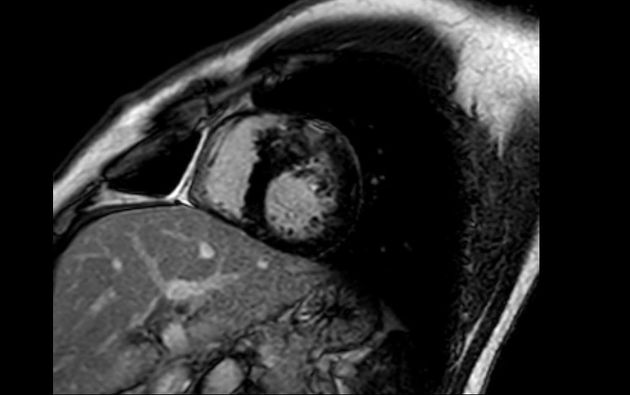
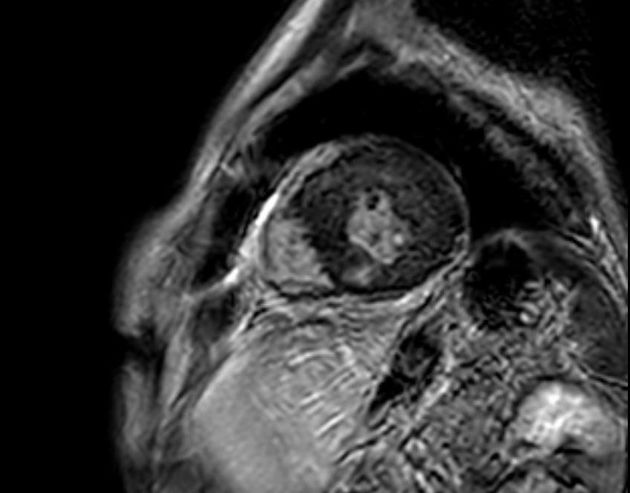
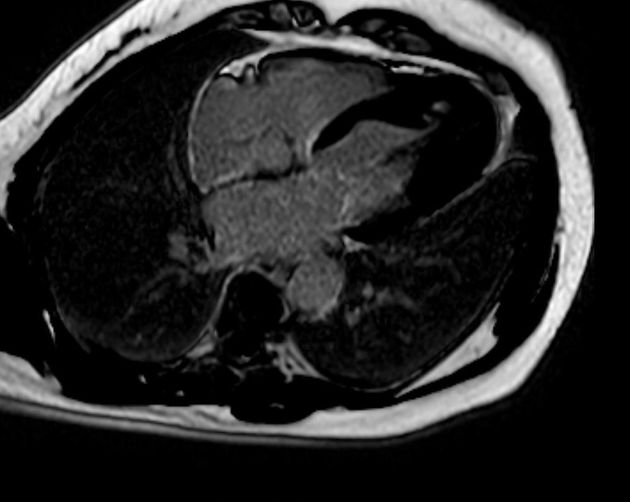
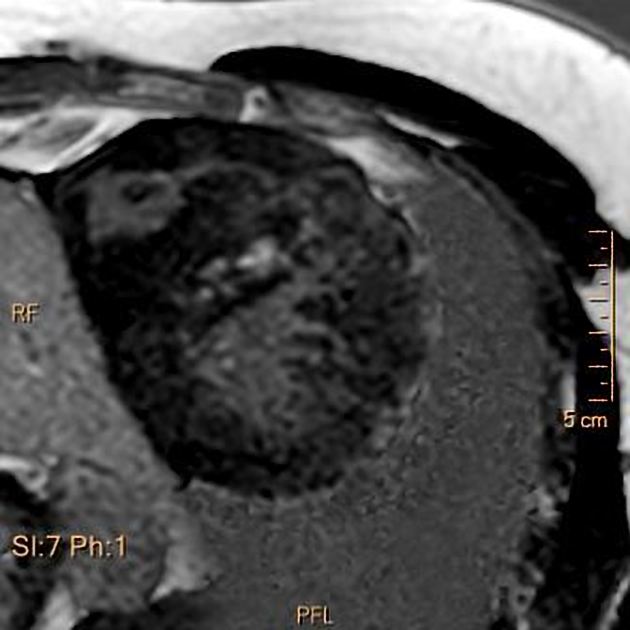
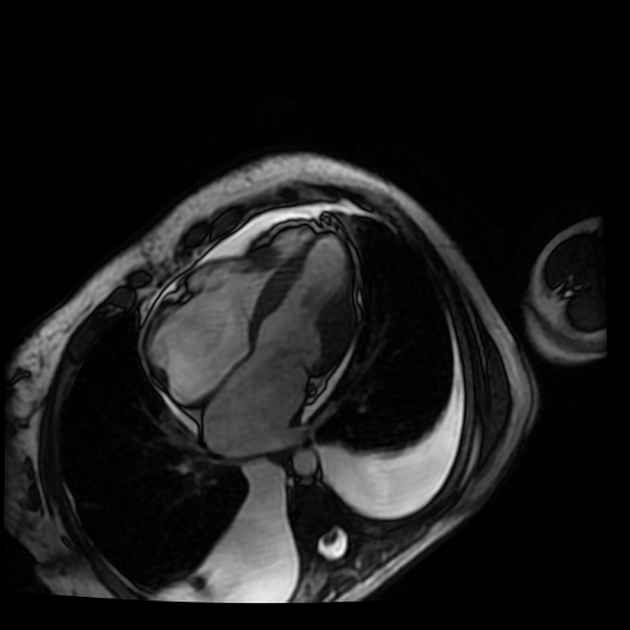
Cardiac MRI, with its capabilities in evaluating cardiac morphology and function, has emerged as a technique particularly well-suited to hypertrophic cardiomyopathy diagnosis and phenotypic characterization. It is superior to echocardiography in identifying areas of segmental hypertrophy not reliably visualized or underestimated by echocardiography (i.e. anterolateral and apical segments). Visualized features may include:
left ventricular systolic dysfunction
-
left ventricular hypertrophy
with or without right ventricular hypertrophy
predilection for the basal interventricular septum
-
systolic anterior motion (SAM) of the mitral valve
mitral regurgitation
left ventricular apical aneurysms
morphologic variations involving the mitral valvular apparatus (e.g. papillary muscles)
Cardiac MRI also has a role in asymptomatic HCM mutation carriers by identifying phenotypic markers of HCM in the absence of left ventricular hypertrophy including:
elongated mitral valve leaflets
-
late gadolinium enhancement (LGE)
patchy/streaky intramyocardial patterns at the right ventricular insertion sites within the hypertrophied myocardium suggest fibrosis
Another important role of CMRI is in differentiating the mass-like HCM variant from a discrete cardiac mass with the former suggested by the following features:
presence of contractility
iso-intense to myocardium on T1- and T2-weighted images
first-pass enhancement
patchy and midventricular type of delayed enhancement
Treatment and prognosis
Management is individualized and generally guided by features such as associated symptoms, degree of arrhythmogenesis, and the presence and severity of left ventricular outflow tract obstruction:
asymptomatic, low-risk patients may not routinely require specific therapy 27
patients with symptomatic heart failure and an absence of LVOT obstruction often receive standard medical therapy for congestive heart failure
-
patients with LVOTO and symptoms consistent with heart failure may be managed with cautious hemodynamic optimization, including:
avoidance of factors exacerbating the degree of obstruction such as intense exercise, hypovolemia, and certain medications (e.g. vasodilators, positive inotropes)
medications which decrease inotropy (e.g. beta blockers) and chronotropy are mechanistically favorable and commonly used 22
-
select patients (e.g. with refractory symptoms) may require surgical or interventional management, examples of which include:
transaortic or transapical septal myectomy 29
alcohol septal ablation
mitral valve plication
patients with (or at high risk of) ventricular dysrhythmias and the attendant risk of sudden cardiac death (SCD) may be managed with pharmacologic (i.e. anti-arrhythmic medications) measures or placement of an implantable cardioverter-defibrillator (ICD) 20
Prognosis
While the majority of patients with hypertrophic cardiomyopathy have a normal life expectancy significant morbidity and mortality may occur secondary to heart failure and sudden cardiac death. Mortality typically occurs from ventricular dysrhythmias (e.g. ventricular fibrillation).
Historical features which may suggest a poor prognosis may include:
previous episodes of ventricular dysrhythmias with or without associated syncope or cardiac arrest 22
family history of sudden cardiac death
Multimodality imaging features associated with a poor prognosis may include the following:
-
echocardiography
severe left ventricular diastolic dysfunction 26
markedly elevated LVOT peak gradient (>80 mmHg)
severe left ventricular hypertrophy
-
cardiac MRI 12
-
extent of late gadolinium enhancement exceeding 15%
marked LGE heterogeneity, semi-quantified by dispersion mapping, may also have prognostic value 25
left ventricular wall thickness ≥ 30 mm
left ventricular apical aneurysms
decreased left ventricular ejection fraction (<50%)
-
History and etymology
Hypertrophic cardiomyopathy was first described and originally termed idiopathic hypertrophic subaortic stenosis (IHSS) by Brent et al. in 1960 11,12. The term hypertrophic cardiomyopathy (HCM) came into common use in the latter half of the 1980s replacing IHSS, which was considered an inappropriate characterization.
Differential diagnosis
-
most common cause of concentric LV hypertrophy
-
suggestive features include a maximal wall thickness usually 13-15 mm, normal diastolic function, left ventricular dilation, and resolution of hypertrophy with exercise cessation
-
valvular aortic stenosis
suggestive cardiac MRI features include a restricted aortic leaflet excursion with elevated transvalvular gradients
echocardiographic features may include valvular structural abnormalities (e.g. calcification, decreased leaflet mobility), pulsed wave Doppler localizing obstruction to the valve, and a continuous wave Doppler envelope consistent with fixed obstruction (parabolic rise with an earlier peak)
-
associated with skeletal myopathy and mild intellectual disability
Dynamic left ventricular outflow tract obstruction (LVOTO) may occur with a variety of unrelated anatomic and physiologic substrates including:
distributive shock
myocardial ischemia of the left anterior descending artery (LAD) territory 21
sigmoid septal hypertrophy14




 Unable to process the form. Check for errors and try again.
Unable to process the form. Check for errors and try again.