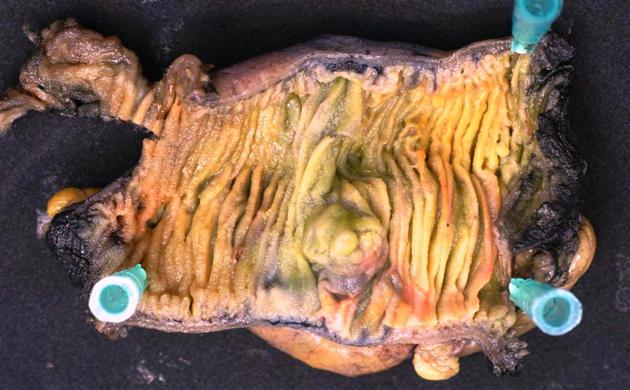
The small intestine is rarely the site of malignant tumors, although it accounts for ~75% of the entire length of the gastrointestinal tract and >90% of the mucosal surface. Approximately 40 different histologic tumor types have been described.
In this article, an overview will be given of the most common benign and malignant types of small bowel tumors, including their imaging characteristics.
On this page:
Epidemiology
Primary neoplasms of the small intestine are very rare, comprising approximately ~4% (range 1.6-6%) of all tumors of the gastrointestinal tract 1,2. Approximately 60% of these tumors are malignant. Benign neoplasms of the small intestine comprise ~5% of benign tumors of the intestinal tract. Most present in the 5th and 6th decades 1.
The most frequent malignant tumors are 1,2:
adenocarcinoma: 30-50%
carcinoid tumor: 25-30%
leiomyosarcoma: 10%
lymphoma: 15-20%
The most frequent benign tumors are 1,2:
adenoma: 25%
Metastases are more common in the small intestine than primary tumors and may occur by hematogenous spread, local extension or intraperitoneal seeding 1.
Clinical presentation
The clinical manifestations are non-specific and can include:
nausea and vomiting
melena
Tumor-related obstructions do not produce symptoms until a late stage, due to the fluid contents of the small intestine. Imaging at that time, usually, documents an advanced tumor stage with a correspondingly low 5-year survival rate (<20%).
Distribution
Can involve the duodenum, proximal jejunum and distal ileum 1,2. The duodenum is most at risk for adenocarcinomas and the ileum for carcinoid tumors and lymphomas 2.
Pathology
Numerous pathogenetic mechanisms have been mentioned regarding the low susceptibility of the small bowel to malignant change. The most cited are 2:
rapid transit of intestinal contents (short exposure of the mucosa to carcinogens)
moderation of mucosal irritation by the liquid nature of the small bowel contents
low bacterial load
relatively high concentration of lymphoid tissue
Some inflammatory disorders predispose to malignancy 1,2:
celiac disease: lymphoma
Crohn disease: adenocarcinoma, 5-20 x increased risk
immunoproliferative small intestinal disease (IPSID, Mediterranean lymphoma or diffuse small intestinal lymphoma)
Various genetic disorders are also associated with an increased incidence of small bowel tumors 1,2:
Peutz-Jeghers syndrome: hamartomatous polyps
Gardner syndrome: adenoma, adenocarcinoma
familial adenomatous polyposis coli: adenoma, adenocarcinoma
von Recklinghausen disease: paraganglioma
Radiographic features
Gastric and colonic endoscopy, ultrasound and small bowel barium examinations are the first-line studies for all non-specific clinical GI symptoms.
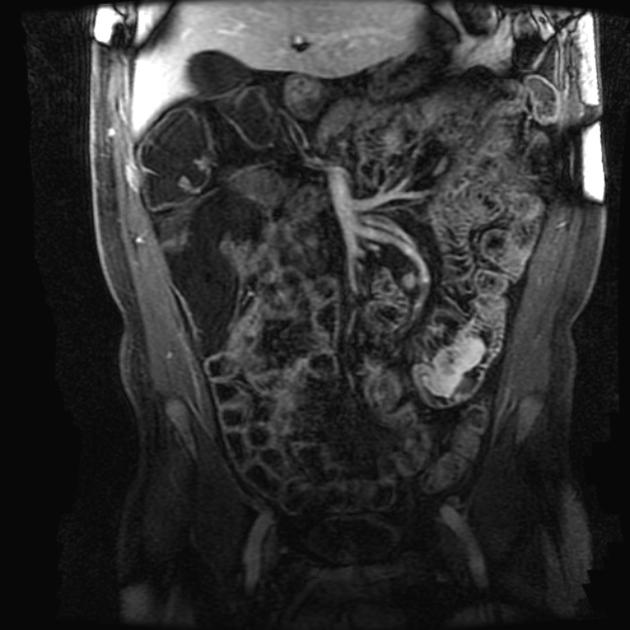
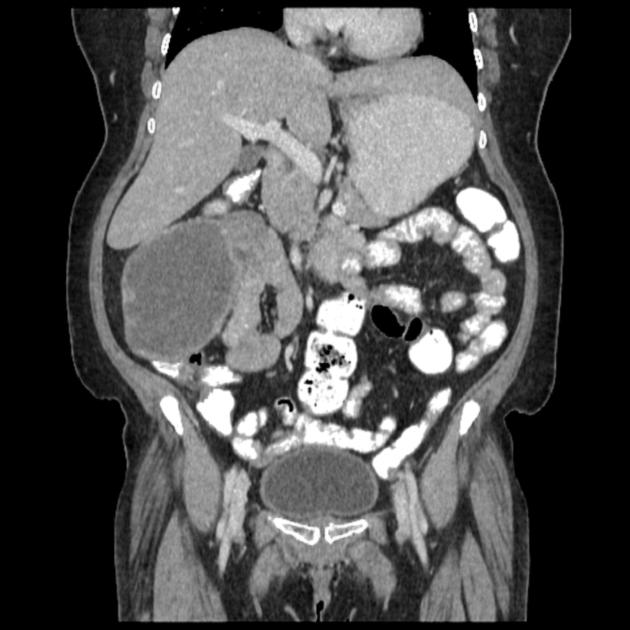
Enterography (and enteroclysis) with CT and especially MRI have become important in the investigation of GI disorders. These techniques are superior to enteroclysis in the evaluation of eccentric tumors and secondary lesions (peritoneal carcinomatosis).
With MRI, the spatial resolution is less than that achieved with conventional enteroclysis or CT, therefore it is less useful for evaluation of lesions <5 mm.
Endoscopic ultrasonography (EUS) is very sensitive and effective for the workup of neoplasms in the ampullary region.
CT angiography may be useful for demonstrating the site of bleeding and the responsible lesion.
Capsule endoscopy has facilitated early diagnosis of small bowel tumors 2.
Benign tumors
-
1-10 cm in size
mostly located in the jejunum
typically rounded, well-circumscribed
frequently ulcerate and cause bleeding
obstructive in case of intraluminal growth
irregular lesion margins and enlarged lymph nodes should raise suspicion of leiomyosarcoma
angiography: hypervascular lesion
-
MRI
intermediate signal on T1 and slightly increased on T2
moderate, homogeneous contrast enhancement
tumor necrosis and calcifications can be present
MRI is better than enteroclysis for showing extraluminal growth patterns
-
1-3 cm in size
predominantly in the duodenum, also in the ileum (ileocecal valve)
one-third occurs in the setting of Peutz-Jeghers syndrome
large polyps may cause intussusception
enteroclysis:
-
mostly located in the ileum (ileocecal valve)
MRI: typically high signal on T1 and loss of signal with fat saturated images
no contrast enhancement with CT/MRI
-
predominantly in jejunum
may cause severe GI bleeding
cavernous types resemble a submucosal polyp
capillary types are multiple and may appear as small, flat, inconspicuous filling defects on enteroclysis
-
MRI
typically show high T2 signal and intense enhancement
smaller lesions can be missed on MRI, mesenteric angiography may be performed in cases with occult hemorrhage
Malignant tumors
-
30-45% occurs in the appendix, 25-35% in the small intestine (90% in distal ileum)
characterized by increased serotonin production
typically invasive growth and pronounced perifocal fibrotic reaction (desmoplastic reaction)
features: muscularis propria thickening, puckering, wall retraction, serosal invasion, mesenteric metastases (similar echogenicity to the primary tumor, may calcify)
-
MRI
T1: isointense to bowel wall
T2: iso- to hypointense
extraluminal desmoplastic streaks (spoke-wheel pattern), mesenteric retraction and kinking of bowel loops
encasement of mesenteric blood vessels may lead to chronic ischemia
CT: dystrophic calcifications can be seen (better with CT than MRI)
hypervascular carcinoids are typically demonstrated in the arterial phase
ultrasound: smooth, intraluminal, hypoechoic masses interrupting the submucosal layer 3
-
small bowel adenocarcinoma 1,2
most common GIT malignancy 3
mostly located near the papilla in the duodenum, less frequently in jejunum
enteroclysis: infiltrative tumor with irregular and segmental luminal narrowing
MRI: non-specific intermediate SI on T1 and T2
asymmetrical nodular wall thickening
non-homogeneous moderate contrast enhancement
streaky infiltration of the mesentery indicates tumor spread
regional mesenteric lymphadenopathy
US: similar to other bowel tumors, hypoechoic, loss of gut signature, obstruction 3
-
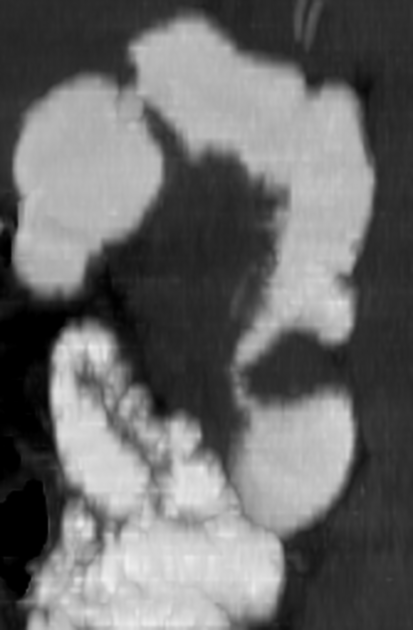
small bowel leiomyosarcoma 1,2
may occur anywhere in the small bowel, mainly ileum and jejunum
large tumors, usually >6 cm, with extraluminal growth
imaging findings are comparable to leiomyoma (see above), and the differentiation can be difficult
frequently ulcerate and cause bleeding
intraperitoneal hematogenous seeding is common
-
typically shows asymmetrical but circumferential wall thickening with associated luminal dilatation (intestinal aneurysm), due to tumor extension into muscularis propria and destruction of autonomic nerve plexus
rarely causes bowel obstruction (tumor does not elicit desmoplastic response)
significant enlargement of mesenteric lymph nodes
MRI: low on T1, slightly increased on T2, hypovascular and thus very little contrast enhancement
ultrasound: wide variety of appearances, anechoic/hypoechoic, destruction of gut signature, aneurysmal dilation, wall thickening 3
-
gastrointestinal stromal tumor (GIST) 3
most common mesenchymal tumor
from interstitial cells of Cajal which regulate gut peristalsis
most frequently affect the stomach and small bowel
intraluminal mass or can extend through serosa as an exophytic mass with a large extraluminal component 4
bowel obstruction is rare even with large tumors 4
tends to displace adjacent structures rather than direct invasion
50% have metastases at time of diagnosis (liver, peritoneum)




 Unable to process the form. Check for errors and try again.
Unable to process the form. Check for errors and try again.