Nasopharyngeal carcinomas (NPC) are the most common primary malignancy of the nasopharynx. They are of squamous cell origin. Some types are strongly associated with the Epstein-Barr virus (EBV).
On this page:
Epidemiology
Nasopharyngeal carcinoma accounts for ~70% of all primary malignancies of the nasopharynx, and although rare in western populations, it is one of the most common malignancies encountered in Asia, especially China 1,3-5. It is commonly diagnosed between 40 and 60 years. Males are more commonly affected, with a male to female ratio of 3:1.
Risk factors
Risk factors are different depending on the histologic type of tumor present (see Pathology section).
Keratinizing squamous cell carcinoma can be thought of as a run-of-the-mill head and neck squamous cell carcinoma, which happens to be located in the nasopharynx. Its biological behavior is similar and it shares the same risk factors, namely smoking and alcohol 2. The other important risk factor is ingested nitrosamines (Chinese diet).
Non-keratinizing squamous cell carcinoma and basaloid squamous cell carcinoma are strongly associated with the Epstein-Barr virus (EBV) and are seen particularly in Asia 1-4. Additional risk factors include consumption of salted fish and meat, and rancid butter 2.
All three types express cytokeratin, and types II and III have incorporation of the EBV into their genome and circulating IgA antibodies to EBV in peripheral blood 1-4.
HPV infection also shows an etiologic role in the development of non-endemic EBV-negative nasopharyngeal cancers. HPV-positive and EBV/HPV-negative tumors exhibit worse outcomes than EBV-positive tumors 7.
Clinical presentation
Clinical presentation is often late, only when the tumor has grown significantly in size and has invaded adjacent structures. Hence, metastasis to bone, lungs, and liver are very common at the time of presentation. However, metastasis to brain is rare 13.
Early, but often ignored, symptoms include nasal obstruction, epistaxis, or conductive hearing loss due to Eustachian tube obstruction and the development of a middle ear effusion. Actual presentation is often delayed until more sinister signs are evident, including nodal masses in the neck (most common), cranial nerve palsies, tinnitus, headache, or even diplopia and proptosis 1,2. The clinical triad of unilateral conductive hearing loss, trigeminal neuralgia, and soft palate immobility is known as Trotter syndrome, which occurs in the advanced stage of the disease.
Diagnosis is usually achieved with endoscopic guided biopsy 4. A minority of patients have submucosal disease, with normal-appearing overlying mucosa. MRI is then essential in guiding biopsy 4.
Pathology
These tumors are defined as carcinomas that arise from the nasopharyngeal mucosa and show squamous differentiation 8. Earlier WHO classifications (1978 and 1991) divided nasopharyngeal carcinoma into three subtypes, but the most recent classification (2005) divides nasopharyngeal carcinoma by histological features and is less focussed on naming types 1-3 1,2,8:
keratinizing squamous cell carcinoma
-
non-keratinizing carcinoma
differentiated type
undifferentiated type
basaloid squamous cell carcinoma
Staging
See nasopharyngeal carcinoma staging.
Radiographic features
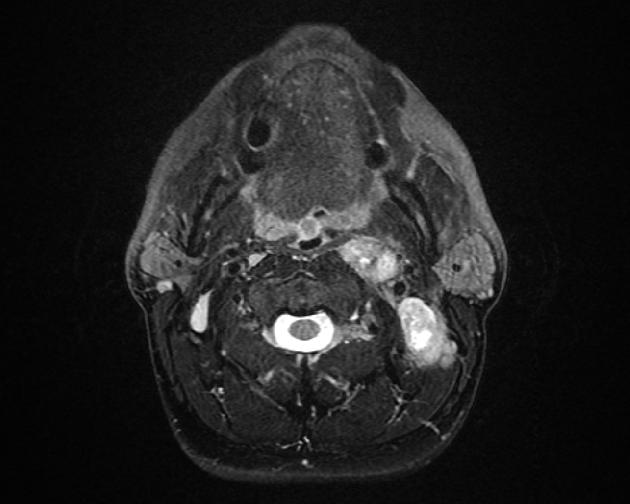
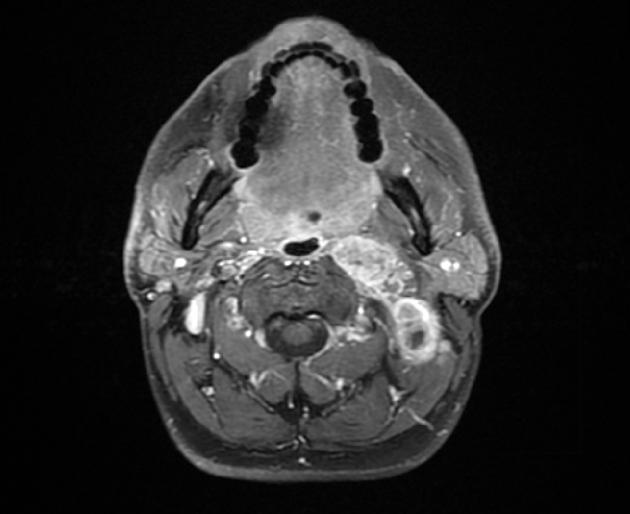
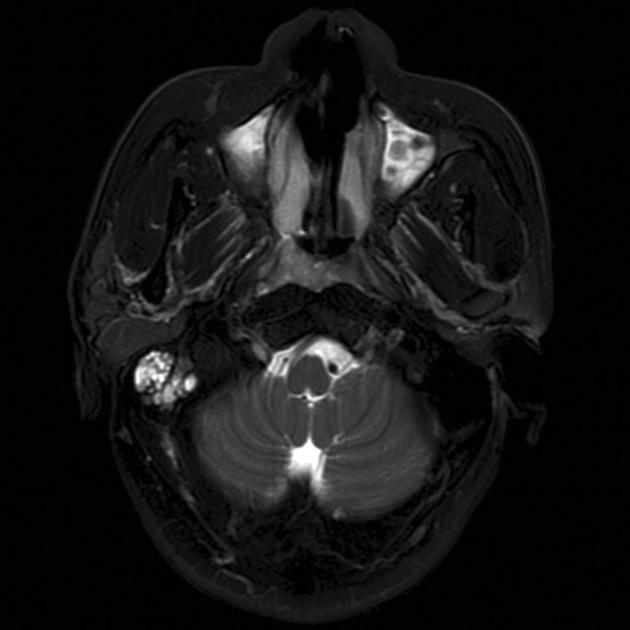
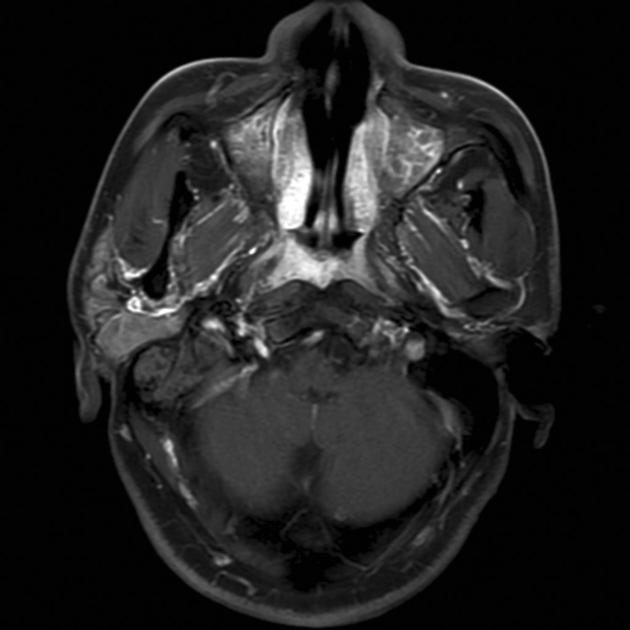
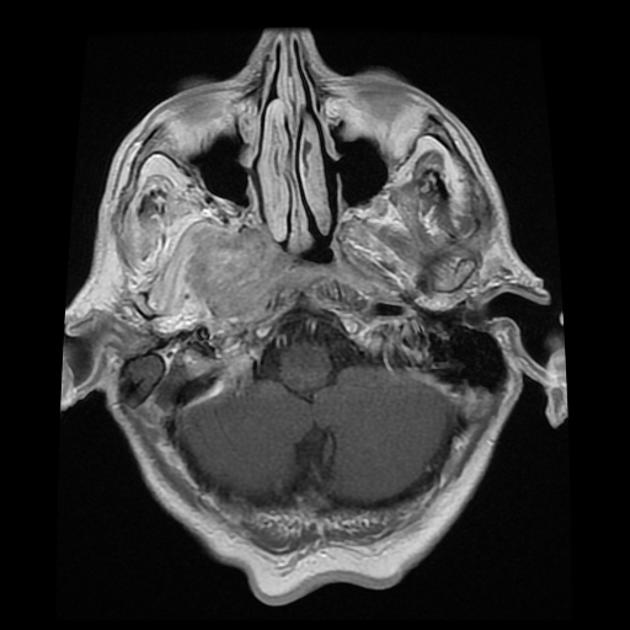
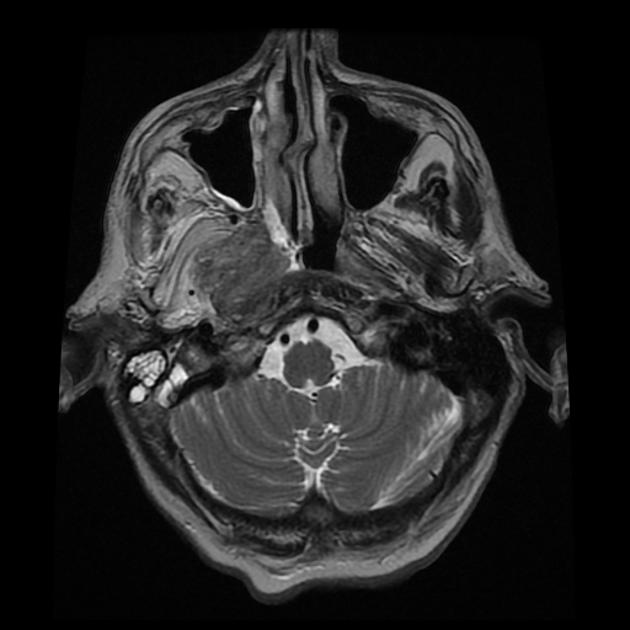
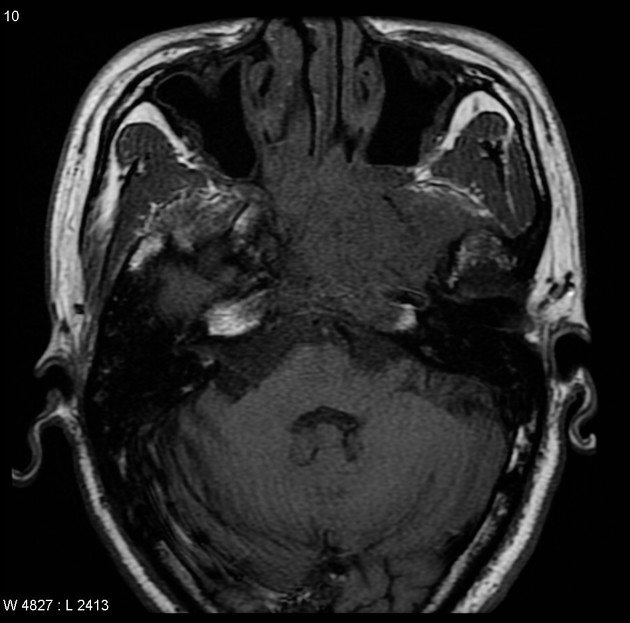
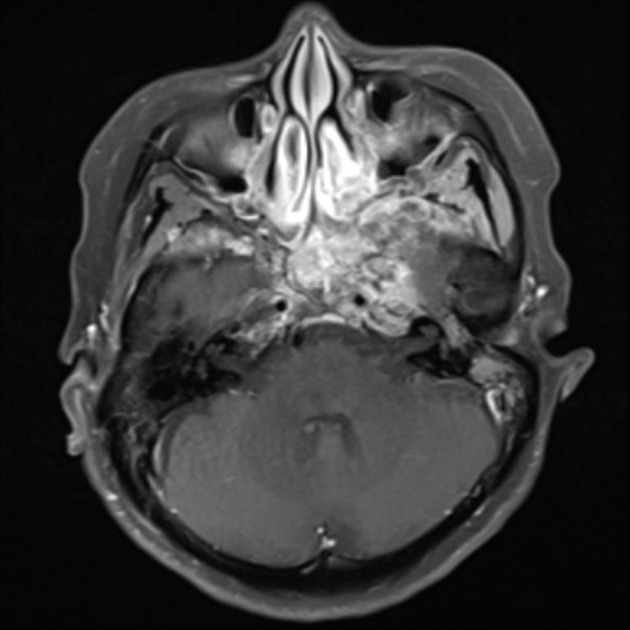
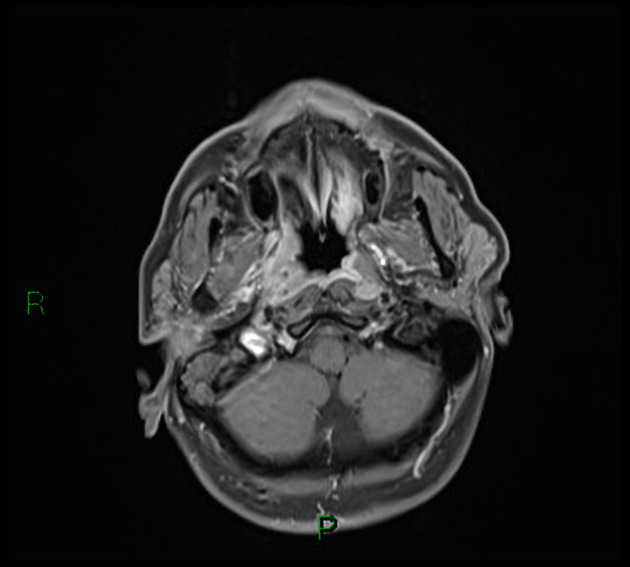
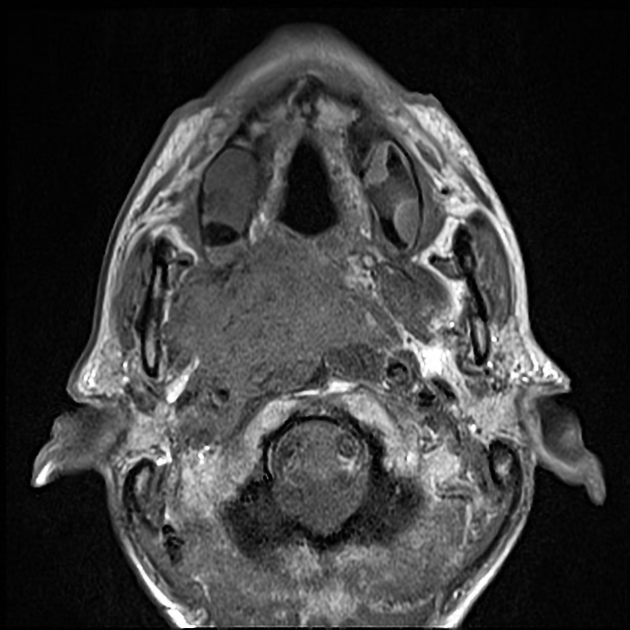
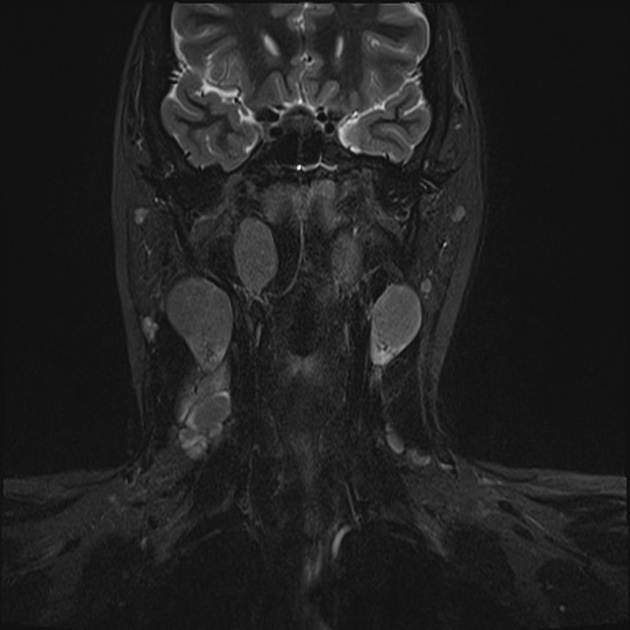
Imaging is crucial in delineating the extent of local tumor extension, as well as detecting nodal metastases which are present in the vast majority of patients at the time of diagnosis (75-90%) 1,3. Unfortunately, imaging in isolation is not only unable to distinguish between the various types of nasopharyngeal carcinoma, but also unable to distinguish nasopharyngeal carcinoma from other primary malignancies of the nasopharynx 1. Initially, they efface the fossa of Rosenmüller. Level II and V, and retropharyngeal nodes are commonly involved.
CT
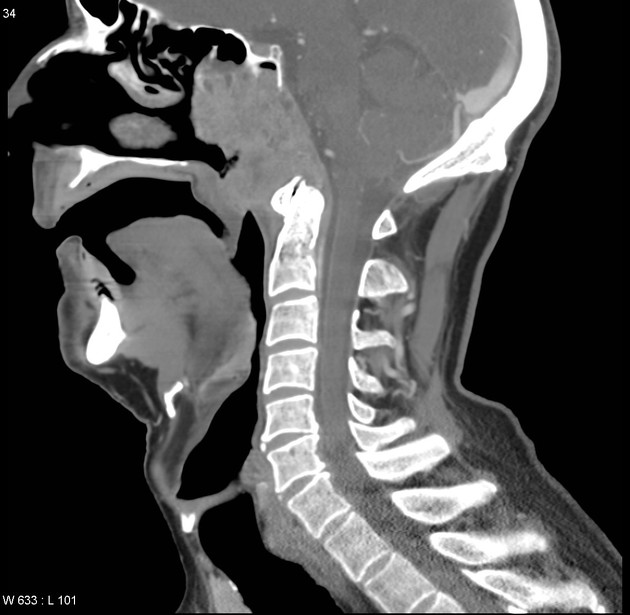
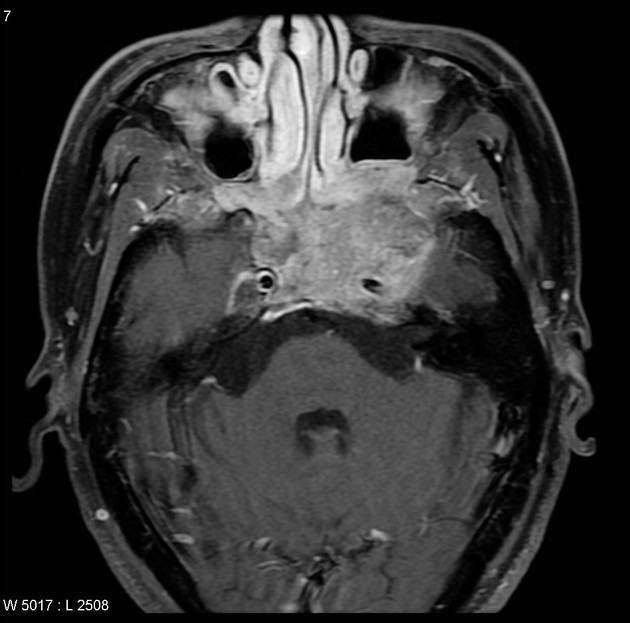
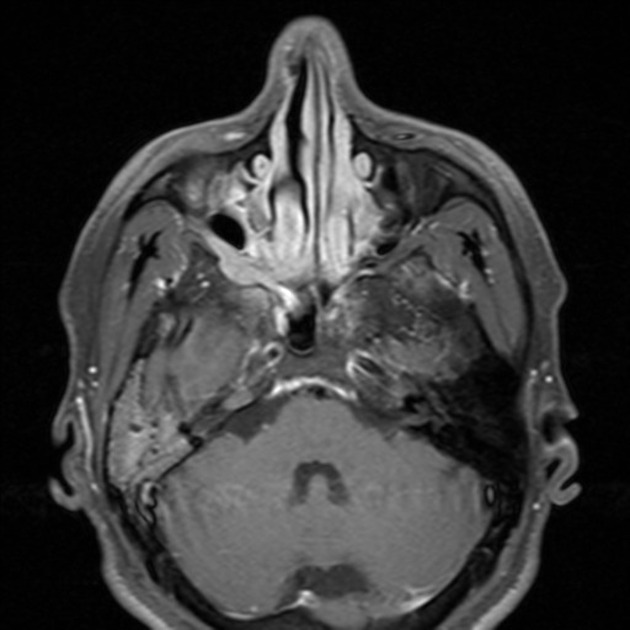
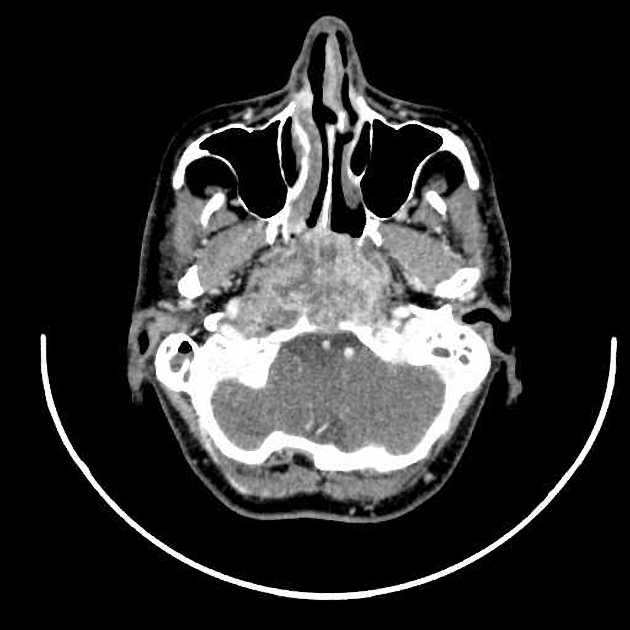
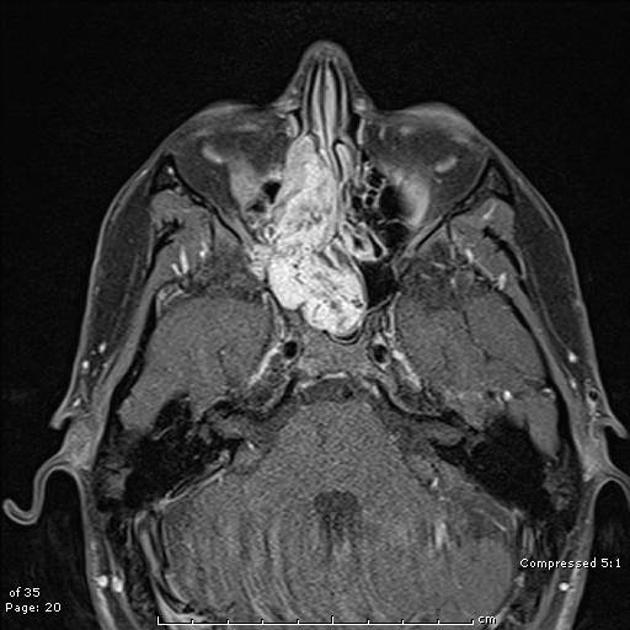
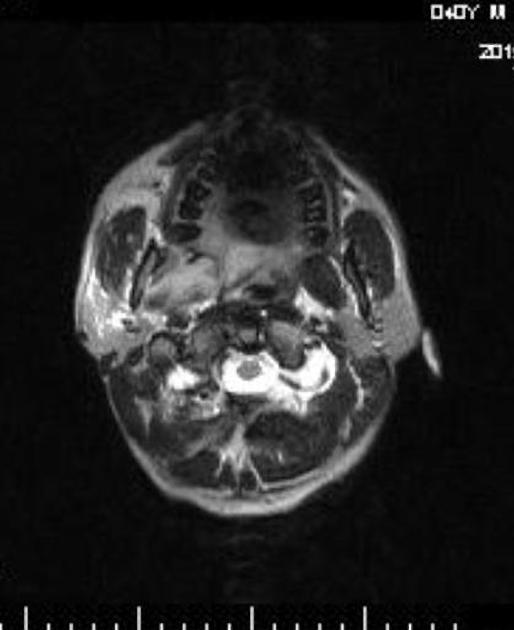
CT is not only more readily available but is also the ideal modality to assess early bony involvement. Nasopharyngeal carcinomas appear as soft tissue masses most commonly centered at the fossa of Rosenmüller.
Small lesions are confined to the nasopharynx by the pharyngobasilar fascia, and are indistinguishable from the prominent adenoidal tissue.
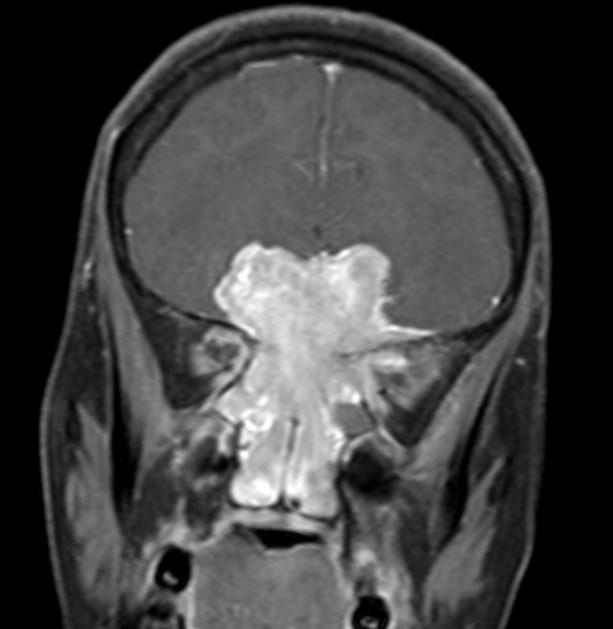
Larger, more aggressive tumors may extend in any direction, eroding the base of the skull and passing via the Eustachian tube, foramen lacerum, foramen ovale, or directly through the bone into the clivus, cavernous sinus and temporal bone. In such cases, the bone has irregular margins where it has been destroyed, characteristic of aggressive processes.
Following administration of contrast, the tumor mass and nodal metastases usually demonstrate heterogeneous enhancement.
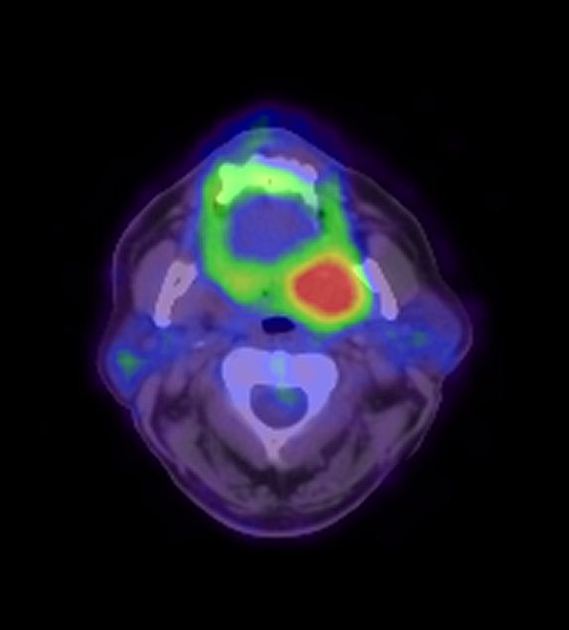
Careful assessment of cervical lymph nodes is essential due to the high rate of nodal involvement at the time of diagnosis. The retropharyngeal nodes are usually the first affected. However, in up to 35% of cases, these nodes are skipped, and level II nodes are involved first 1,3.
NB: Post-radiotherapy fibrosis can mimic residual tumor on CT 3.
MRI
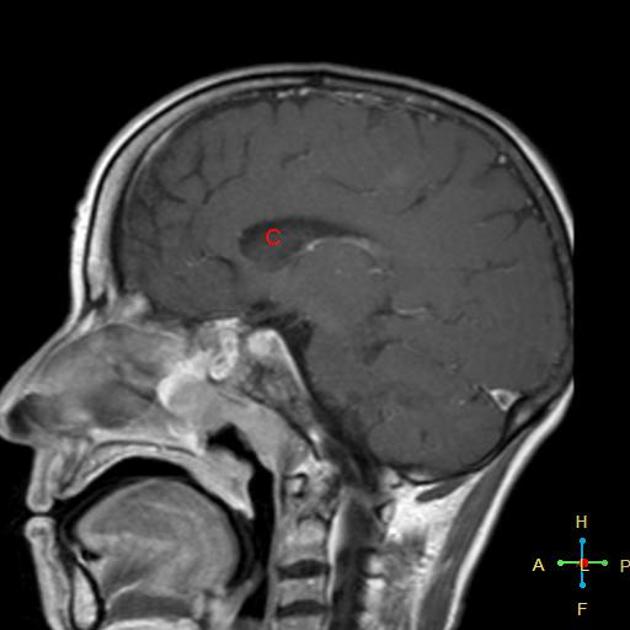
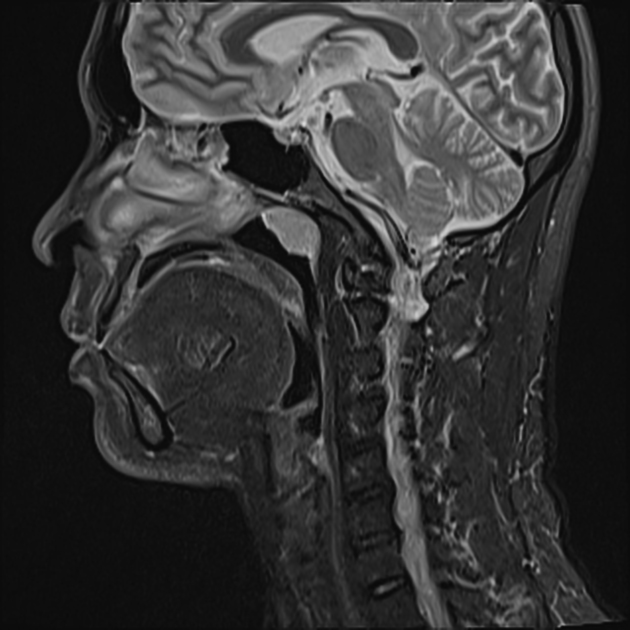
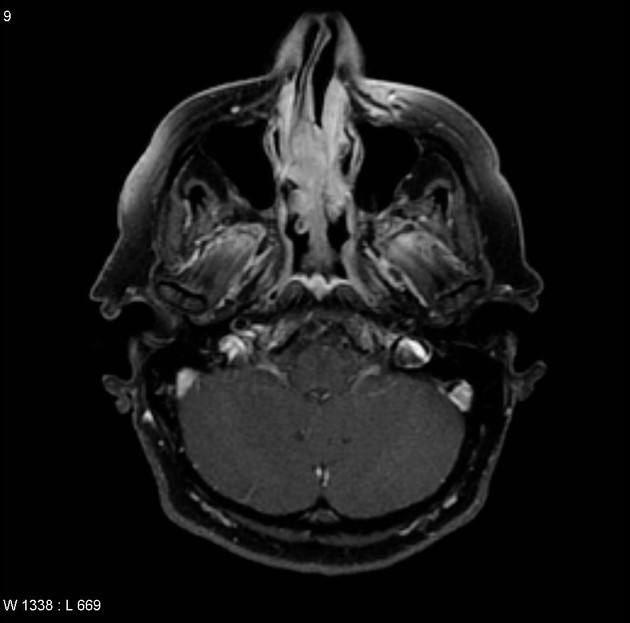
MRI is the modality of choice for tumor staging in nasopharyngeal carcinoma 10 due to its exquisite soft tissue resolution, sensitivity to perineural spread and intracranial extension. It is also sensitive for marrow changes of infiltration (see normal bone marrow signal of the clivus), although not all bone marrow changes represent tumor extension 3. A 2022 meta-analysis supports high sensitivity of MRI for early tumor detection in well-selected patient cohorts 11 although assessment is aided by using an MRI grading system to improve diagnostic performance 12.
T1: typically isointense to muscle
-
T2
isointense to somewhat hyperintense to muscle
fat saturation is helpful 5
fluid in the middle ear is common but not specific
-
T1 C+ (Gd)
post-contrast sequences should be fat-saturated
prominent heterogeneous enhancement is typical
perineural extension should be sought
MRI criteria have been proposed for diagnosis of NPC, including mucosal asymmetry, loss of adenoidal septa, and loss of integrity of the deep mucosal white line9.
Post-radiotherapy fibrosis can be distinguished from a recurrent or a residual tumor on MR if the fibrosis is mature. In such cases, fibrotic scarring is of low signal intensity on T2 and does not demonstrate enhancement. Early fibrotic change cannot be distinguished from a residual/recurrent tumor, as both may be hyperintense on T2 and demonstrate enhancement 3.
Nuclear medicine
F-18 FDG-PET is highly sensitive for nodal metastases and is the modality of choice to detect recurrence.
Treatment and prognosis
The mainstay of treatment is external beam radiotherapy, supplemented in some cases with chemotherapy. Surgery has little role in the management of nasopharyngeal carcinoma other than for the purposes of diagnostic biopsy. Surgery is also considered in radiation-resistant tumors and in local recurrence.
Complications
A potential complication of radiotherapy is radiation necrosis of the temporal lobes, as well as cranial nerve dysfunction, and atrophy and fibrosis of the muscles of mastication and salivary glands 3.
Differential diagnosis
On imaging alone, nasopharyngeal carcinomas appear similar to other primary nasopharyngeal malignancies. Tumors of the skull base should also be included in the differential, especially when significant bony involvement is present.
The differential for a small mass confined to the mucosal space includes:
-
prominent but normal adenoidal tissue
often has a striped appearance at MRI on T1WI and T2WI 4
low grade or other early primary nasopharyngeal malignancies
The differential for a larger mass with involvement of base of skull includes all of the above, with the addition of the following:






 Unable to process the form. Check for errors and try again.
Unable to process the form. Check for errors and try again.