Peritoneal metastases are a relatively common location for metastases, particularly from tumors of the abdomen and pelvis, that generally imply a poor prognosis, often with a significant impact on palliation 1.
On this page:
Terminology
If peritoneal metastases are of an epithelial origin (as most are) and extensive, the term peritoneal carcinomatosis may also be used. Less commonly, peritoneal metastases arise from mesodermal or lymphoid cell lines, with peritoneal sarcomatosis or peritoneal lymphomatosis respectively as the preferred terms 1.
Epidemiology
The epidemiology of patients with peritoneal metastases mirrors that of affected patients. Common primaries include 1,2:
extra-adrenal, intra-abdominal paraganglioma 5
-
hematogenous spread
Clinical presentation
Isolated peritoneal metastases are usually asymptomatic. Peritoneal carcinomatosis may also be asymptomatic, but eventually, most patients begin to report symptoms which can vary from uncomfortable to debilitating. Symptoms include 1:
abdominal distention due to malignant ascites
-
abnormal bowel motility, resulting
nausea/bloating
intermittent pains
bowel obstruction (most common from colorectal cancer metastases)
Pathology
Malignant involvement of the peritoneal lining occurs via a number of routes, including 1:
intraperitoneal seeding and direct invasion: most common
hematogenous dissemination
lymphatic dissemination: rare
Radiographic features
Although peritoneal metastases are visible on ultrasound, MRI and sometimes are implied on barium studies, CT is the most frequently used modality to investigate patients with suspected peritoneal metastases, or assess the resultant complications.
Ultrasound
Malignant ascites may be anechoic or have low-level echoes, and aids in the identification of deposits 1. Nodules are of intermediate echogenicity, hypoechoic compared to the peritoneum, whereas infiltration of the omentum results in hyperechogenicity 1.
CT
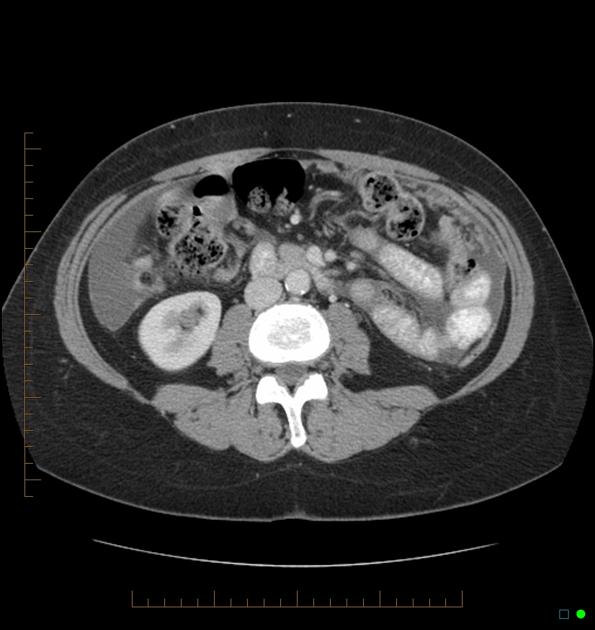
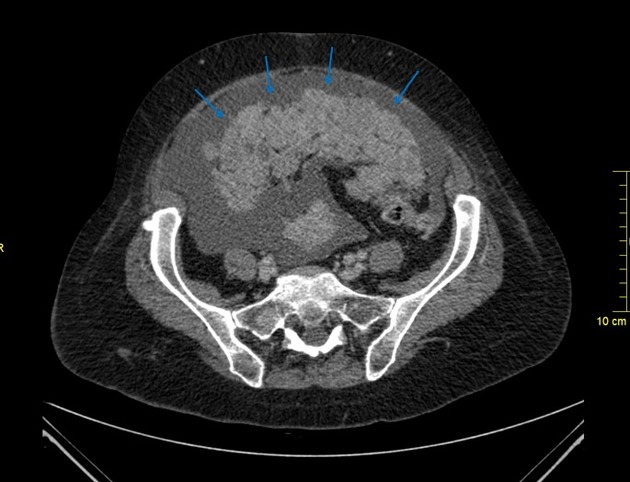
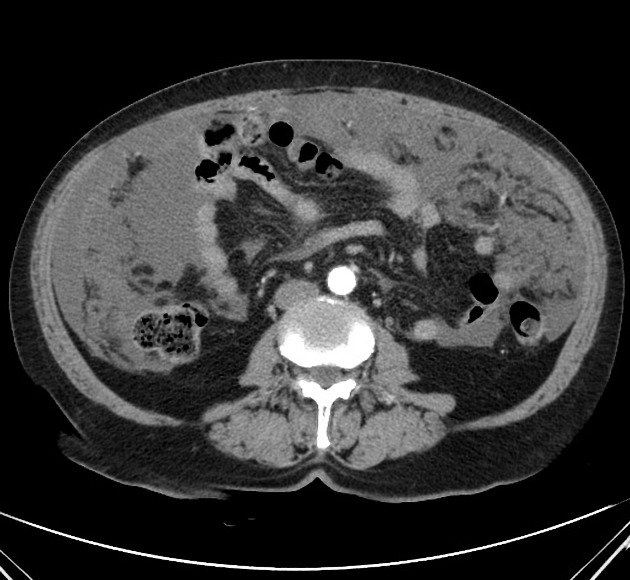
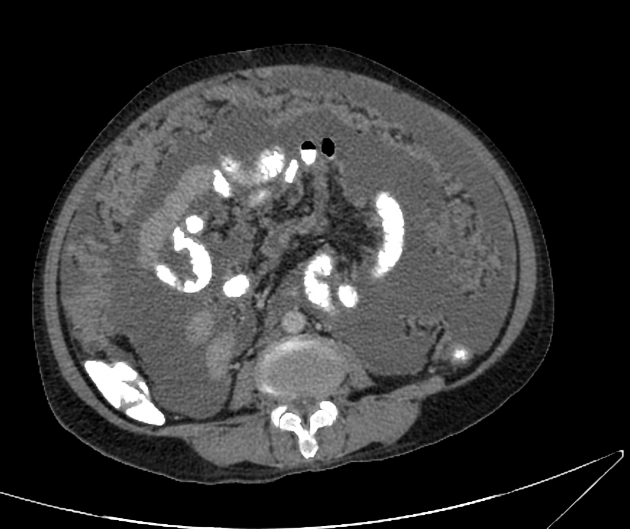
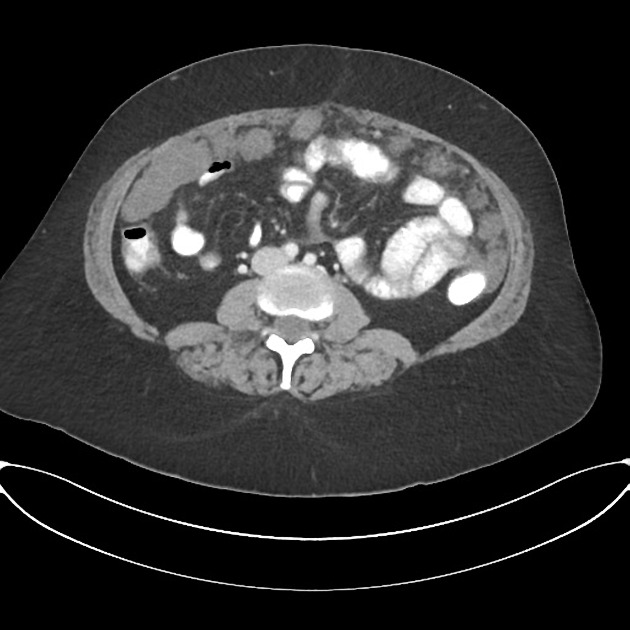
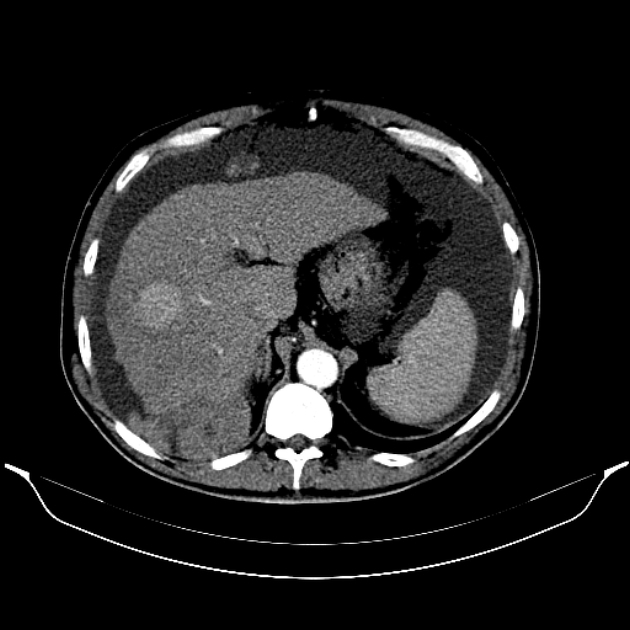
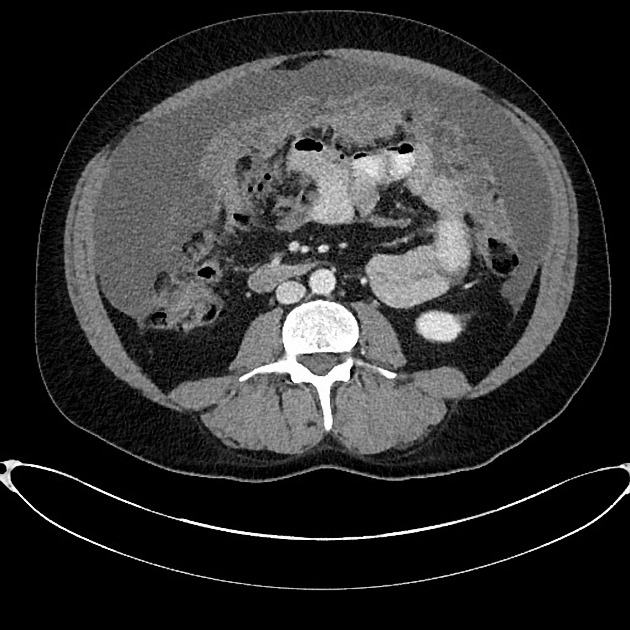
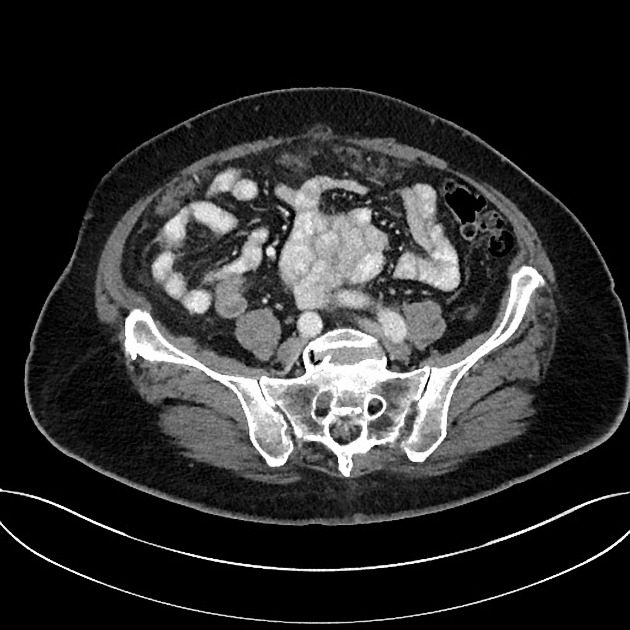
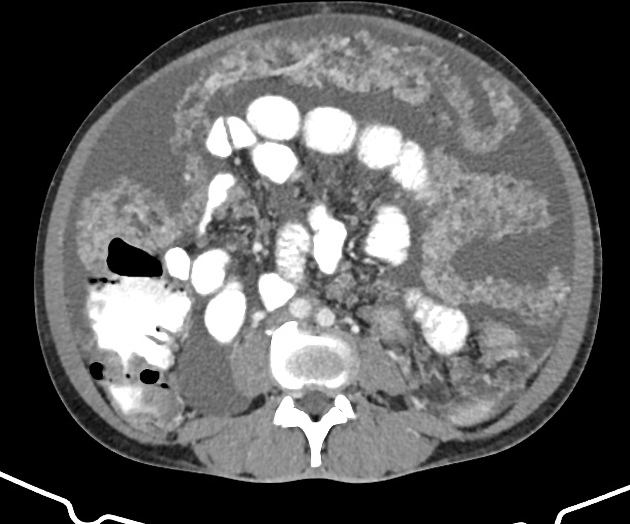
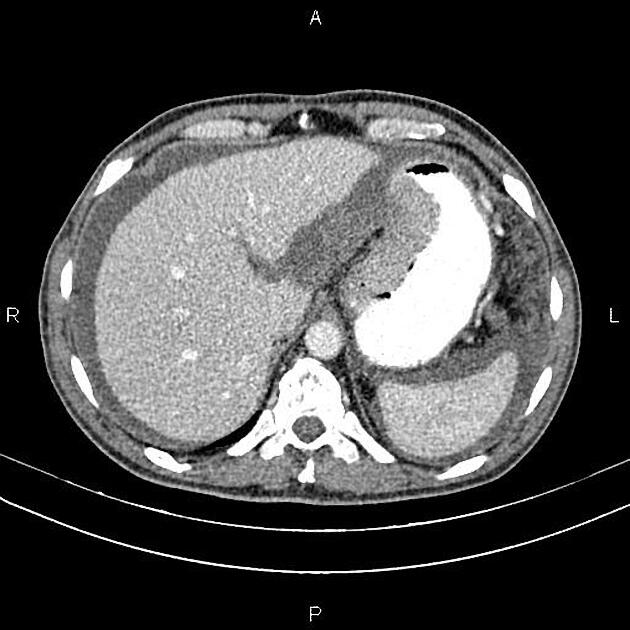
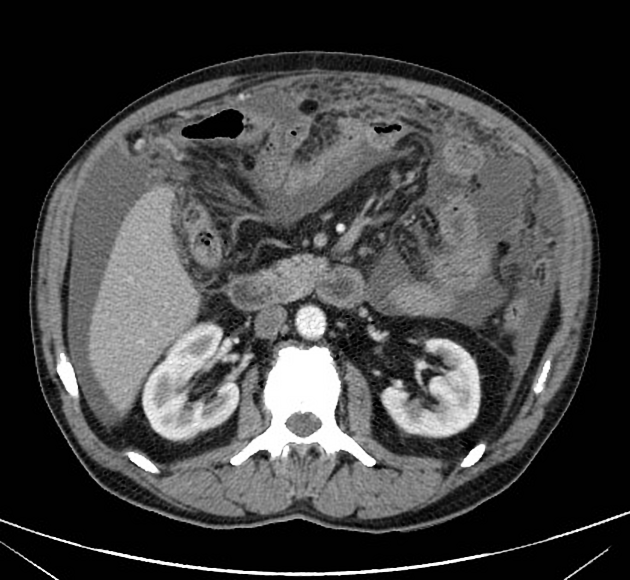
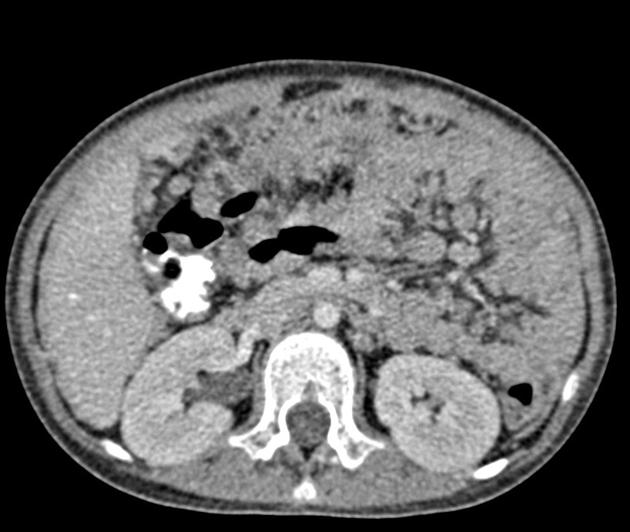
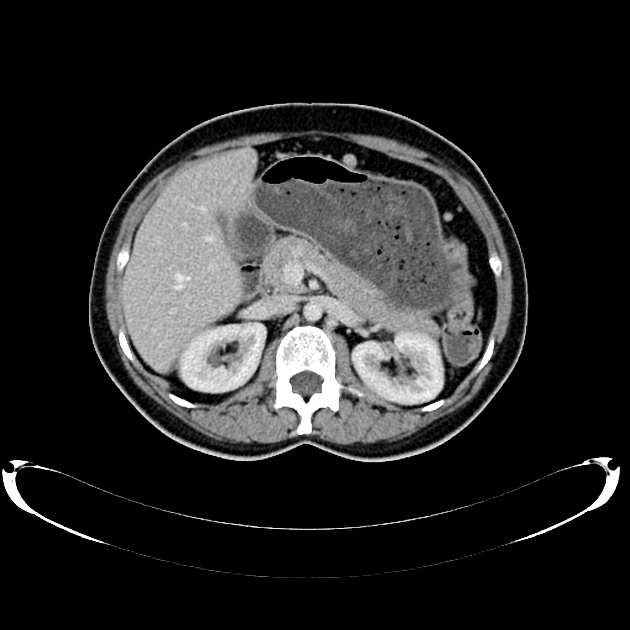
Peritoneal metastases can range in appearance from invisible to multiple large masses, and CT may detect 85-93% of peritoneal metastases later shown to be present at surgery 1,3.
Appearances include 1:
thickening and enhancement of peritoneal reflections (especially if nodular)
soft tissue nodules
stranding and thickening of the omentum (omental cake)
stranding and distortion of the small bowel mesentery
ascites, especially if loculated
-
calcifications 2 (particularly in cystadenocarcinoma of the ovary)
nodular with the non-calcified component are typical
nodal calcification
Intraperitoneal contrast has been investigated as a way of improving sensitivity to the presence of small deposits, and may improve detection but has not been widely adopted 3.
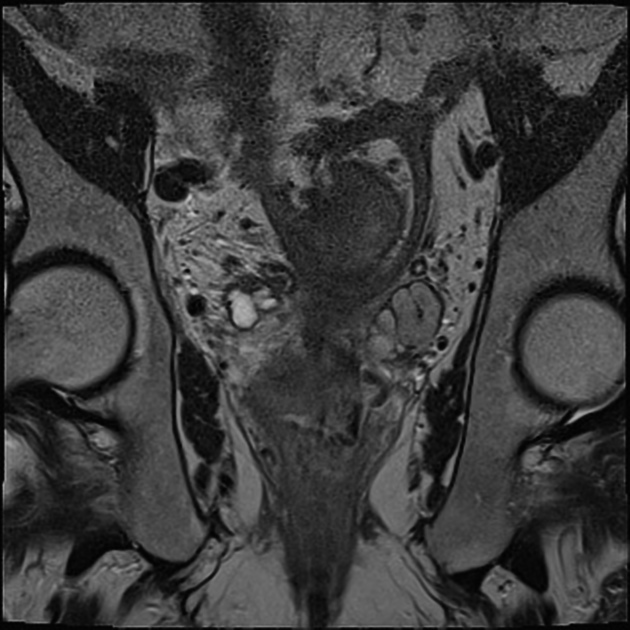
MRI
MRI can be very sensitive, probably more so than CT (85-90%) 1. Peritoneal metastases show up as increased enhancement (greater than liver), best seen after 5-10 minutes 1.
Treatment and prognosis
Peritoneal metastases per se are not locally treated, although systemic treatment may have some effect. Complications do however frequently require treatment for palliation:
bowel obstruction: bypass enterostomies may be required
malignant ascites: repeated ascitic drainage
Differential diagnosis
Differential diagnosis depends on the dominant pattern.
peritoneal mesothelioma is very similar to peritoneal carcinomatosis, but usually no primary neoplasm is known
-
peritoneal sarcomatosis: if the primary tumor is of mesenchymal origin (i.e. sarcoma)
most commonly metastases from a gastrointestinal sarcoma 1
-
most commonly metastases from a primary (e.g. non-Hodgkin lymphoma) elsewhere 1
-
peritonitis/sepsis
smooth thickening and enhancement of the peritoneum, with stranding of the omentum and mesentery may be seen in intra-abdominal sepsis
benign calcifications tend to be sheetlike, and nodal calcifications in these patients less common 2
a history of peritoneal dialysis or recent abdominal sepsis is usually easily obtained






 Unable to process the form. Check for errors and try again.
Unable to process the form. Check for errors and try again.