T2 relaxation, also known as spin-spin relaxation or transverse relaxation, refers to the progressive dephasing of spinning dipoles resulting in decay in the magnetization in the transverse plane (Mxy). Following a radiofrequency pulse, this form of relaxation occurs with the time constant T2 (also known as the exponential decay constant, T2) 3, where T2 is the time it takes for the transverse magnetization vector to decay to 1/e or 37% of its initial magnitude. T2 relaxation occurs due to tissue-particular characteristics, primarily those that affect the rate of movement of protons, most of which are found in water molecules.
Discussion
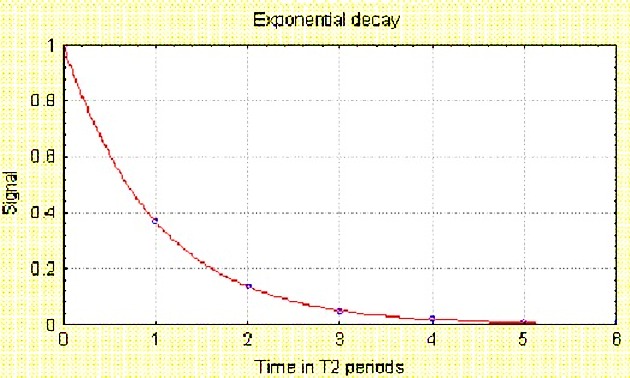
In spin echo sequences, immediately after the 90° pulse, all the spinning dipoles within the slice are exactly in phase. Almost immediately, they lose coherence as some spin slightly faster than the others. This dephasing effect has been likened to the opening of a Chinese fan. The result is that the Mxy component of the magnetic vector decreases exponentially as a function of the T2 time constant (Figure 1).
Factors affecting T2 relaxation
Each magnetic dipole exists in a microenvironment unique to the tissue where it belongs. In all tissues, there exist tiny magnetic fields (~1mT) generated by the spinning hydrogen nuclei (protons). T2 relaxation occurs in a varying local magnetic field when there is transfer of energy between dipoles facing parallel and antiparallel to the external magnetic field, flipping each other in opposite directions. This rate of flipping or transfer of energy between spins or dipoles increases as the frequency of the variation of the local magnetic field approaches the Larmor frequency. This is related to the rate of rotation and translation of the water molecule or adjacent dipoles. The dipole-dipole interaction is also increased the strength of the local field which is dependent on the proximity of the adjacent dipoles.
In pure water T2 is long, about 3-4 seconds because water molecules move considerably faster than the Larmor frequency. The rapid motion results in the T1 and T2 being about the same in pure water.
In solutions of macromolecules and tissues the relaxation rate is much faster, i.e., the T2 time is shorter. This is related in part to the slower motion of protons both in macromolecules as well as water molecules attracted to the surface of the macromolecule. This slower motion is closer to the Larmor frequency. Examples of T1 and T2 in biological tissues include: CSF, T1=1.9 seconds and T2=0.25 seconds; brain white matter, T1=0.5 seconds and T2=0.07 seconds (70 msec).
As motion, and therefore the local field fluctuations, decreases below the Larmor frequency in tissues and tendons, dipoles that are aligned with the main magnetic field start contributing to T2 relaxation by causing local variations in precession rate. The resulting short T2 time causes tendons and other semi-solid tissues to appear dark on T2-weighted images. Long T2 fluids with few macromolecules such as water, urine, and CSF will appear bright on T2-weighted images.
Loss of signal and darkness on T2-weighted images in cortical bone, teeth, calculi is primarily a result of little water (low proton density) unlike tendons and ligaments 4. The water that is in bone, teeth, and calculi would mostly be bound as to collagen and would have a very short T2 time constant and appear dark. There is also mild susceptibility differences between bone and soft tissue that could contribute to a dark appearance at interfaces, as between marrow and bone trabecula. This is seen in particular on gradient echo images.
Note: T2 relaxation is not to be confused with T2*, which is a broader phenomenon and includes static magnetic field effects in addition to the tissue-characteristic T2 relaxation.




 Unable to process the form. Check for errors and try again.
Unable to process the form. Check for errors and try again.