Von Hippel-Lindau (vHL) disease is characterized by the development of numerous benign and malignant tumors in different organs (at least 40 types 1) due to mutations in the VHL tumor suppressor gene on chromosome 3.
On this page:
Epidemiology
The disease is rare with an estimated prevalence of 1:35,000-50,000. Most patients are diagnosed with their first tumor in early adulthood (mean age at diagnosis of initial tumor is 26) 10.
Clinical presentation
Clinical presentation is varied, depending on the site of disease manifestation (see below). Most commonly these are either within the abdominal cavity or affect the central nervous system.
Pathology
Location
Patients may develop some or all of the various lesions which include:
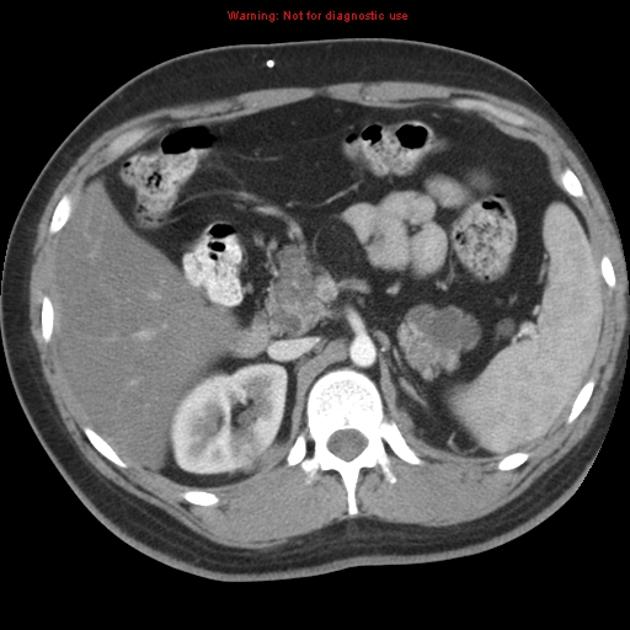
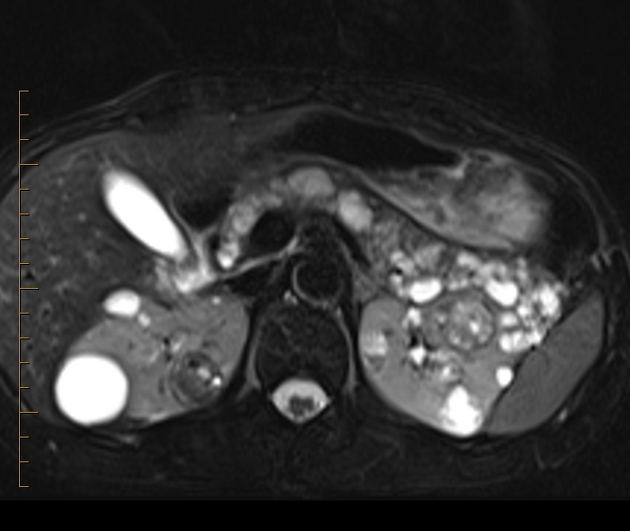
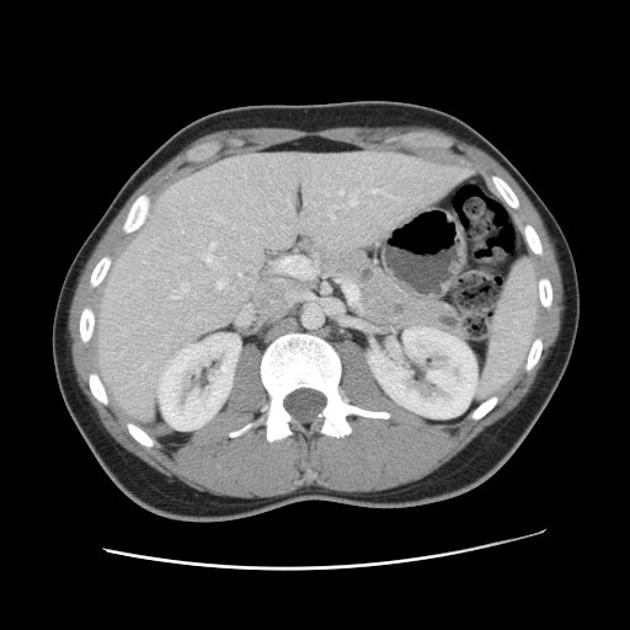
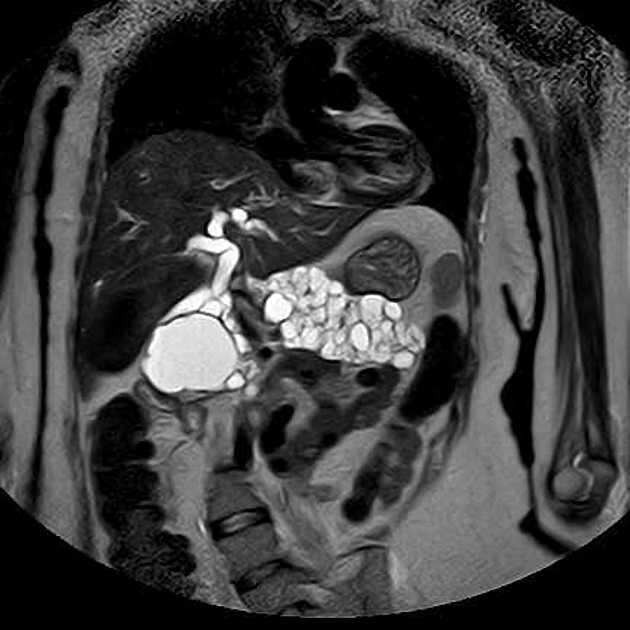
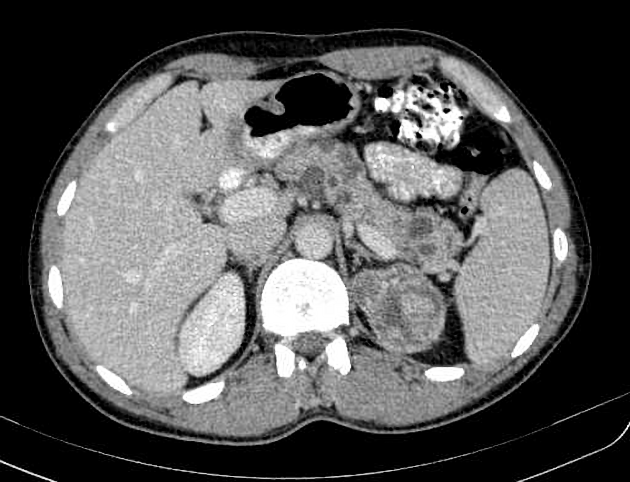
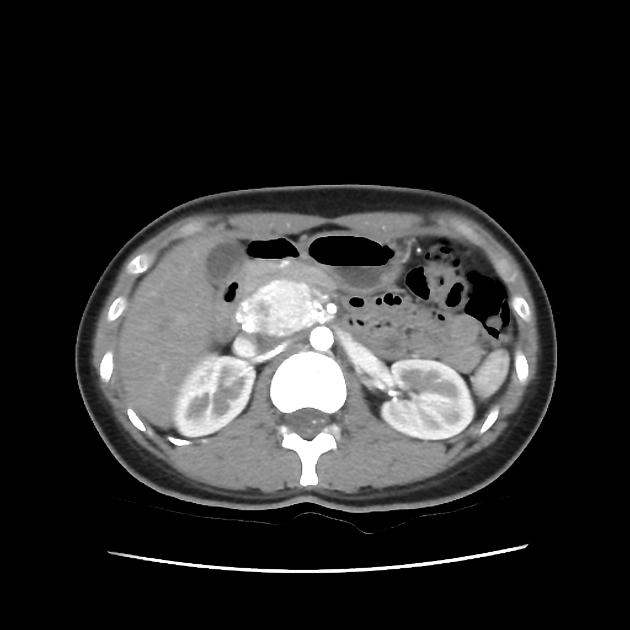
Abdominopelvic
-
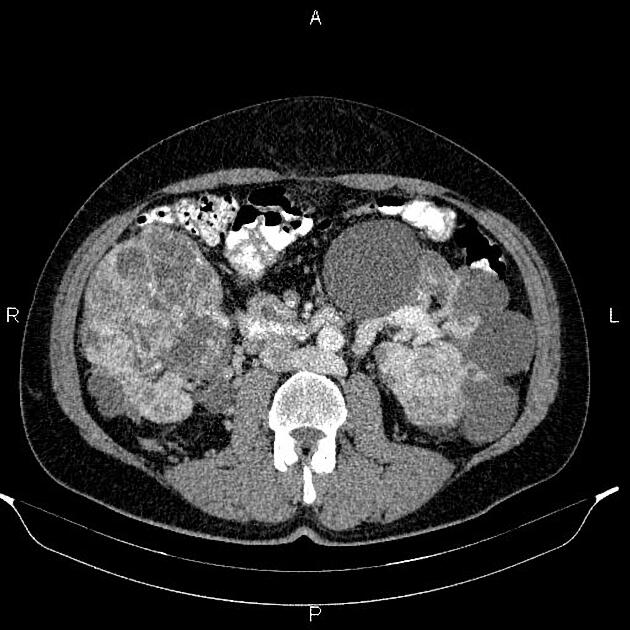
renal (>67% of patients 10)
-
usually of the clear cell type 7 and frequently bilateral 10
70% lifetime risk 9
renal cell carcinomas present at an earlier age (mean = 39 years) in those with vHL 10
-
up to 75% of patients 5
often tend to be bilateral and multiple
can be simple, complex or cystic renal cell carcinoma 10
-
-
adrenal
pheochromocytomas: 25-30% of patients 10
extra-adrenal pheochromocytoma / paraganglioma: 15% 8,10
-
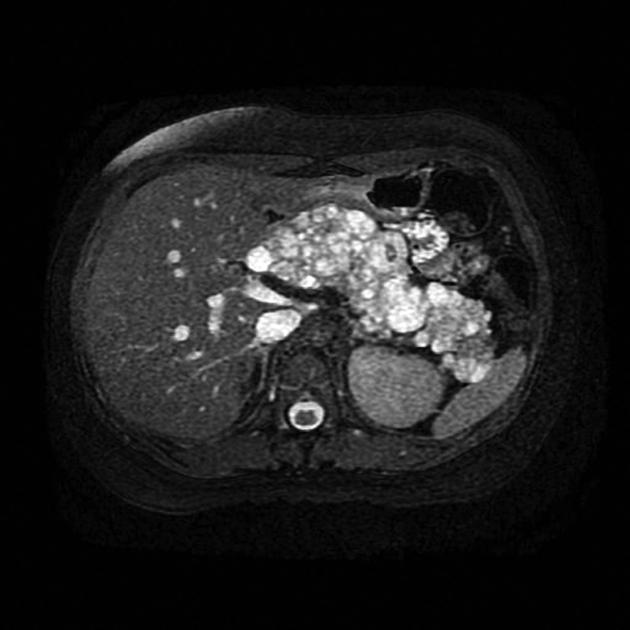
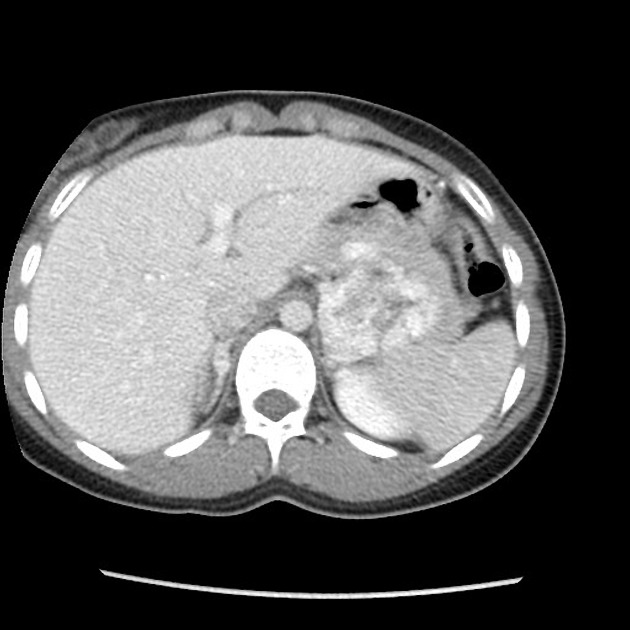
pancreas (may be the earliest manifestation 3)
pancreatic cysts: ~40% of patients 10
-
pancreatic neuroendocrine tumors (pNET)
~12.5% of patients (range 9-17%) 10
usually non-functional 9
frequently multiple
pancreatic serous cystadenoma: ~12.5% of patients (range 9-17%)
-
liver
-
lung
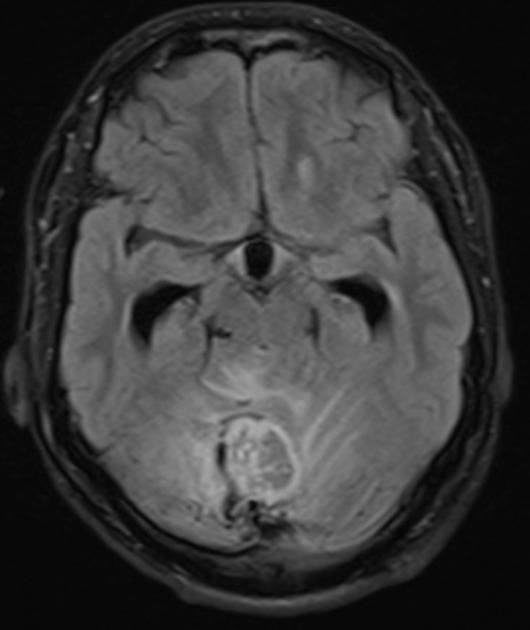
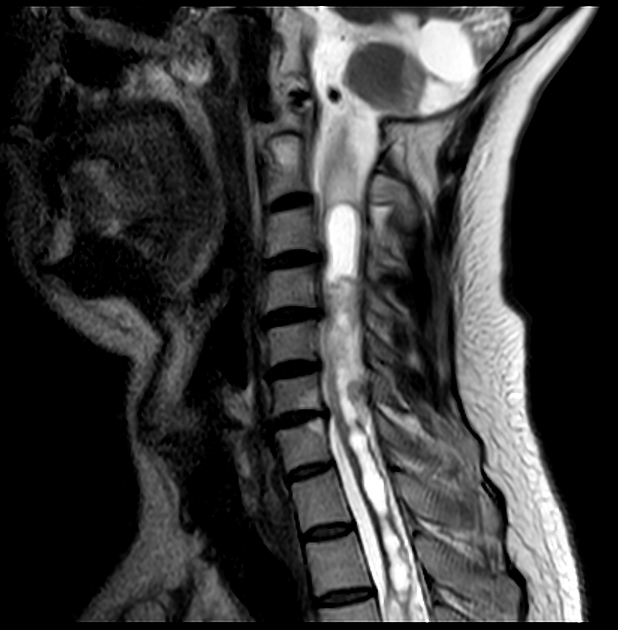
hemangioblastoma
Urogenital
papillary cystadenoma of the epididymis: ~35% of patients (range 25-60%) 10
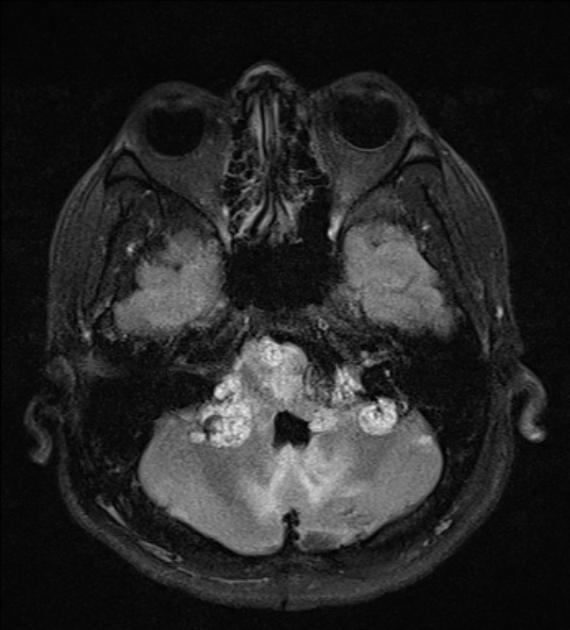
CNS
-
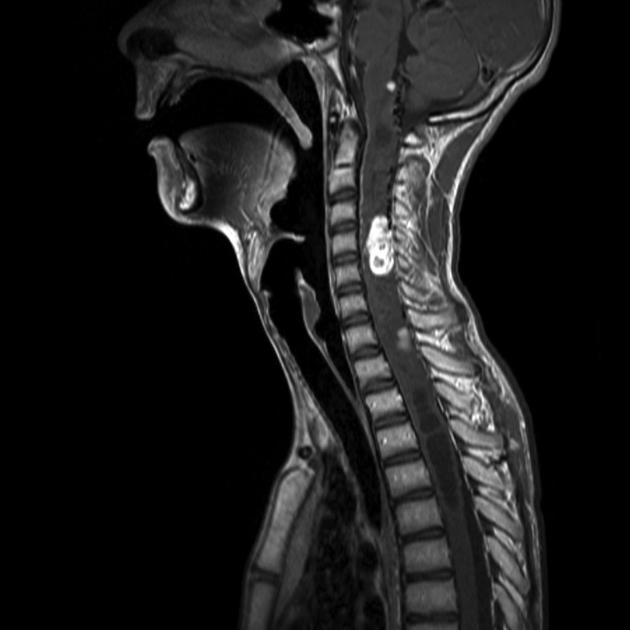
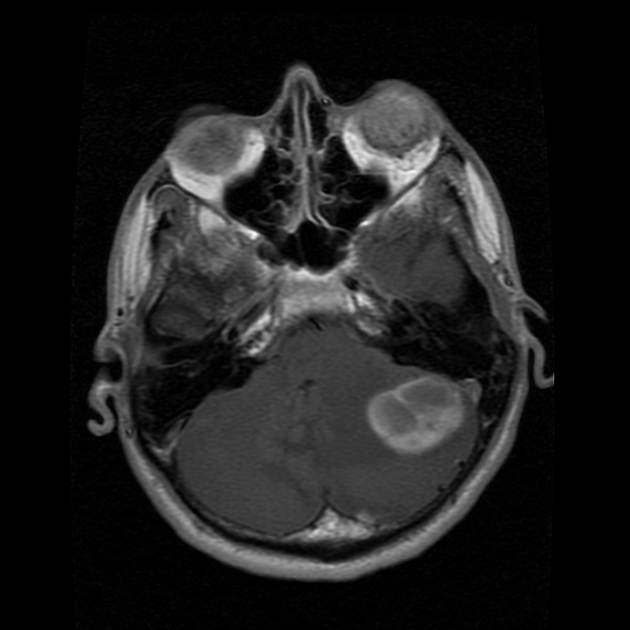
CNS hemangioblastomas: ~70% of patients (range 60-80%) 9,10
cerebellar (~60%; range 44-72%)
spinal cord (~30%; range 13-50%); most commonly in the cervical and thoracic cord
brainstem
Head and neck
-
most common presenting feature, 45-60% of patients 9,10
vision loss in 35-55% of patients 9
-
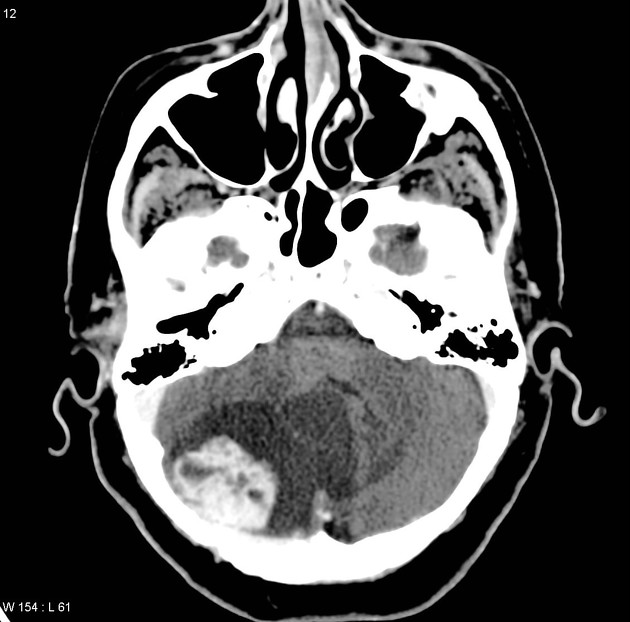
endolymphatic sac tumors (ELST)
10-15% of patients 10
bilateral in 30% 10; considered pathognomonic for vHL 9
A mnemonic to help remember the features of vHL is HIPPEL.
Classification
VHL can be classified according to clinical phenotypes, and the classification correlates with particular genotypes 10:
type 1: low-risk for pheochromocytoma but higher-risk for CNS hemangioblastoma, renal cell carcinoma, pancreatic cyst, and pancreatic neuroendocrine tumor
type 2A: high-risk for pheochromocytoma; low-risk for renal cell carcinoma
type 2B: high-risk for pheochromocytoma and renal cell carcinoma
type 2C: high-risk for pheochromocytoma only
Genetics
The disease carries an autosomal dominant inheritance with high expression and penetrance; ~80% of cases occur via this pathway with ~20% arising de novo 10. It results from the inactivation of VHL, a tumor suppressor gene located on chromosome 3p25.5. However, no mutation is identified in up to 30% of cases.
Radiographic features
Please refer to articles on individual lesions for specific imaging characteristics.
Treatment and prognosis
Most lesions from vHL are treatable and surveillance is recommended with various regional guidelines 10. Some experts advocate routine screening starting in adolescence. Prognosis is poor, with a median survival of ~50 years, with the most common cause of death being renal cell carcinoma and cerebellar hemangioblastoma 1.
History and etymology
Eugen von Hippel (1867-1939) was a German ophthalmologist who had described a rare disorder of the retina in 1904 and seven years later reported the basis of this disease, named "angiomatosis of the retina".
Arvid Vilhelm Lindau (1892-1958) was a Swedish pathologist and bacteriologist who described the association between angiomatosis of the retina and hemangioblastomas of the cerebellum and other parts of the CNS and other visceral components of a disease, calling it "angiomatosis of the central nervous system".
In 1964 the disease was renamed Von Hippel-Lindau disease.




 Unable to process the form. Check for errors and try again.
Unable to process the form. Check for errors and try again.