Hepatoblastomas are the most common primary malignant liver tumors in children under four years of age. They usually present with a painless abdominal mass and raised AFP. Hepatoblastomas are embryonic tumors.
On this page:
Epidemiology
Most cases are seen during the first 18 months of life, and diagnosis in adulthood is exceedingly rare. Occasionally, the tumor may be diagnosed antenatally 2. There is a recognized slight male preponderance, with an M:F ratio of ~3:2. There may also be a predilection towards the right lobe of the liver.
Associations
Most hepatoblastomas are seen sporadically, however, it has been known to be associated with:
hemihypertrophy: seen in 2% of patients with hepatoblastoma 1
prematurity and low fetal birth weight 1,6
Wilms tumor 14
-
biliary atresia 1,2
Clinical presentation
Most children present with abdominal distension or an asymptomatic palpable abdominal mass. However, other presenting symptoms include 1:
abdominal pain
anorexia
vomiting
pyrexia
anemia
precocious pseudopuberty, also known as peripheral precocious puberty (due to β-hCG secretion 13)
Pathology
Macroscopic appearance
Macroscopically hepatoblastomas are usually relatively well-circumscribed large masses, usually single, with heterogeneous cut surface 10.
Microscopic appearance
The histological classification of hepatoblastoma is complicated with multiple subtypes identified 10:
-
major categories
-
epithelial
fetal, well differentiated
embryonal
macrotrabecular
mixed (calcifications more common ref)
small cell undifferentiated
rhabdoid
-
-
minor categories
cholangioblastic (ductal)
keratinizing squamous epithelium
intestinal glandular epithelium
teratoid (neuroid-melanocytic)
rhabdomyoblastic
chondroid
osteoid
It is important to note that with the possible exception of small cell undifferentiated subtype, prognosis is independent of histology when adjusted for stage gender and age 10.
Markers
Serum alpha fetoprotein (AFP) levels are frequently elevated (90% of cases 6).
Staging
Refer to: hepatoblastoma staging.
Caval involvement often indicates unresectable disease.
Radiographic features
Plain radiograph
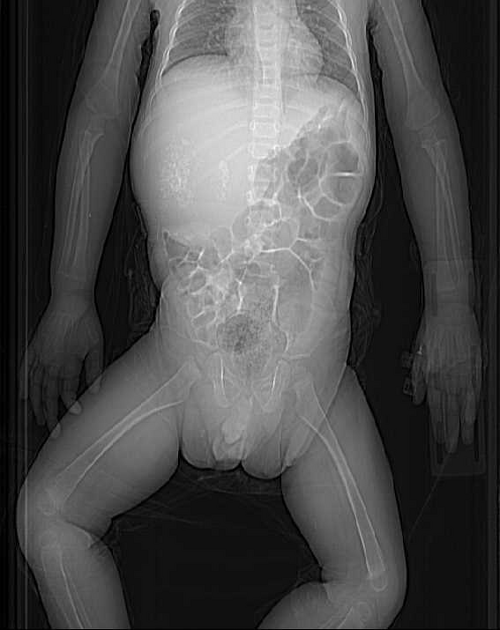
Abdominal x-rays are nonspecific, typically demonstrating a right upper quadrant mass. Calcifications are visible in 10% of cases 1,4.
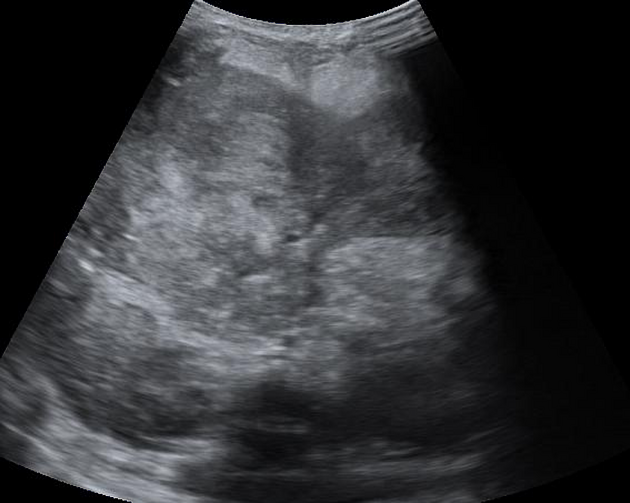
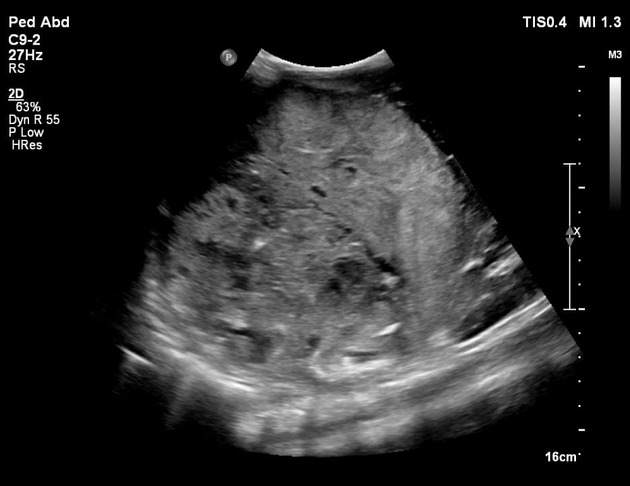
Ultrasound
On ultrasound, hepatoblastomas appear as predominantly echogenic soft tissue masses. In larger tumors heterogeneity and variable echogenicity is common. Even when large, they tend to be relatively well-defined 7. Intralesional calcifications may be visible as areas of shadowing 4,7.
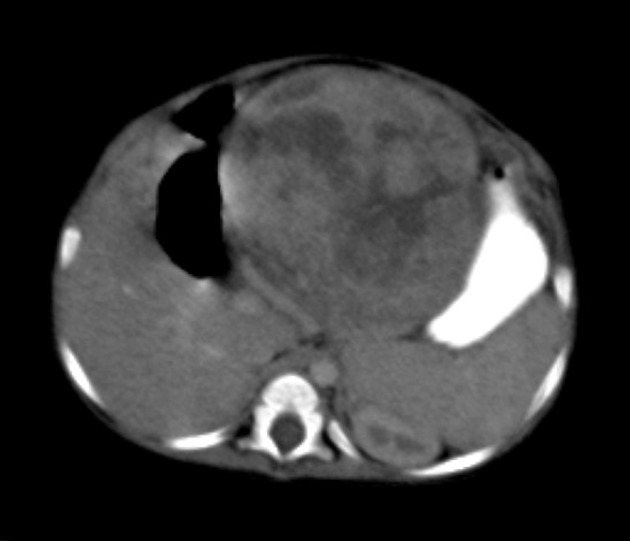
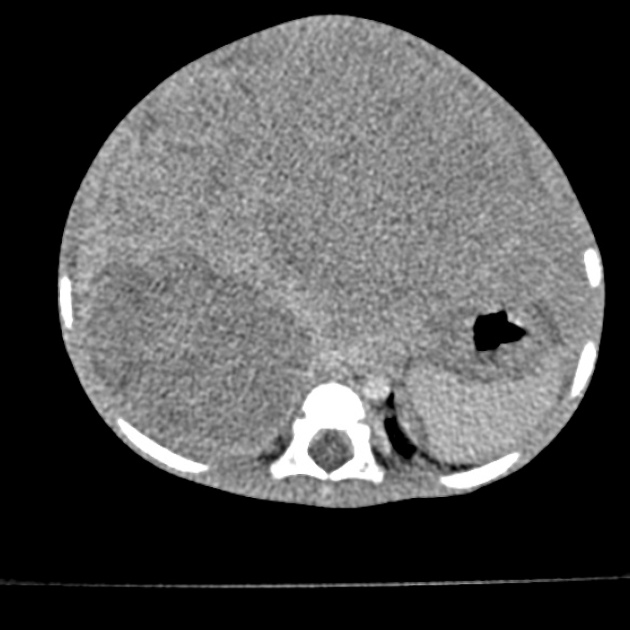
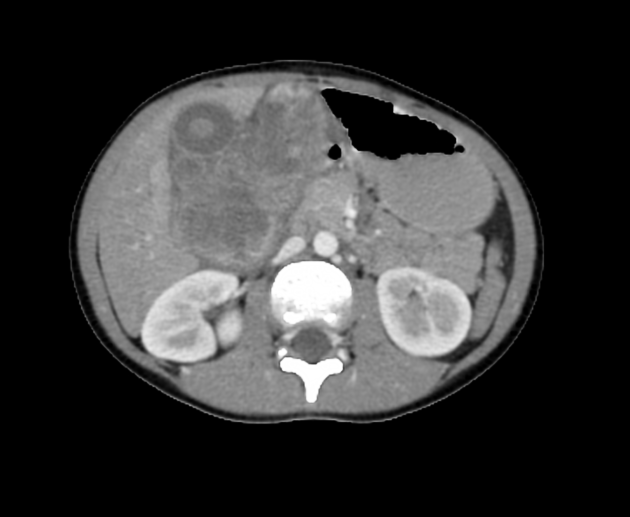
CT
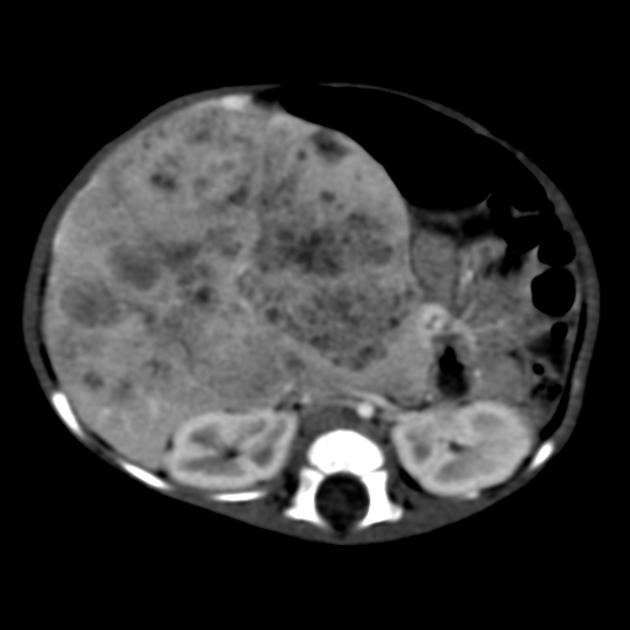
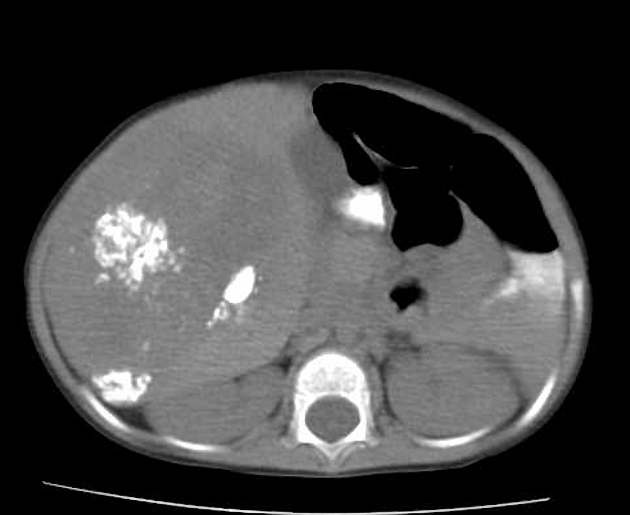
Usually seen as a well-defined heterogeneous mass, which is usually hypoattenuating compared to the surrounding liver 11. Frequently there are areas of necrosis and hemorrhage. Chunky, dense calcifications may be seen in approximately 40% of cases 11.
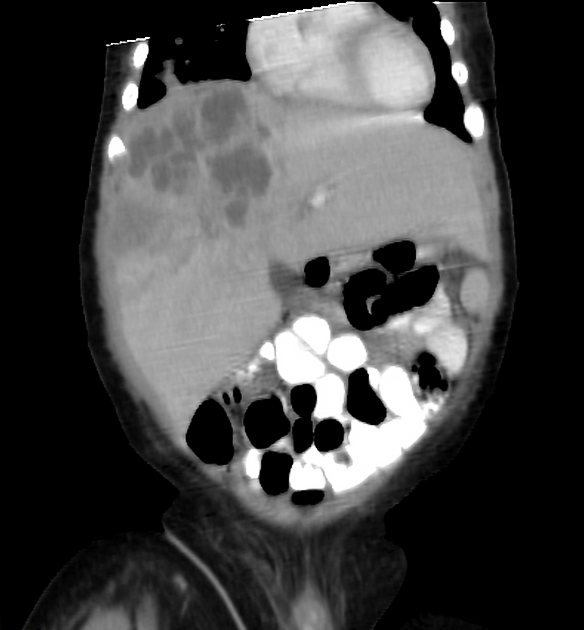
CT is also able to evaluate the lungs for metastases and for nodal enlargement.
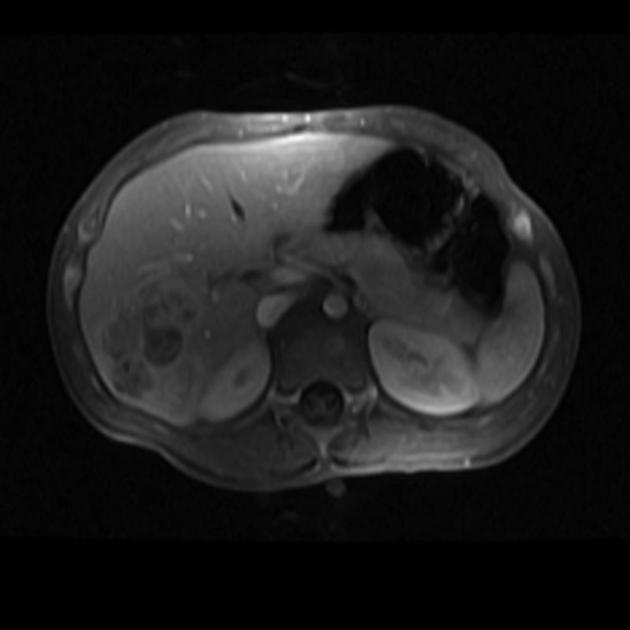
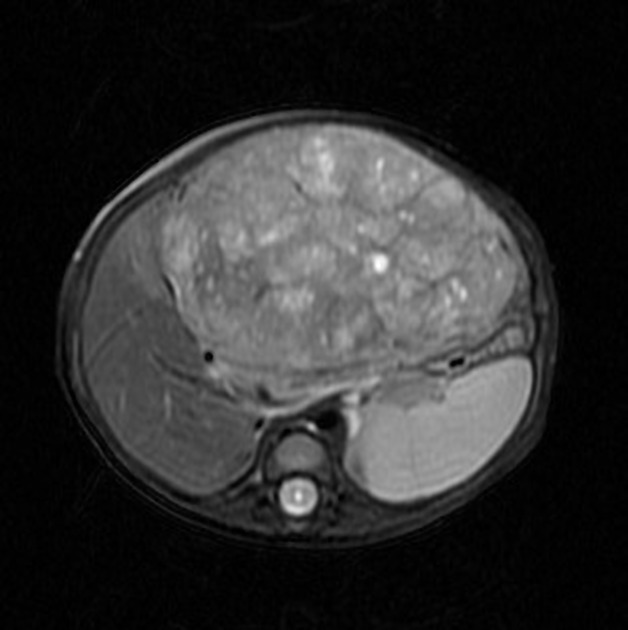
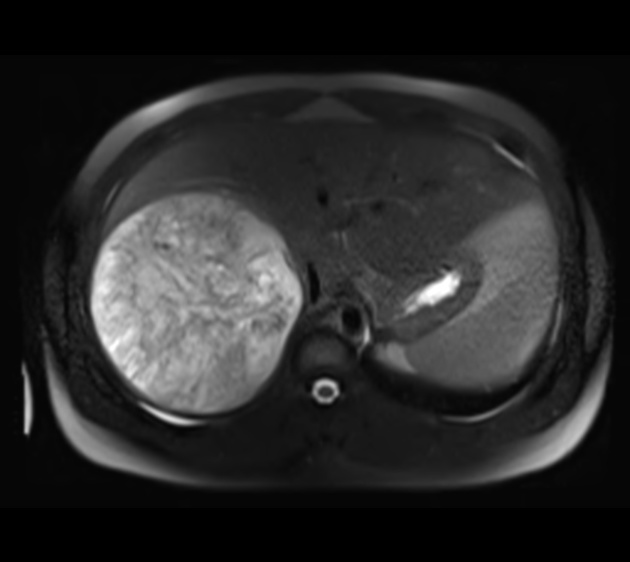
MRI
MRI is superior to CT in defining tumor margins, vessel involvement and adenopathy 1.
T1: generally hypointense
T1 C+ (Gd): can show heterogeneous enhancement
-
T2
generally hyperintense compared to liver
areas of necrosis and hemorrhage are common
Angiography (DSA)
tend to be hypervascular
Treatment and prognosis
Surgical resection is the treatment of choice although preoperative chemotherapy may be used to reduce tumor bulk. Chemotherapy is also employed in following surgical resections. If the tumor is resectable then liver transplantation provides a cure, as long as no metastatic disease is present. The lungs are a relatively frequent site of metastases.
Overall there is 65-70% long-term survival 10, however, the prognosis depends on staging:
stage I: can expect very good long-term survival, of up to 100% with combined chemotherapy and surgery 8,12
stage II: 75-80%
stage III: 65%
stage IV: 0-27% 10
Differential diagnosis
General imaging differential considerations include:
hepatic metastases, e.g. neuroblastoma
rhabdomyosarcoma of biliary tract (rare)





 Unable to process the form. Check for errors and try again.
Unable to process the form. Check for errors and try again.