Neuromyelitis optica spectrum disorder (NMOSD) is a severe demyelinating diseases, which in seropositive cases, is caused by an autoantibody to the aquaporin-4 (AQP4) water channel. The classic presentation of NMOSD is with the triad of bilateral optic neuritis, longitudinally extensive myelitis, and positive anti-AQP4 antibody, although a far wider range of manifestations are now recognized as part of NMOSD 8-13.
On this page:
Terminology
Neuromyelitis optica (NMO) was previously referred to as Devic disease, and traditionally NMO was thought to have limited if any intracranial manifestations. Over the past decade, however, a far wider range of manifestations have been recognized as belonging together and thus the term NMOSD has been proposed to encompass them all.
Epidemiology
Neuromyelitis optica spectrum disorder is typically found in patients somewhat older than those with multiple sclerosis, with an average age of presentation of 41 years, and there is an even stronger female predilection (F:M 6.5:1) 6,8. It is found more frequently in patients of Asian, Indian, and African descent 8.
Diagnosis
The diagnostic criteria are defined by the International Panel for NMO Diagnosis.
Clinical presentation
NMOSD is usually a relapsing condition 5, characterized by bilateral optic neuritis (usually bilateral, but can be unilateral) and myelitis resulting in blindness and myelopathy (paraplegia often with atypical symptoms such as pain and pruritus 27,28) respectively. Although the two classically present concurrently, it is not uncommon for one to precede the other by up to several weeks, or to occur each as standalone clinical presentations 3.
In addition, other clinical syndromes may be present:
brainstem syndrome
diencephalic syndrome (e.g. narcolepsy, autonomic dysfunction)
cerebral syndrome
Furthermore, NMOSD encompasses non-neurological manifestations in anti-AQP4 antibody seropositive patients including systemic lupus erythematosus (SLE) and Sjögren syndrome 13.
Pathology
In approximately 70% (sensitivity of 70-90%; specificity of 90%) of patients with established NMOSD, a specific immunoglobulin can be isolated (anti-AQP4-IgG) which targets a transmembrane water channel (aquaporin-4) present on astrocyte foot processes abutting the limiting membrane 5,8. This accounts for some of the predilection for the circumventricular organs (e.g. periaqueductal grey matter) which are particularly rich in aquaporin-4 8.
Early in the disease, demyelinating regions will demonstrate similar findings to multiple sclerosis, such as macrophage/microglia activation and axonal damage. Additionally, however, and relatively specific for NMOSD, these regions will also demonstrate extensive eosinophilic infiltration, perivascular deposition of immunoglobulins (especially IgM) and local activation of the complement cascade 3. Another differentiating feature is that axonal damage precedes demyelination in NMOSD 5.
Generally, the condition is sporadic, although some overlap in immunogenic features between certain viruses and aquaporin-4 water channel have been identified 8.
Radiographic features
MRI
MRI is the modality of choice for NMOSD and the orbits, brain and spinal cord should be imaged in suspected cases.
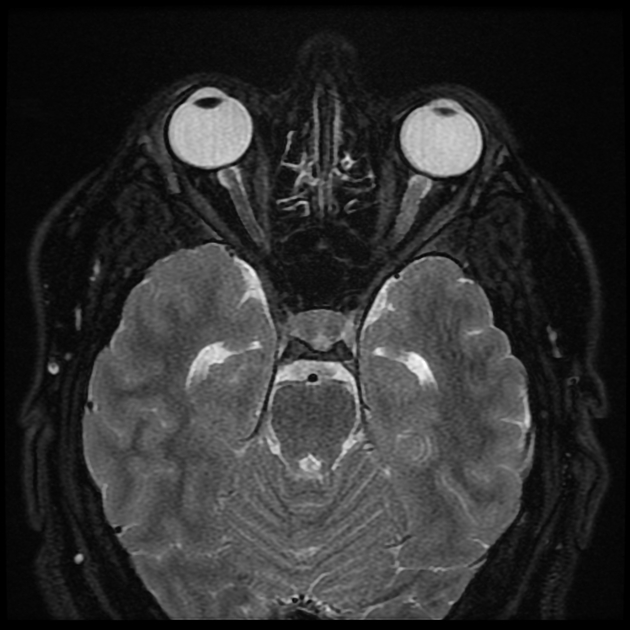
Orbits
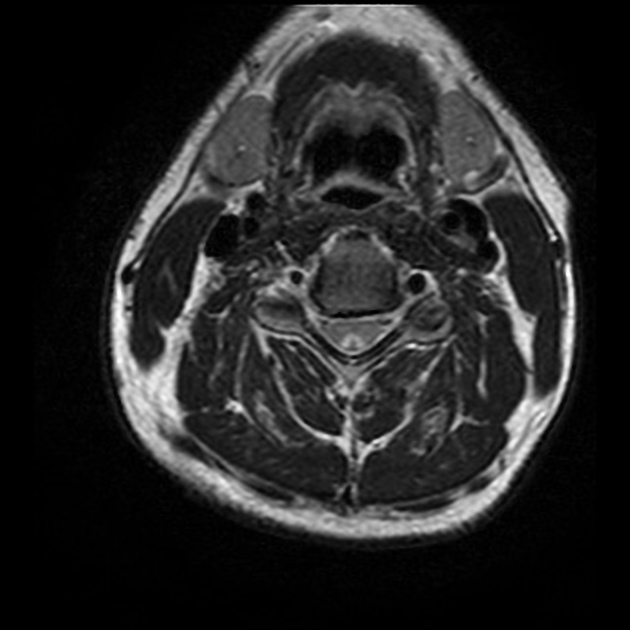
Targeted imaging of the orbits (including fat-saturated T1 C+ (Gd) and T2 weighted sequences (see MRI protocol: orbit) may demonstrate typical features of optic neuritis:
optic nerves appearing hyperintense and swollen on T2 weighted sequences and enhancing on T1 C+ (Gd)
bilateral optic nerve involvement and extension of the abnormal signal posteriorly as far as the chiasm is particularly suggestive of NMOSD 5
atrophy of the optic nerves with associated hyperintensities on T2 weighted sequences may be seen in chronic stages of the disease 11
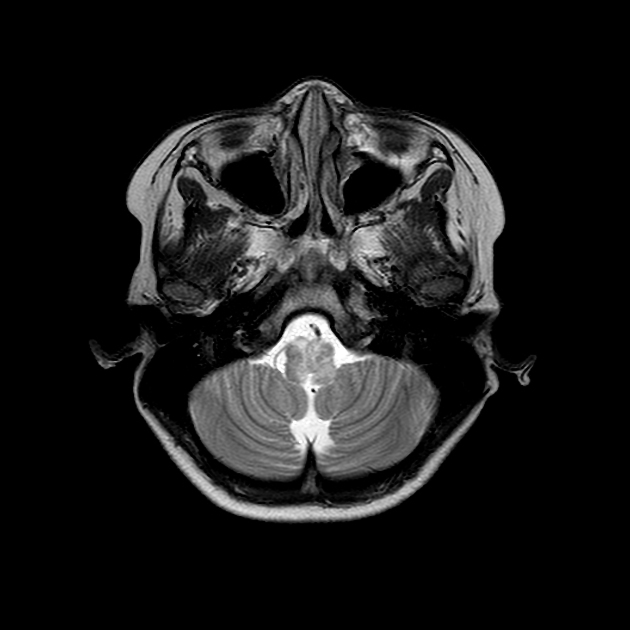
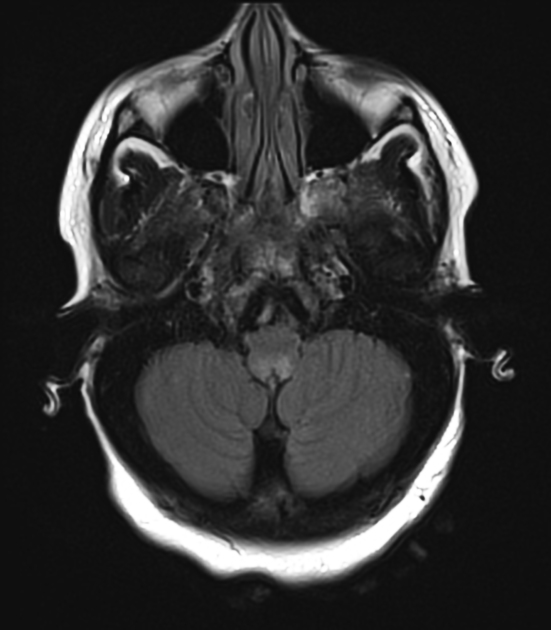
Brain
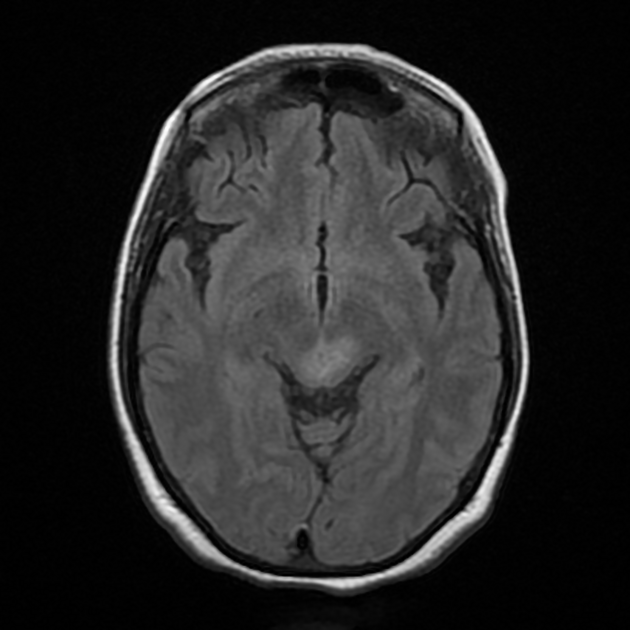
Although traditionally NMOSD was thought to have normal intracranial appearance it is increasingly evident that asymptomatic abnormalities are present in the majority of seropositive NMOSD patients. These can be divided into four categories 5,8,13:
-
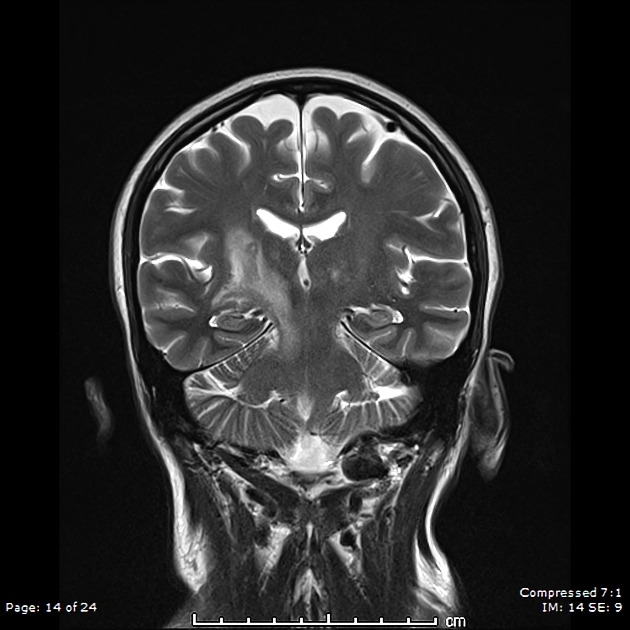
lesions which mirror the distribution of aquaporin-4 in the brain, which is particularly found in the periependymal regions abutting the ventricles:
periventricular (hemispheric) confluent smooth sessile white matter involvement (unlike MS, there are usually no Dawson fingers)
periaqueductal grey matter
hypothalamus/medial thalamus
-
multiple callosal lesions with heterogeneous signal leading to a marbled pattern 7
the splenium may be diffusely involved and expanded
deep (or less frequently subcortical) punctate white matter lesions (which may appear similar to those seen in multiple sclerosis)
corticospinal tract involvement by extensive longitudinal lesions; has been reported more frequently in Korean patients 5
-
larger >3 cm diameter hemispheric white matter lesions
radially oriented (spilled-ink appearances) or spindle-shaped
limited if any mass effect although tumefactive lesions do occur 13
may involve overlying cortex 14
facilitated diffusion
may be more common in children and in patients from the Far East and Africa
often vanish but cystic change is seen in a minority of cases 13
Additionally, in patients with longstanding disease, often with a history of numerous clinical attacks, rarely a more extensive, confluent, bilateral, symmetric pattern of T2/FLAIR hyperintensities may be seen, which can involve subcortical and deep white matter, external capsules, anterior temporal lobe, and cerebellum 25.
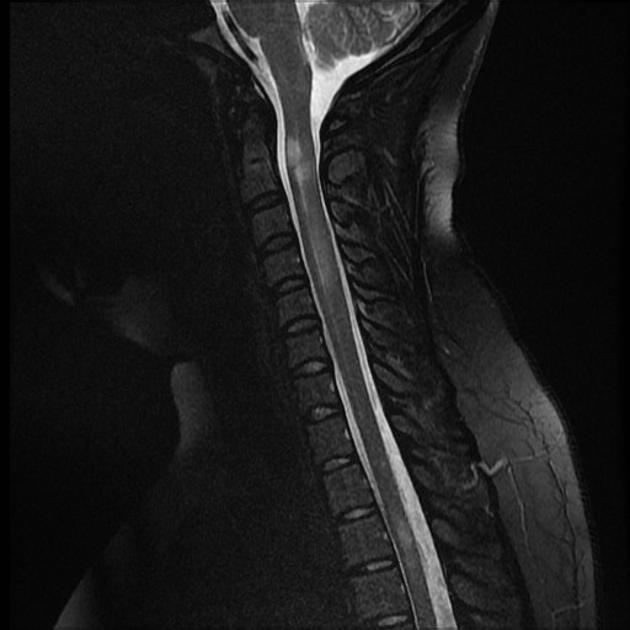
Spinal cord
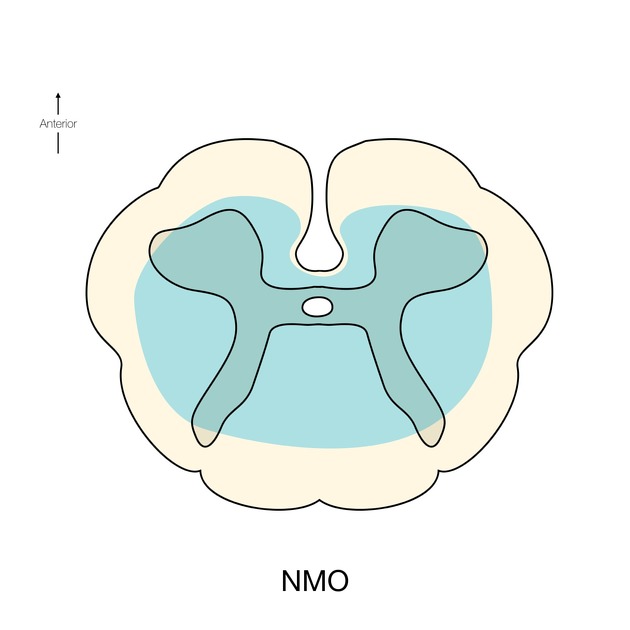
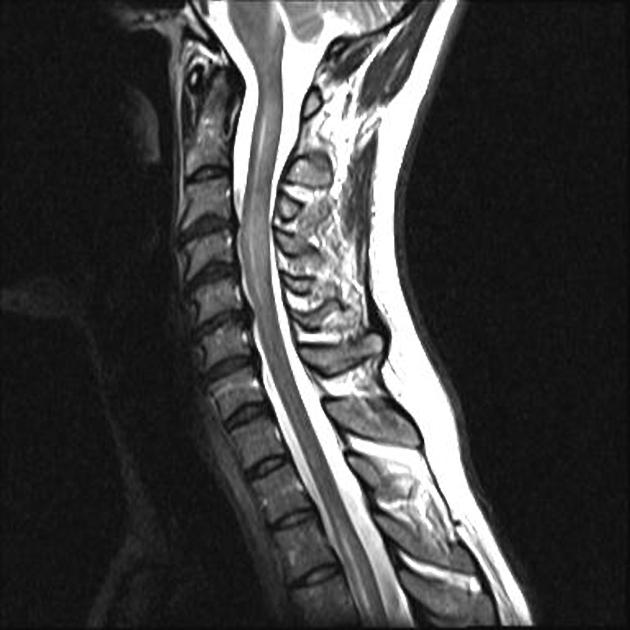
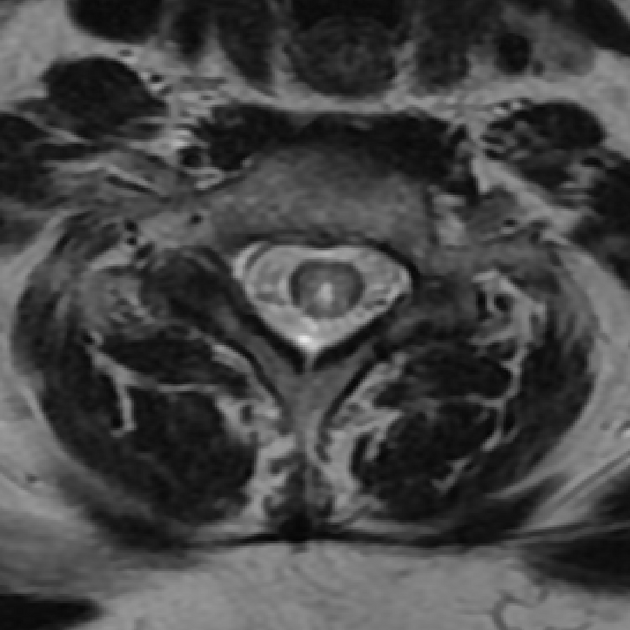
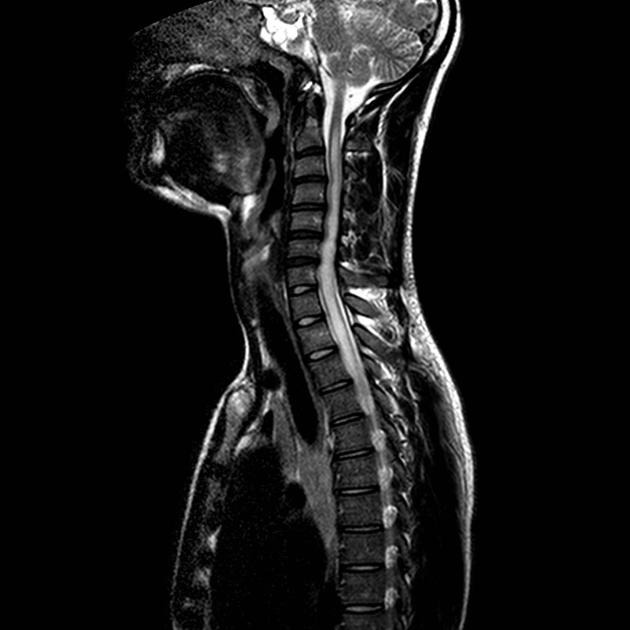
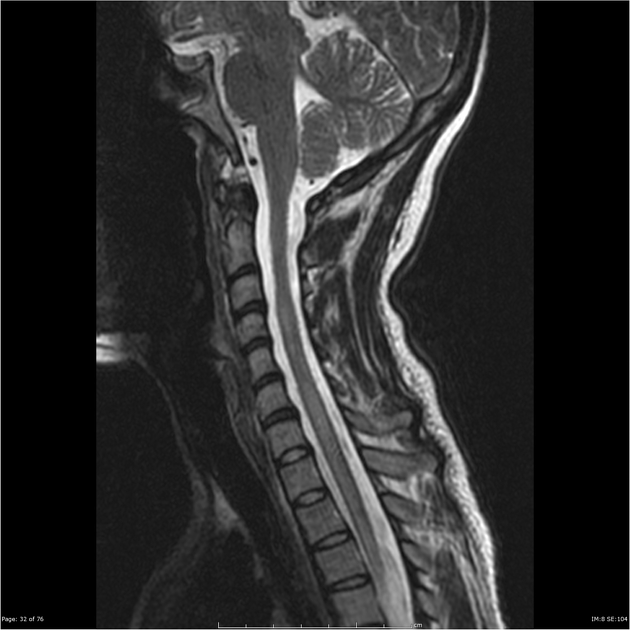
Spinal cord involvement is extensive, typically with a high T2 signal spanning at least three vertebral segments, known as a longitudinally extensive spinal cord lesion 4,5,8,11. Cord swelling is usually present in the acute phase. Although less common, short segment transverse myelitis is seen in ~15% of cases 11.
Imaging features include 5,8:
-
T1
hypointense
follow-up scans may demonstrate cord atrophy and low T1 signal 5
-
T2
hyperintense (often >3 vertebral body lengths), including highly specific bright spotty lesions 26
central grey matter involvement is typical, but white matter can also be involved
-
T1 C+ (Gd)
enhancement is common and variable in appearance
-
ring-enhancement
seen in a third of patients 17
both on sagittal and axial imaging
ring often extends over multiple vertebral levels
patchy "cloud-like" enhancement of the aforementioned T2 bright lesions may be present
thin ependymal enhancement similar to ependymitis
lens-shaped enhancement on sagittal images 11
Treatment and prognosis
Treatment of NMOSD with immunosuppression is necessary to reduce relapses 5. Immunosuppressive options may include traditional immunosuppressants (e.g. prednisolone, azathioprine, mycophenolate mofetil, etc.) and/or biologic agents:
eculizumab or ravulizumab: C5 inhibitors 19,20
inebilizumab: CD19 monoclonal antibody 21
satralizumab or tocilizumab: IL-6 receptor antagonist 22,24
rituximab: CD20 monoclonal antibody 23
It is important to distinguish NMOSD from multiple sclerosis as the treatment is not only different but treating a patient with NMOSD with multiple sclerosis-specific therapies (e.g. beta-interferon or natalizumab) can actually lead to its exacerbation 5.
Patients with a relapsing course have a poorer prognosis 4:
blind in one or both eyes: monophasic 22% vs relapsing 60%
monoplegia or paraplegia: monophasic 31% vs relapsing 52%
History and etymology
The disease was first described in 1870 by Sir Thomas Clifford Allbutt 12. The French term "neuromyélite optique aiguë" (literally acute optic neuromyelitis) was later coined by Eugène Devic and Fernand Gault in 1894 in a case series of 16 patients 12. At the time, the disorder was thought to be acute and monophasic.
For many years, it was considered to be a subtype of multiple sclerosis, until the discovery of anti-AQP4 antibodies at which time it was moved into its own disease category. It is now evident that a significant proportion of patients with clinical NMOSD do not have the anti-AQP4 antibody (some have anti-MOG antibodies for example) and that the presentation can be more heterogeneous.
Differential diagnosis
The differential diagnosis depends on the presentation, and when classic, the diagnosis can be made with a fair degree of certainty. The most important clinical and imaging differential is multiple sclerosis as both can present with optic neuritis, cerebral and spinal demyelination including involvement of the corpus callosum (see Practical points).
Another key differential diagnosis is myelin oligodendrocyte glycoprotein antibody-associated disease (MOGAD), which can present very similarly. Individuals with AQP4-IgG-positive NMOSD and optic neuritis tend to have longer-segment less prominent involvement of the optic nerves, more frequently involving the optic chiasm and optic tracts 18.
More generally, for patients with cerebral involvement, the differential is broad and includes:
myelin oligodendrocyte glycoprotein antibody-associated disease (MOGAD)
Susac syndrome: involves the central portion of the corpus callosum
neuro-Behçet: mesodiencephalic involvement is typical
acute disseminated encephalomyelitis (ADEM): grey-white matter involvement with a more tumefactive appearance
For patients with spinal cord involvement, the differential is that of a longitudinally extensive spinal cord lesion. For the typical bright spotty lesion, an important differential diagnosis is acute spinal cord infarction.
Practical points
There are many features that distinguish NMOSD from multiple sclerosis, although none are pathognomonic. It should also be noted that overlap of imaging findings of these two entities may be higher in Asian populations 15. Nonetheless, features that are helpful in favoring NMOSD over multiple sclerosis include 5,7,15:
-
brain
smooth confluent periependymal distribution (see above)
fewer oval perivenular orientation of periventricular lesions (no Dawson fingers)
fewer juxtacortical lesions (U-fiber)
more extensive involvement of the corpus callosum (especially its ependymal surface)
larger, more confluent lesions
lack of open ring enhancement
corticospinal tract and diencephalic involvement (see above)
-
spinal cord
-
more longitudinally extensive spinal cord lesion
preferential involvement of the central cord rather than the peripheral cord
-
-
optic nerves
-
more longitudinally extensive optic neuritis
with preferential involvement of the posterior optic pathway
-





 Unable to process the form. Check for errors and try again.
Unable to process the form. Check for errors and try again.