Osteoid osteomas are benign bone-forming tumors that typically occur in children (particularly adolescents). They have a characteristic lucent nidus <1.5 or 2 cm and surrounding osteosclerotic reaction, which classically causes night pain that is relieved by the use of NSAIDs.
On this page:
Epidemiology
Osteoid osteomas are usually found in children, adolescents, and young adults between the ages of 10 and 35 years 2.
They account for ~10% of all benign bone lesions, and there is a male predilection (M:F 2-4:1) 2.
Diagnosis
Diagnostic criteria according to the WHO classification of soft tissue and bone tumors (5th edition) 10:
-
essential
typical imaging appearances with nidus <2 cm +/- surrounding sclerosis
histological evidence of woven bone formed by microtrabeculae with a rim of vascularized stroma containing plump osteoblasts
-
desirable
nocturnal pain relieved by NSAIDs
narrow transition zone between the central nidus and surrounding sclerosis
Clinical presentation
Classically, patients present with nocturnal pain that is relieved by aspirin/NSAIDs (aspirin is contraindicated in children <12 years old due to risk of Reye syndrome).
When in the spine, they are a classic cause of painful scoliosis, concave on the side of the lesion. This typical presentation is seen in over 75% of cases 2.
Soft tissue swelling may occur, and if it is close to a growth plate, accelerated growth may be evident 2, presumably related to hyperemia.
When the lesion is intracapsular, the presentation is more atypical and more likely to mimic inflammatory arthropathy or synovitis. Joint effusion is often present 2,4.
Pathology
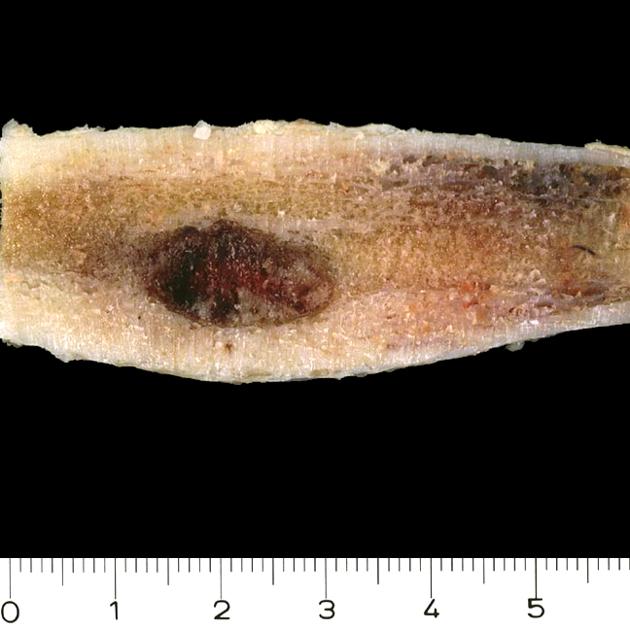
An osteoid osteoma is composed of three concentric parts 1:
-
nidus, representing the neoplastic process
meshwork of dilated vessels, osteoblasts, osteoid, and woven bone
may have a central region of mineralization 2
fibrovascular rim
surrounding reactive sclerosis
The nidus releases prostaglandins (via the enzymes cyclo-oxygenase-1 and cyclo-oxygenase-2), resulting in pain 2.
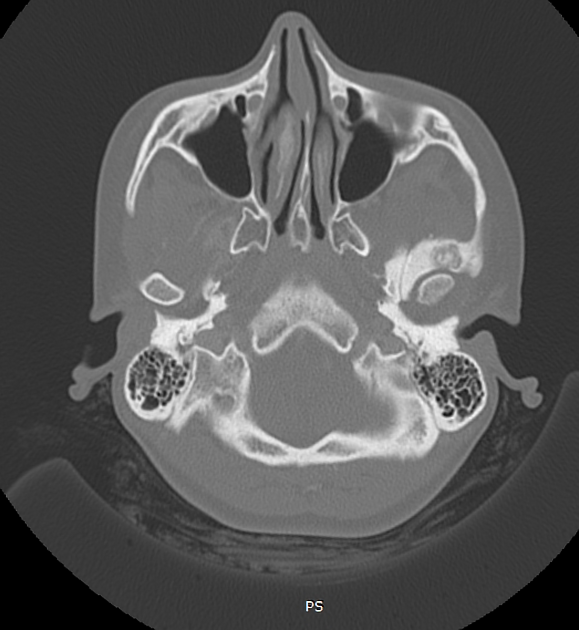
Location
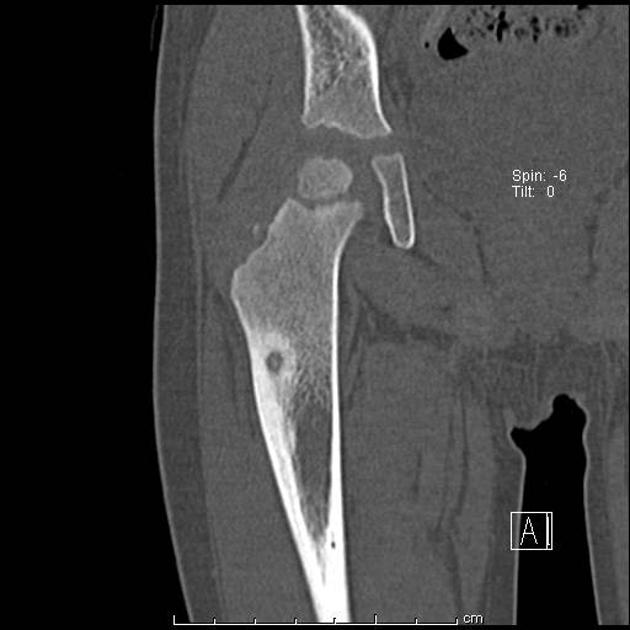
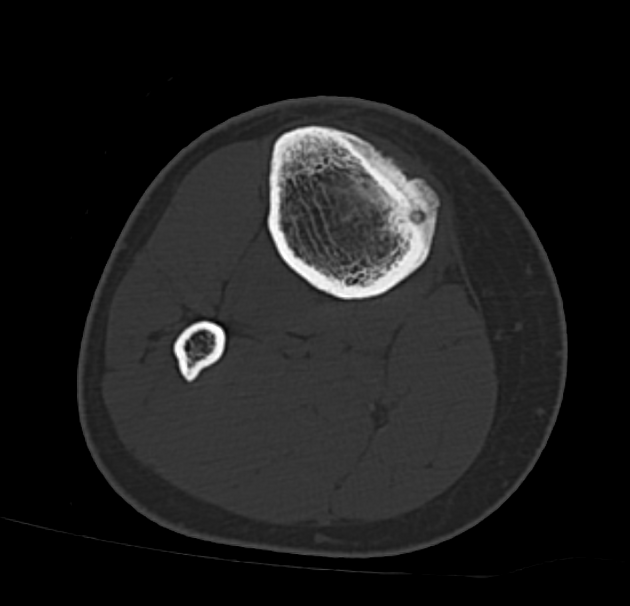
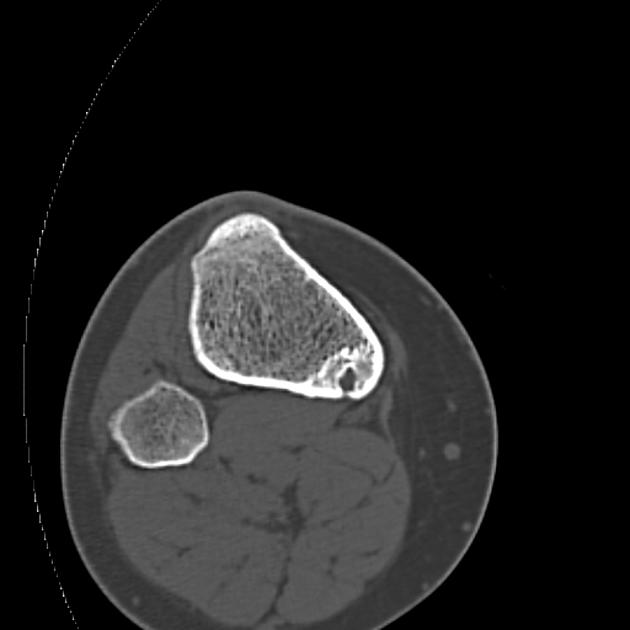
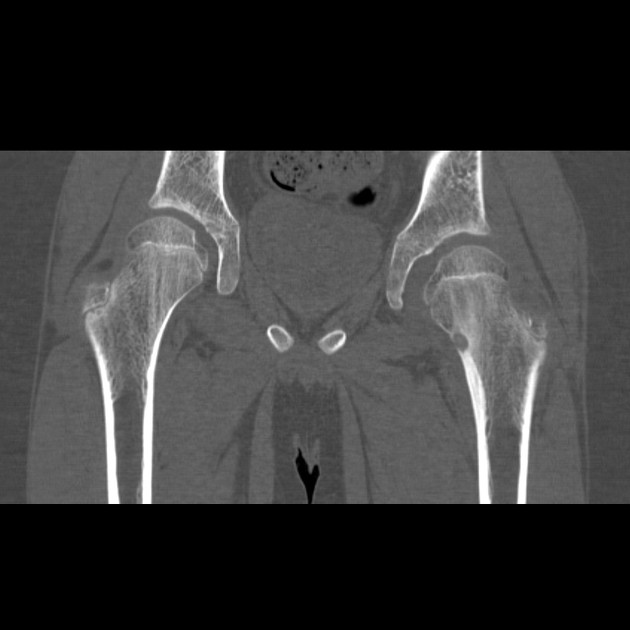
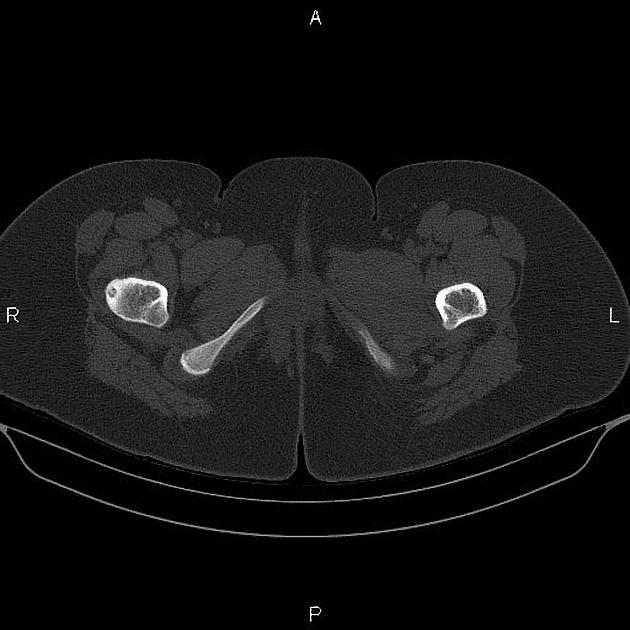
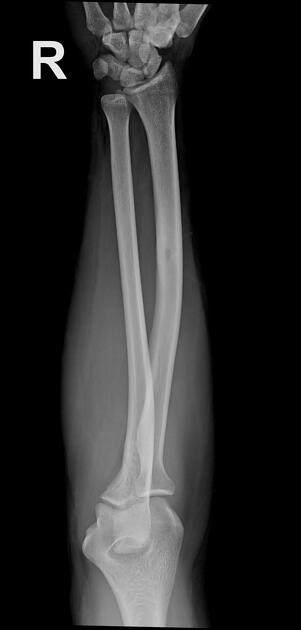
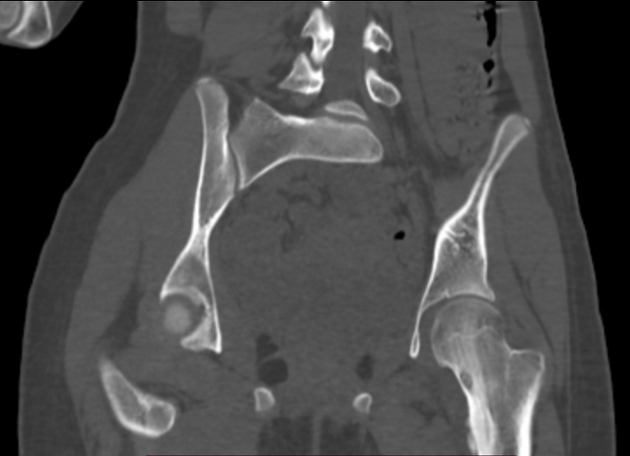
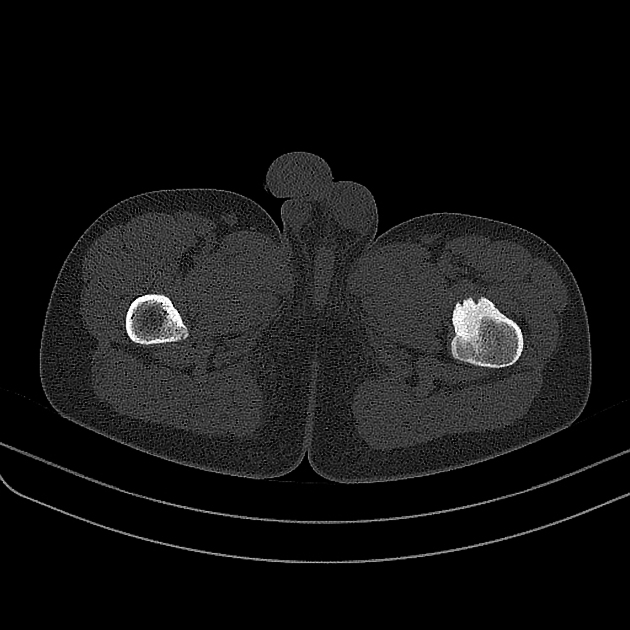
Most osteoid osteomas occur in long tubular bones of the limbs (especially the proximal femur), but any bone may be involved.
-
long bones of the limbs: ~65-80% 1,2,7
femur most common (especially neck of femur)
mid-tibial diaphysis is also common
phalanges: ~20%
-
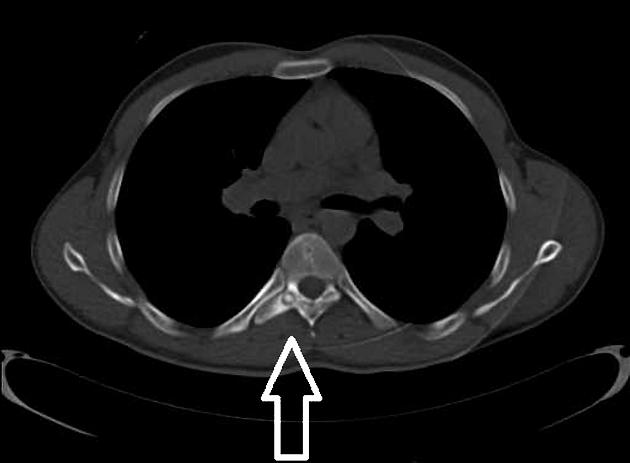
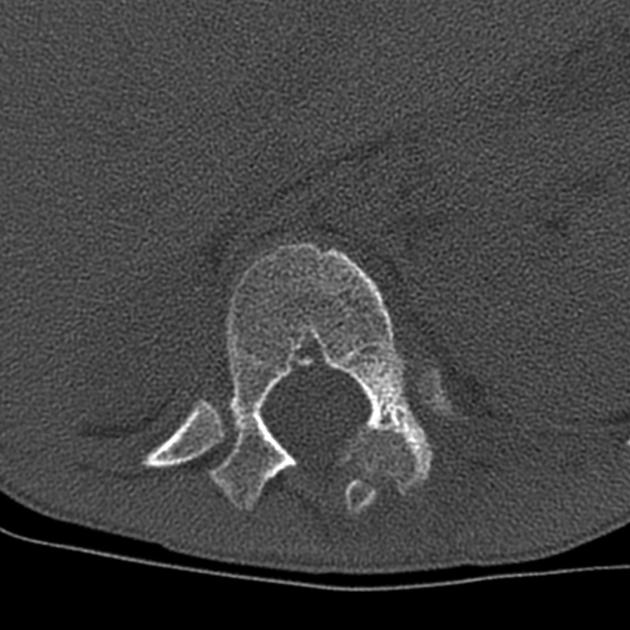
vertebrae: ~10%, predominantly posterior elements
lumbar: 59% 2
cervical: 27%
thoracic: 12%
sacrum: 2%
Furthermore, osteoid osteomas are usually cortical lesions but they can occur anywhere within the bone including medullary, subperiosteal (most commonly in the talus), and intracapsular 2. In intracapsular osteoid osteomas, periosteal reaction may be distant from the lesion itself 9.
Genetics
FOS gene rearrangement is present in ~90% of cases (similar to osteoblastomas) 10
Radiographic features
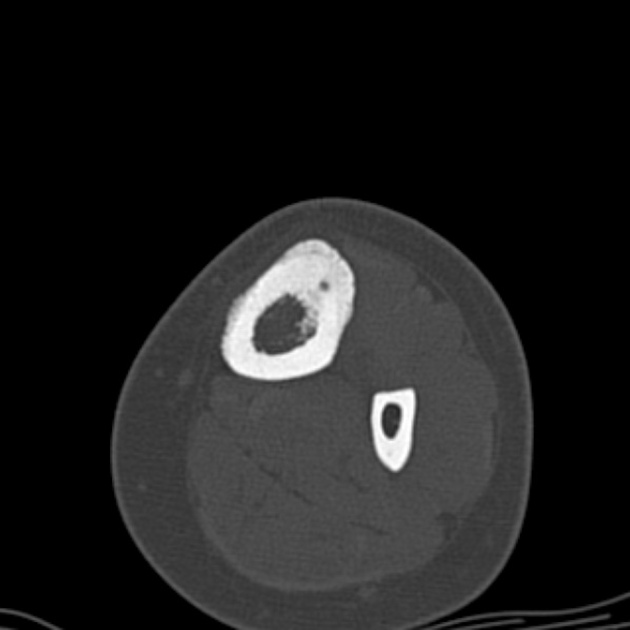
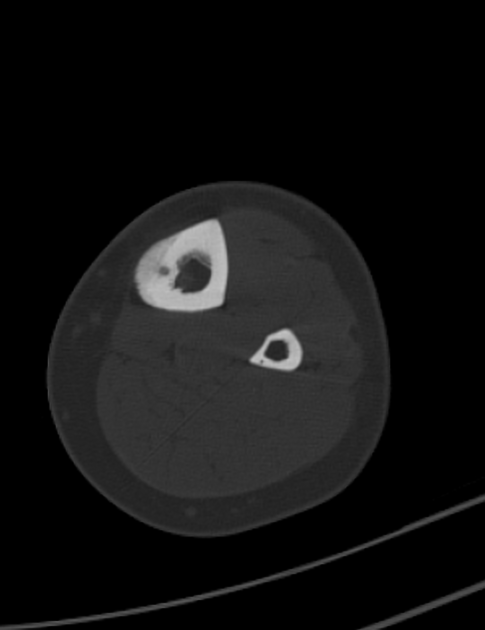
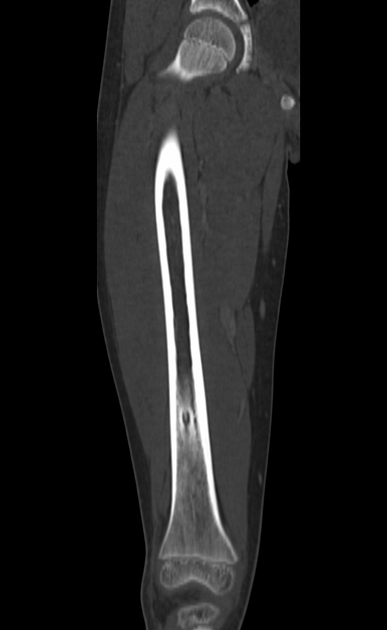
It is important to remember that the sclerosis is reactive and does not represent the lesion itself. The nidus is usually <2 cm in diameter and is typically ovoid. It may have a central region of mineralization 2.
Plain radiograph
May be normal or may show a solid periosteal reaction with cortical thickening. The nidus is sometimes visible as a well-circumscribed lucent region, occasionally with a central sclerotic dot. However, dense sclerosis may sometimes obscure the nidus.
Ultrasound
On ultrasound, focal cortical irregularity with adjacent hypoechoic synovitis may be present at the site of intra-articular lesions. The nidus can show hypoechogenicity with posterior acoustic enhancement. Ultrasound may be able to identify the nidus as a hypervascular nidus on Doppler examination 2.
CT
CT is excellent at characterizing the lesion and is the modality of choice. It typically shows a focally lucent nidus within surrounding sclerotic reactive bone. A central sclerotic dot may also be seen.
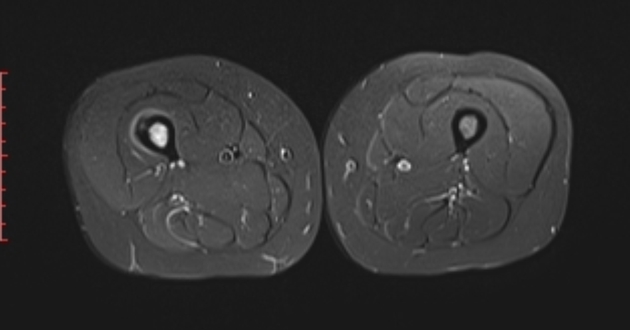
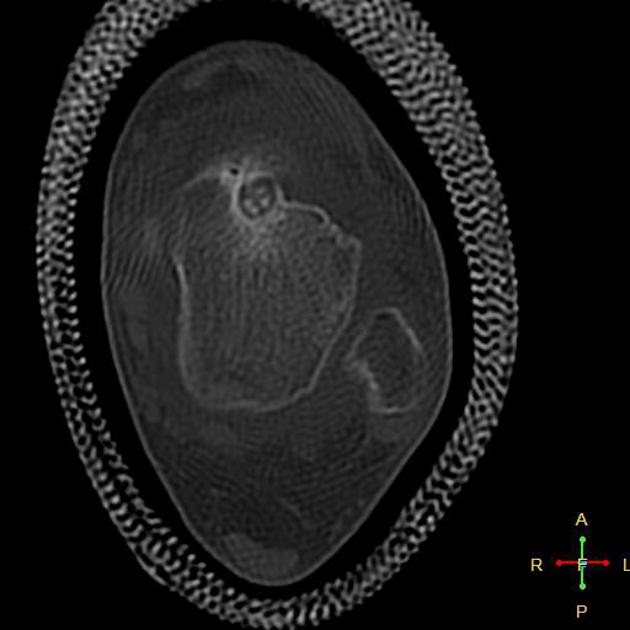
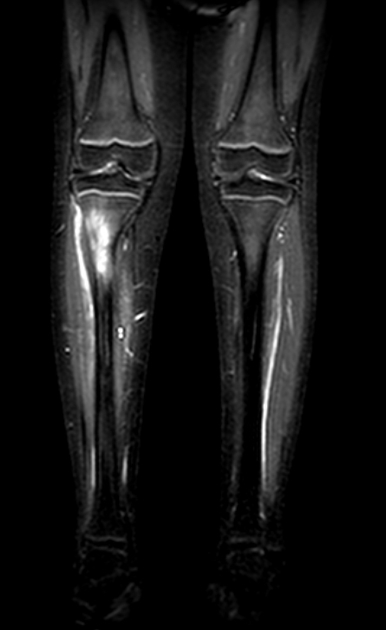
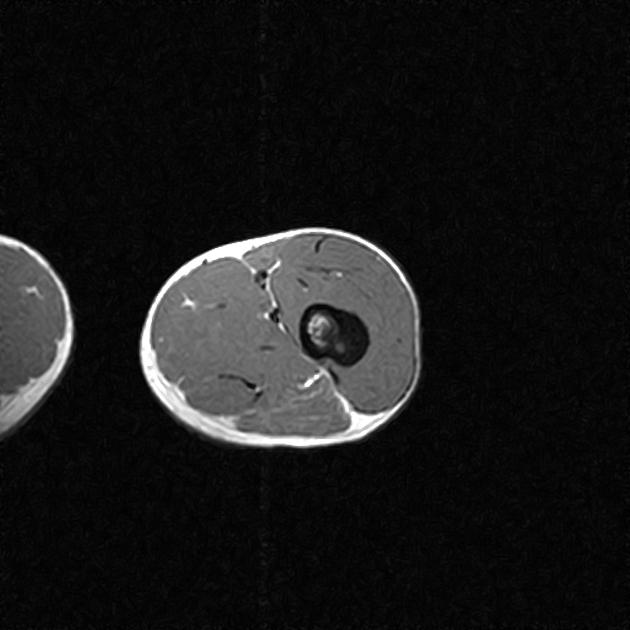
MRI
Although MRI is sensitive, it is non-specific and is often unable to identify the nidus. The hyperemia and resultant bone marrow edema pattern may result in the scans being misinterpreted as representing aggressive pathology 2.
The signal intensity of the nidus is variable on all sequences as is the degree of contrast enhancement 2.
In patients without a history of lower limb overuse, the identification of the half-moon sign is highly specific and sensitive for the detection of femoral neck osteoid osteoma 11. In these cases, detection of half-moon sign on fluid-sensitive MRI sequences should prompt further CT examination to detect a nidus. In cases of overuse (e.g. marathon runners, armed forces members), half-moon sign may indicate a stress reaction/fracture 12.
Nuclear medicine
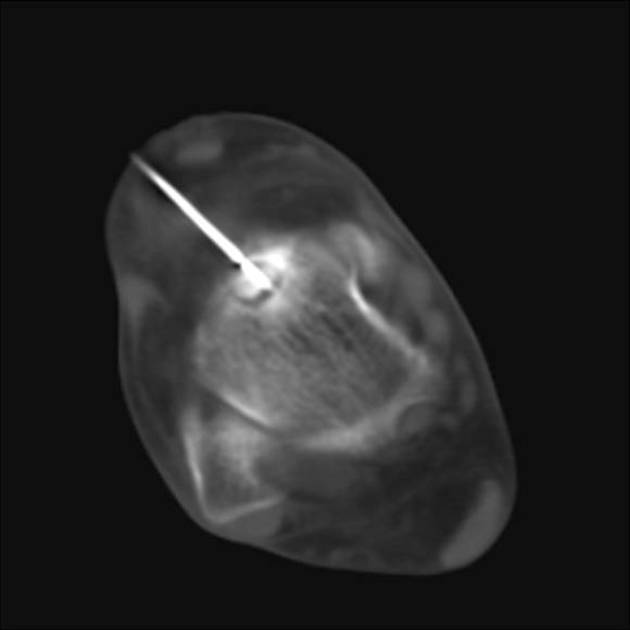
Bone scintigraphy will show typical focal uptake and, at times, will show a double density sign (also known as the less catchy hotter spot within the hot area sign), which, if present, is highly specific and helpful in distinguishing it from osteomyelitis. The central focus shows intense uptake within a surrounding lower, but nonetheless increased, uptake rim.
Treatment and prognosis
The lesion is benign and treatment has traditionally been with surgical resection. Historically, this has been, on occasion, difficult because of the inherent inability to locate the nidus during surgery 3. Surgery has been largely replaced by image-guided procedures such as percutaneous radiofrequency ablation, microwave ablation (MWA), cryoablation, core drill excision, high-intensity focussed ultrasound or laser photocoagulation 10.
There is growing evidence, that osteoid osteoma naturally resolves spontaneously with time and can be treated conservatively with NSAIDs in certain groups of patients 6. The average time to resolution is 33 months 13. Prognosis is considered excellent with recurrences being uncommon 10.
Differential diagnosis
General imaging differential considerations include:
osteomyelitis (e.g. Brodie abscess): bone scan demonstrates a central area of reduced uptake representing an avascular area of purulent material
osteoblastoma: >1.5-2 cm in size
-
intraosseous migration of intratendinous calcification
this condition is generally located at tendon insertions and soft tissue calcifications are often seen








 Unable to process the form. Check for errors and try again.
Unable to process the form. Check for errors and try again.