Pleomorphic xanthoastrocytomas (PXA) are an uncommon circumscribed astrocytic tumor found in young patients and can be WHO grade 2 or 3. They are often considered part of the heterogeneous group of tumors known as long-term epilepsy-associated tumors (LEATs) 9.
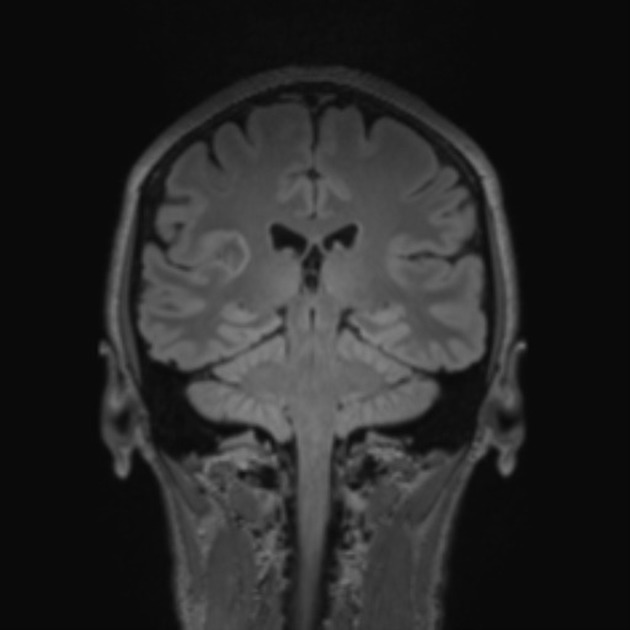
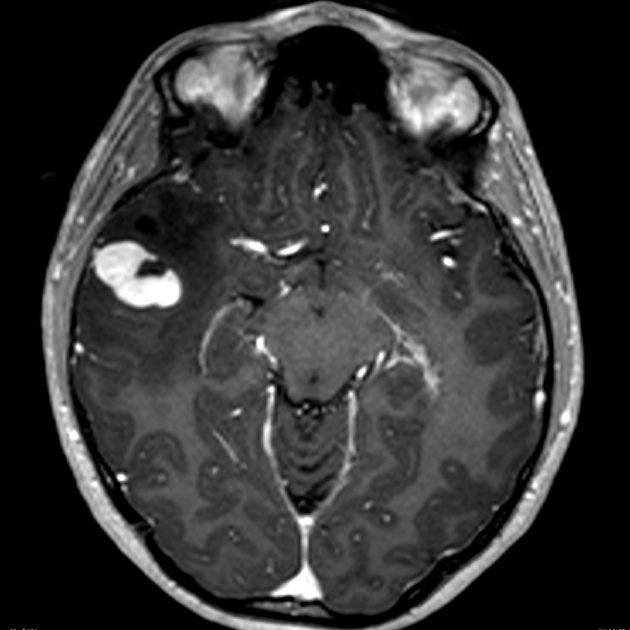
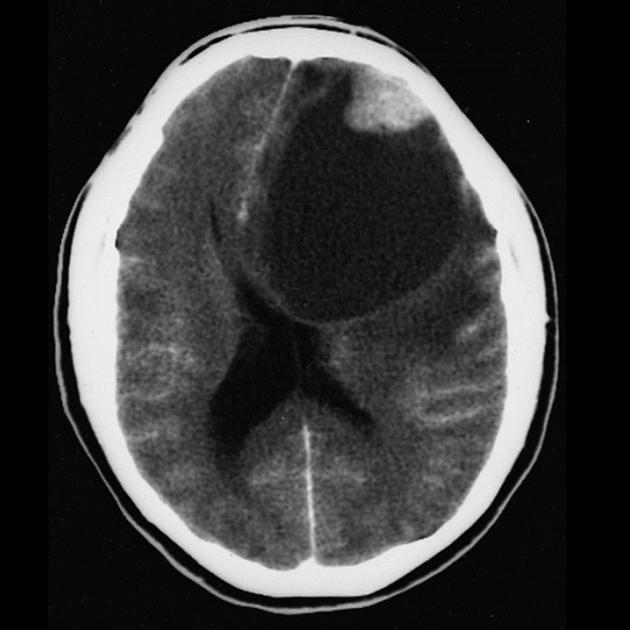
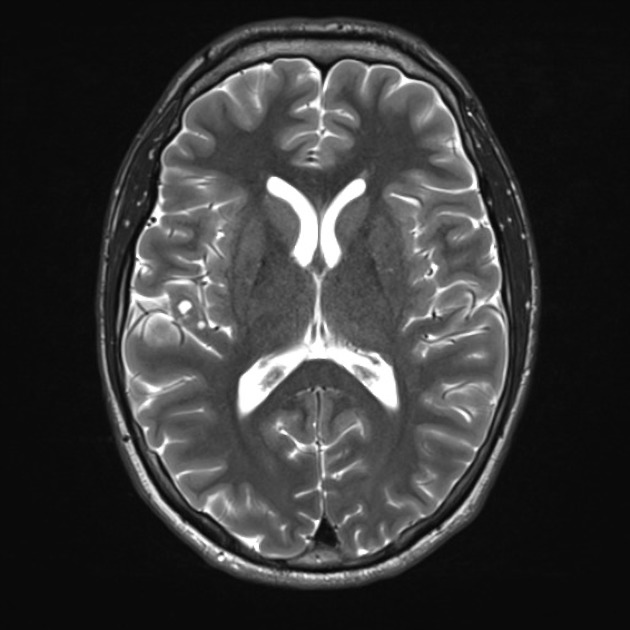
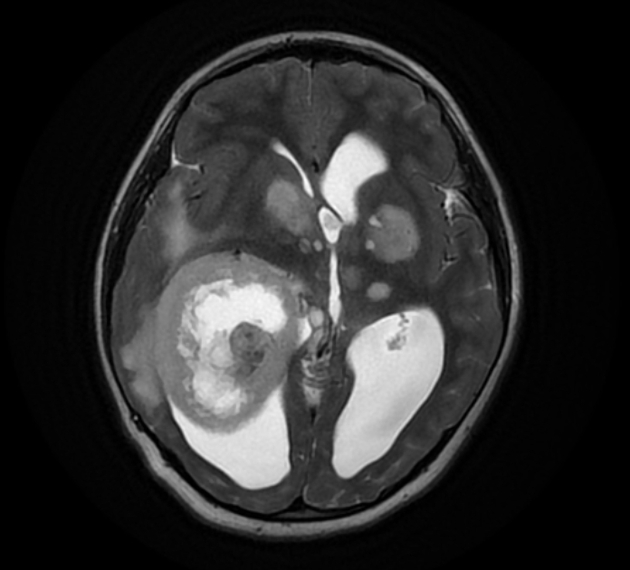
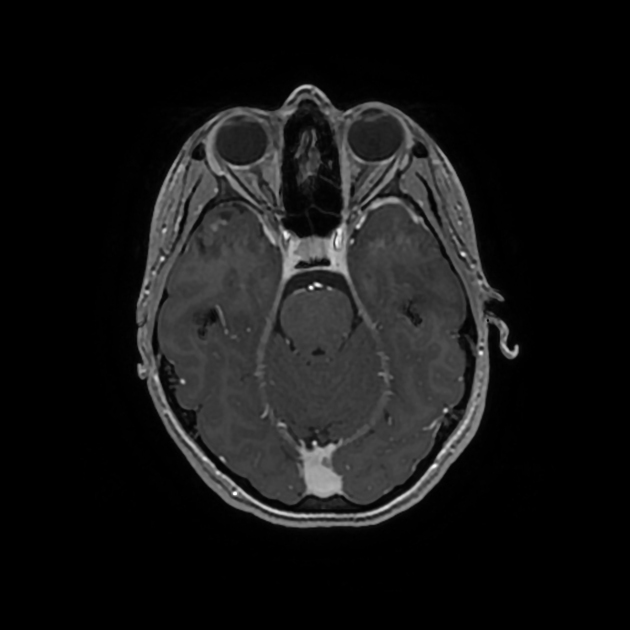
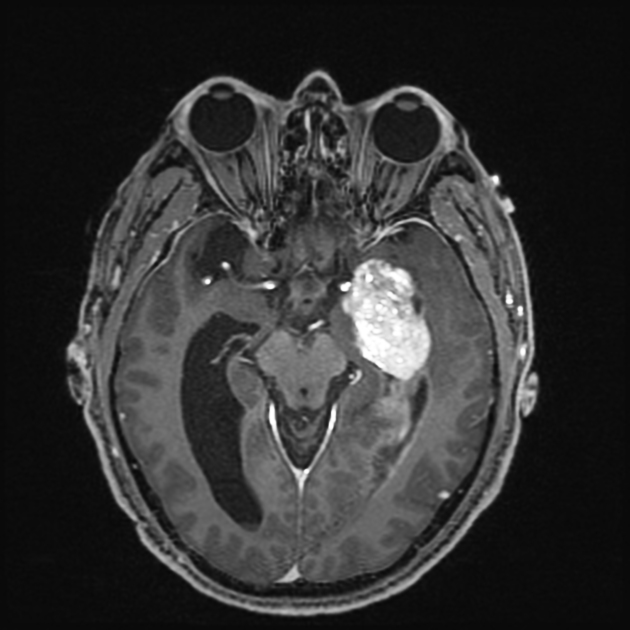
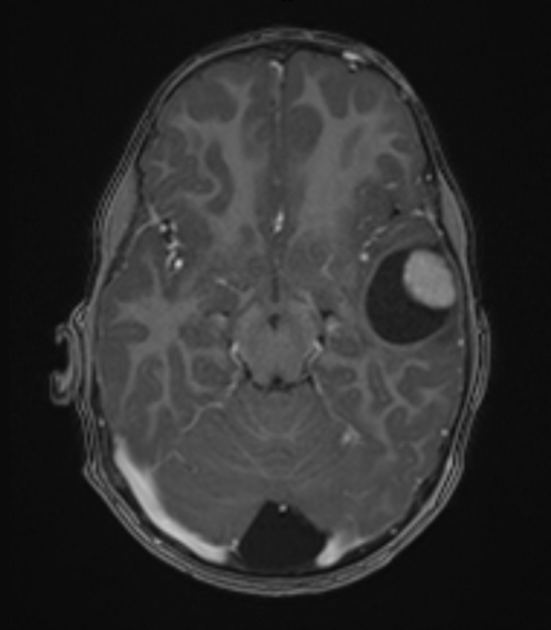
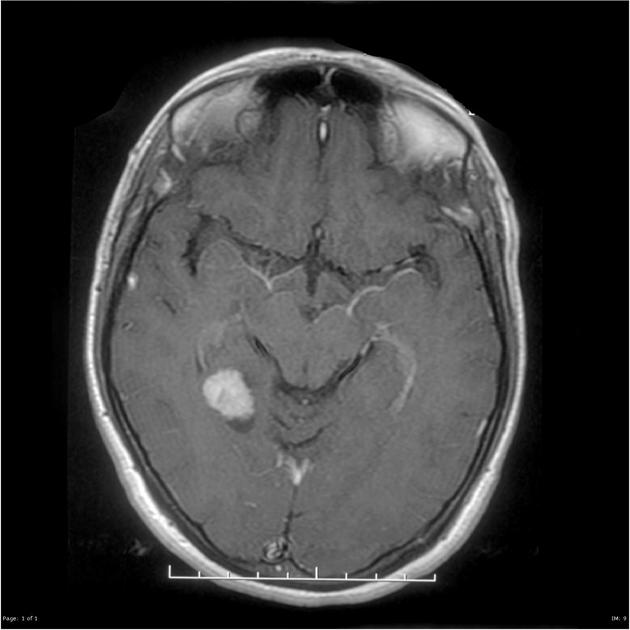
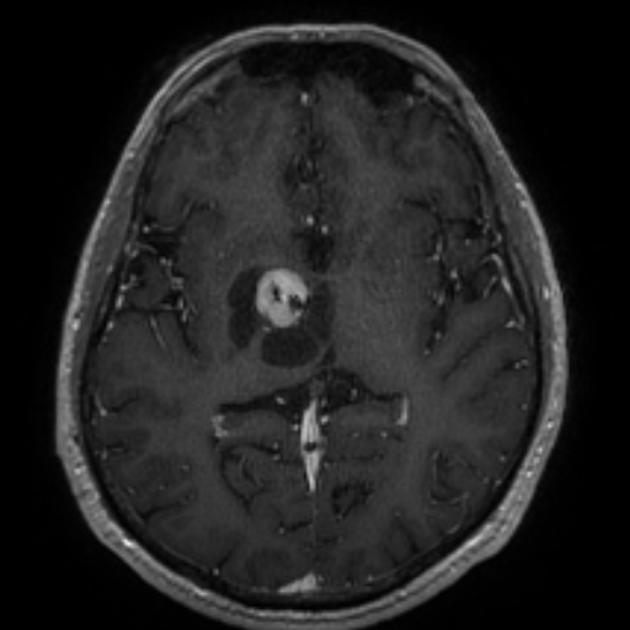
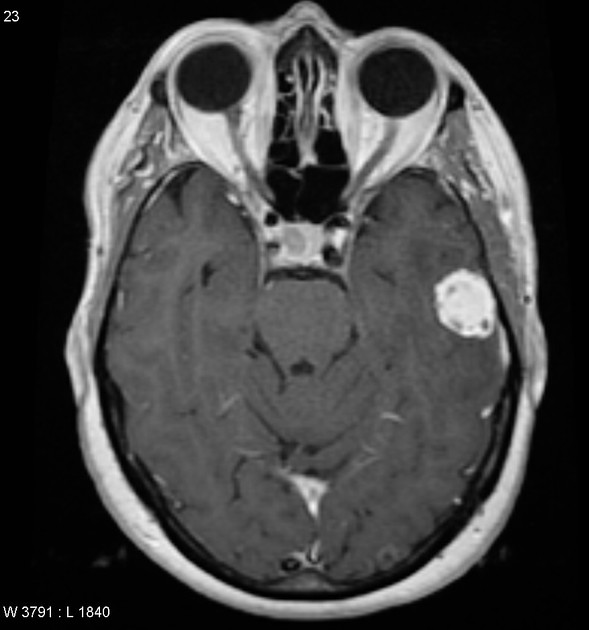
Pleomorphic xanthoastrocytomas most often appear as cortical tumors with a cystic component and vivid contrast enhancement, most commonly in the temporal lobe where they present with temporal lobe epilepsy. Features of slow growth may be present, such as no surrounding edema and scalloping of the overlying bone. A reactive dural involvement expressed by a dural tail sign can be found. Calcifications are rare.
On this page:
Epidemiology
They are rare tumors accounting for only 1% of primary brain tumors 2,3,6. Typically these tumors are found in young patients (children or young adults), with a peak incidence in the 2nd to 3rd decades of life (10-30 years) 6.
Associations
The only reported association is with neurofibromatosis type 1, although this is not a strong association 6.
Clinical presentation
As these tumors have a predilection for the temporal lobe they most frequently present with seizures and focal epilepsy (~75% of cases) 1,2. Other findings include dizziness, and headache, or rarely, patients are asymptomatic 5.
Pathology
Grading
Pleomorphic xanthoastrocytomas are considered either WHO grade 2 or 3 tumors based on histological features 8.
Grade 3 tumors (previously known as anaplastic pleomorphic astrocytomas) have an increased mitotic rate of >5 mitoses per 10 high-power fields but are otherwise histologically very similar with grade 2 tumors, although necrosis and vascular proliferation are more common 6. They can be difficult to distinguish from epithelioid glioblastomas 6.
Location
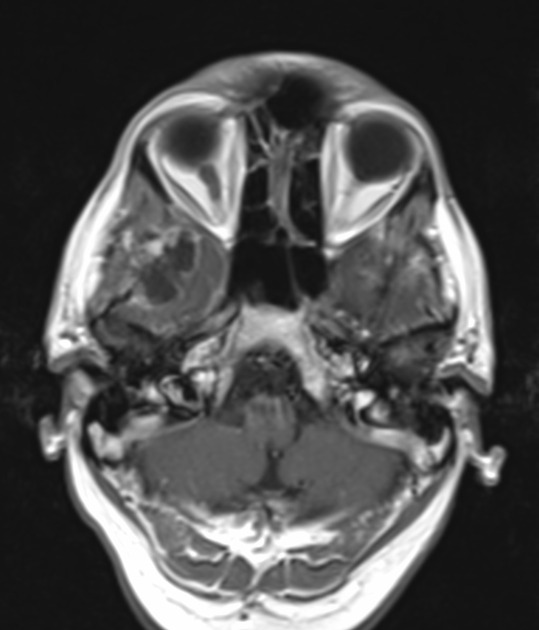
Pleomorphic xanthoastrocytomas are almost invariably (98%) located supratentorially, typically located superficially (peripherally) abutting the leptomeninges, involving the cortex and overlying leptomeninges but actual dural involvement is rare. Approximately half are located in the temporal lobe, with the remainder of the lesions more common in the frontal than the parietal lobes 4,6.
Macroscopic appearance
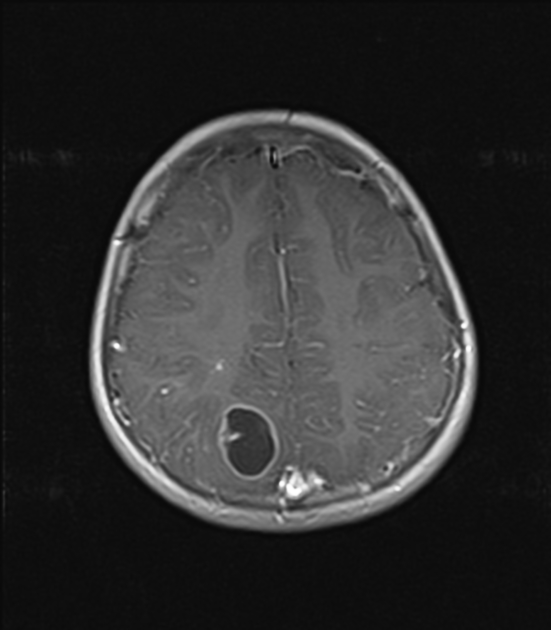
Macroscopically these tumors appear well-circumscribed, often with a cystic component and involvement of the overlying leptomeninges 1,3.
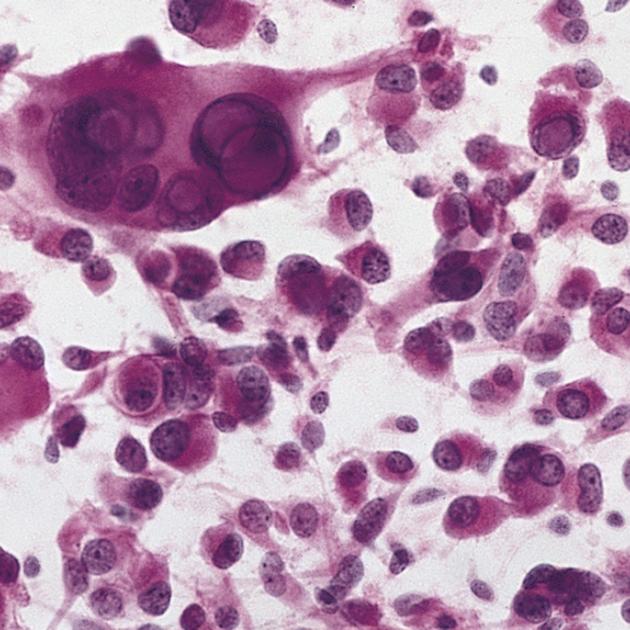
Microscopic appearance
Microscopically the margins are not as well defined. The histological features are variable (thus the term "pleomorphic") with spindle cells, polygonal cells, multinucleated cells and lipid-laden xanthomatous astrocytes are all identified 6. Even more pleomorphic is the appearance of the nuclei, with common nuclear inclusions, and highly variable nuclear size 6.
Immunophenotype
Immunohistochemistry demonstrates expected glial marker reactivity. Less obviously, there is also variable reactivity for neuronal markers 3,6:
GFAP: positive, although often only weakly
S100: positive
neuronal markers including synaptophysin, MAP2 and neurofilament: variable
Ki-67 proliferation index: <1% 6
Genetics
Pleomorphic xanthoastrocytomas, as well as pilocytic astrocytomas (and many non-CNS tumors), exhibit BRAF mutations 6,7.
Radiographic features
Pleomorphic xanthoastrocytomas appear as a solid enhancing nodule, frequently with a peripheral eccentric cystic component (50-60%). Due to their peripheral location and leptomeningeal involvement, they are one of the tumors that may exhibit a dural tail. This is reactive rather than due to true direct dural invasion, which is rare 2,6. As these lesions are very slow growing, superficial remodeling of the adjacent skull is characteristic and vasogenic edema is variable 5.
CT
Pleomorphic xanthoastrocytomas are typically hypodense or isodense and may be well or poorly demarcated, usually with little or no surrounding edema. Calcification is rare. Due to its superficial location it may cause scalloping of the overlying bone 2.
MRI
-
T1
solid component iso to hypointense to grey matter
cystic component hypointense
leptomeningeal involvement is seen in over 70% of cases 2
-
T1 C+ (Gd)
solid component usually enhances vividly
-
T2
solid component iso to hyperintense to grey matter
cystic component hyperintense
on T2/FLAIR sequence, cystic areas show hyperintensity relative to CSF due to higher protein contents
little surrounding vasogenic edema
Angiography (DSA)
Despite vivid enhancement, pleomorphic xanthoastrocytomas are usually avascular on angiography 2.
Treatment and prognosis
Although prognosis is good following surgical excision of grade 2 tumors, with a 5-year survival of 90% and 5-year-disease-free survival of 70% 1,3,6, local recurrence and malignant transformation (to WHO grade 3) is common, encountered in up to 20% of cases 2.
Grade 3 pleomorphic xanthoastrocytomas have a substantially worse prognosis: 40-50% 5-year survival 6.
Neither radiotherapy nor chemotherapy has a significant effect on these tumors 2, although radiotherapy may have a role to play in patients with incomplete resection or those with recurrent disease 3.
Differential diagnosis
The main differential diagnosis is that of other cortical tumors, with helpful distinguishing features including 1-4:
-
can look very similar
contrast enhancement often less prominent
calcification in ~50% of cases
-
dysembryoplastic neuroepithelial tumors (DNET)
contrast enhancement uncommon
"bubbly appearance" common
-
calcifications common
older age group: middle-aged adults, most commonly in the 4th and 5th decades of life
-
desmoplastic infantile ganglioglioma
young children
dural involvement prominent
large often multiple lesions






 Unable to process the form. Check for errors and try again.
Unable to process the form. Check for errors and try again.