Testicular seminomas are a type of germ cell tumor and the most common testicular tumor, accounting for ~45% of all primary testicular tumors. This article concerns only testicular seminomas; however, seminomas can arise outside of the testis, most often within the anterior mediastinum (see article on anterior mediastinal germ cell tumors).
On this page:
Epidemiology
Testicular germ cell tumors account for around 1-3% of all male malignancies in Western populations and are the most common non-hematological malignancy in males 15-49 years old. Approximately 50% of all germ cell tumors are seminomas. Several European countries (e.g. Denmark, Norway, Germany, Switzerland, and the Czech Republic) have the highest incidence rates for testicular cancer, and the incidence is on the rise, especially in young men 9.
Risk factors
-
undescended testis is the major risk factor for testicular germ cell tumors
10-40 times increased risk
~10% of tumors are associated with undescended testis
increased risk in the contralateral normally descended testis
a previous tumor in the contralateral testis
a family history of testicular germ cell tumor
testicular microlithiasis: controversial risk factor, although some sources claim approximately eight times increased risk 2
other risk factors include infections such as HIV, mumps, and orchitis and a history of trauma or organ transplant immunosuppression
See the article on risk factors for testicular germ cell tumor (GCT) for more details.
Clinical presentation
The most common presentation is with a painless testicular mass, although some 45% will report a degree of testicular discomfort. Bilateral tumors are rare (~2%) and are almost always asynchronous 1. Diagnosis following trauma is common as it draws the patient’s attention to the lump. Back pain, abdominal discomfort, or abdominal mass may present in 25% of patients with retroperitoneal nodal metastases 3. Presentation with distant or extranodal metastases is rare (~5%).
Pathology
Seminoma, by definition, must be pure seminoma on histology and not associated with an elevated serum alpha-fetoprotein (AFP). If either of these criteria is not met, the tumor must be classified as non-seminomatous and managed accordingly. Uncommonly (15%), b-HCG may be slightly elevated. Placental alkaline phosphatase (PALP) is a very specific tumor marker for seminoma, both as a serum marker and immunohistochemistry marker 10. Pure seminomas are subdivided into three histological subtypes:
classic: 85%, infrequent mitoses; monotonous sheet of large cells with abundant cytoplasm and round hyperchromatic nuclei, prominent nucleoli
anaplastic: 10%, ≥3 mitotic figures per high-power field
spermatocytic: 5%, older male patients above 60 years old, rarely metastasize; well-differentiated with cells resembling secondary spermatids
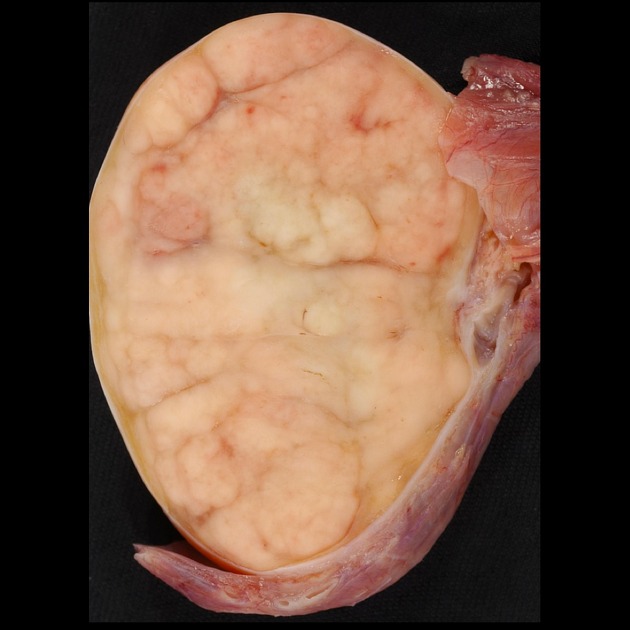
On gross examination, seminomas are pale grey to yellow nodules that are uniform or slightly lobulated and often bulge from the cut surface. Tumor staging is via the TNM staging system (see testicular cancer staging).
Radiographic features
Ultrasound
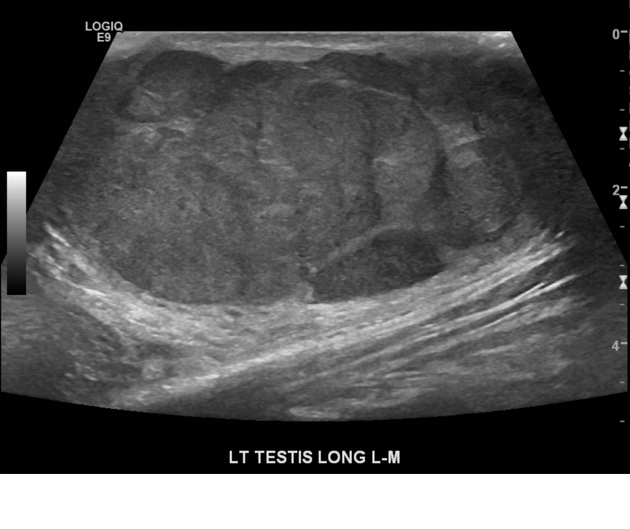
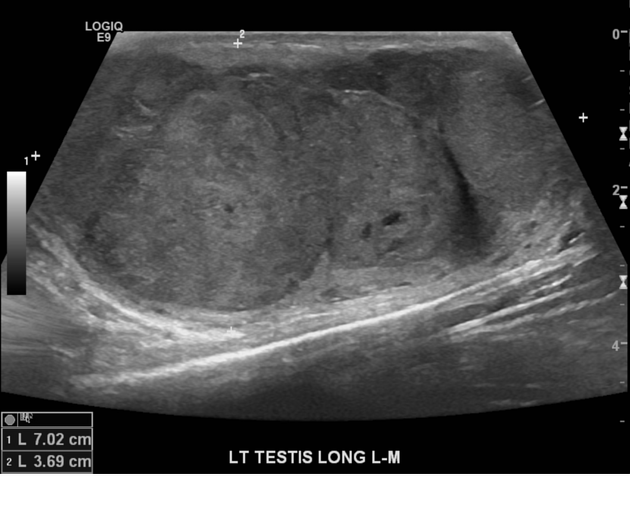
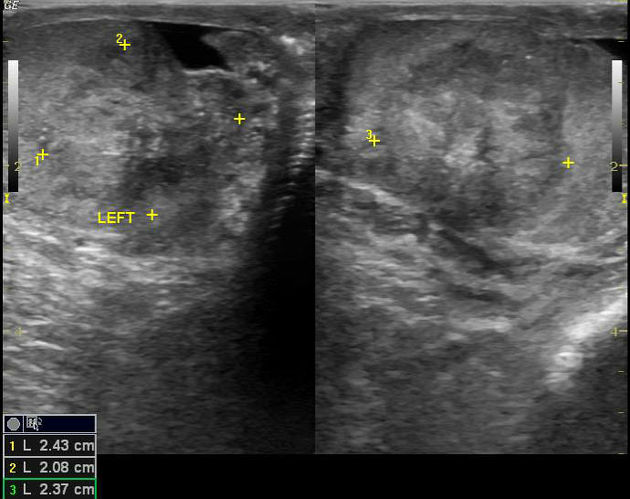
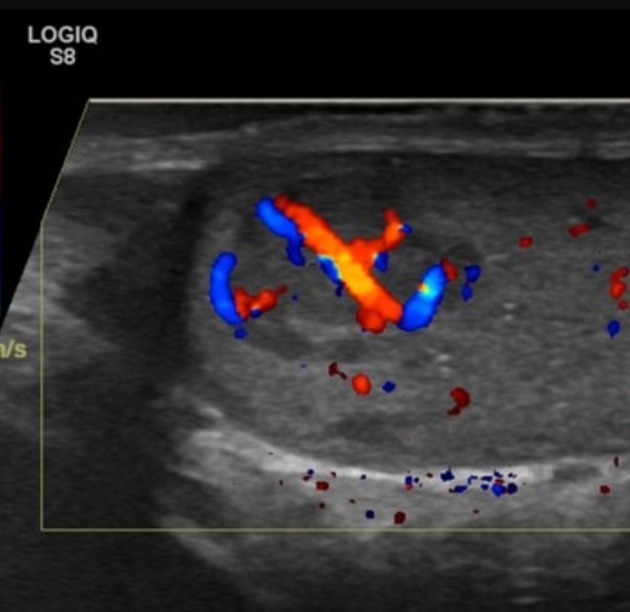
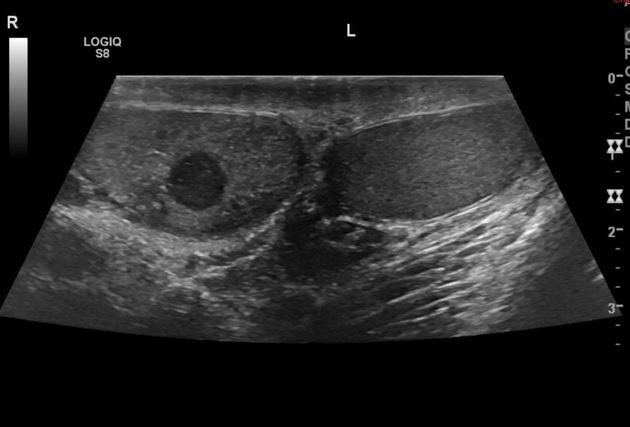
Ultrasound is the first-line imaging modality if a patient presents with a testicular abnormality. Ultrasound has a 92–98% sensitivity and specificity of 95–99.8% for testicular malignancy. Its sensitivity is nearly 100% when combined with the physical examination. Features include:
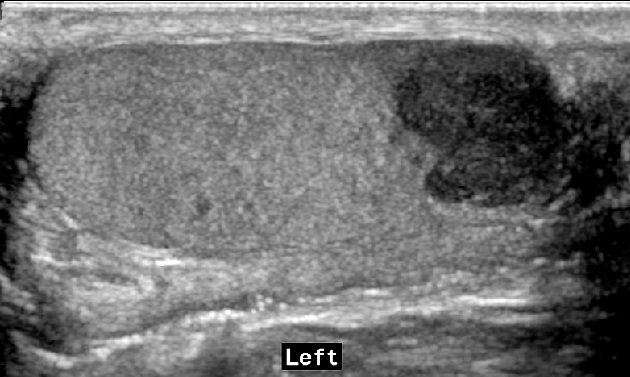
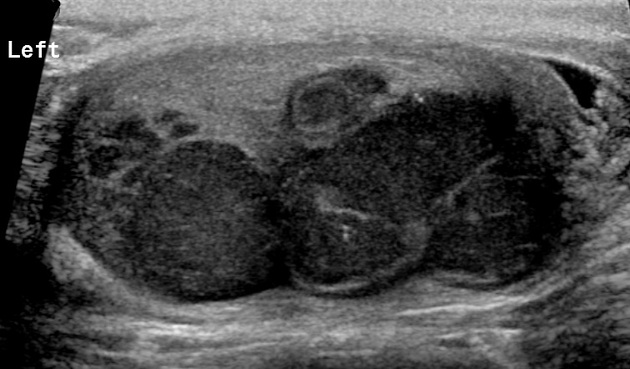
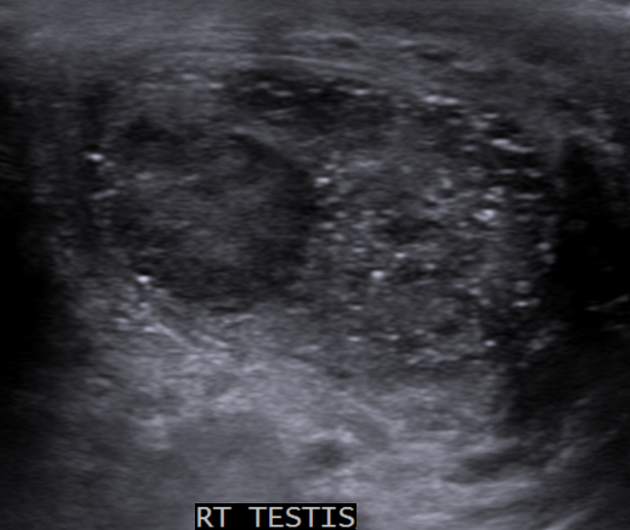
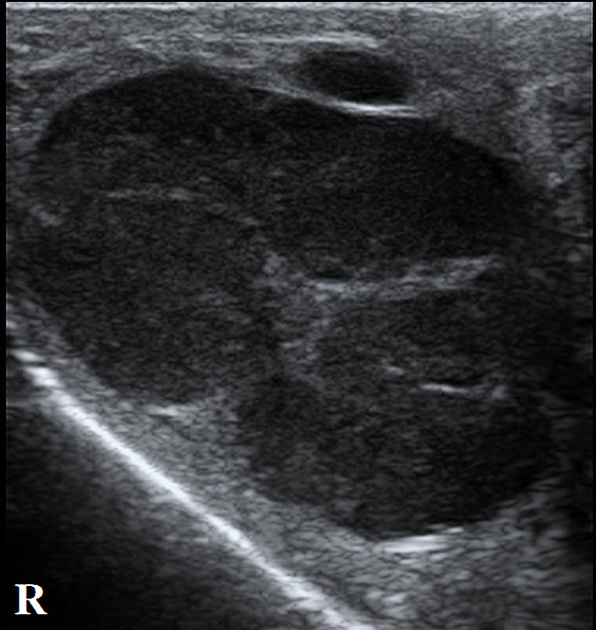
seminomas usually appear as a homogeneous intratesticular mass of low echogenicity compared to normal testicular tissue
-
the mass is usually oval and well-defined in the absence of local invasion
usually confined within the tunica albuginea, rarely extending to para-testicular structures
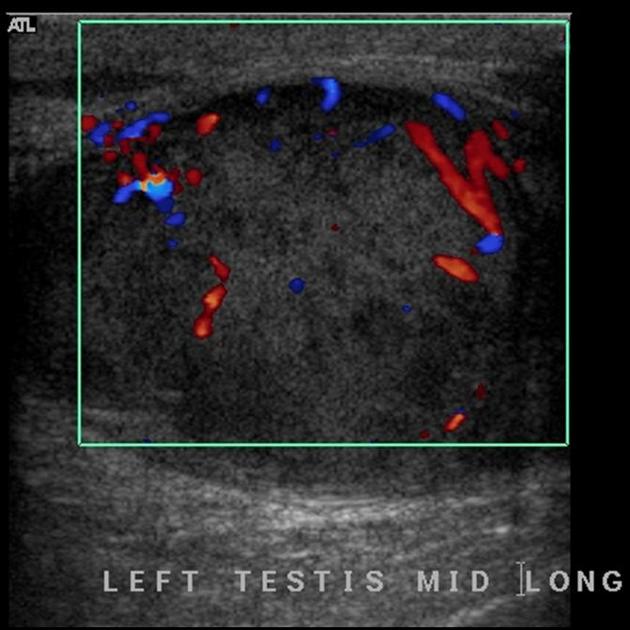
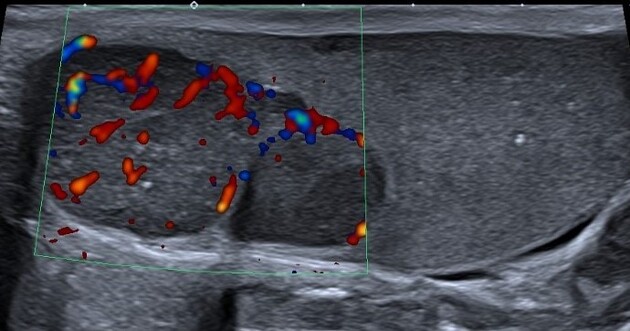
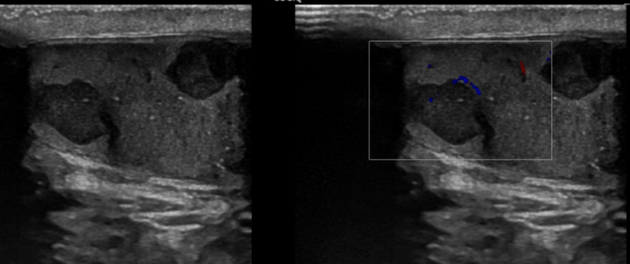
internal blood flow is seen on color Doppler imaging
cystic regions and calcifications are less common than in non-seminomatous germ cell tumors
larger seminomas can have a heterogeneous appearance
CT
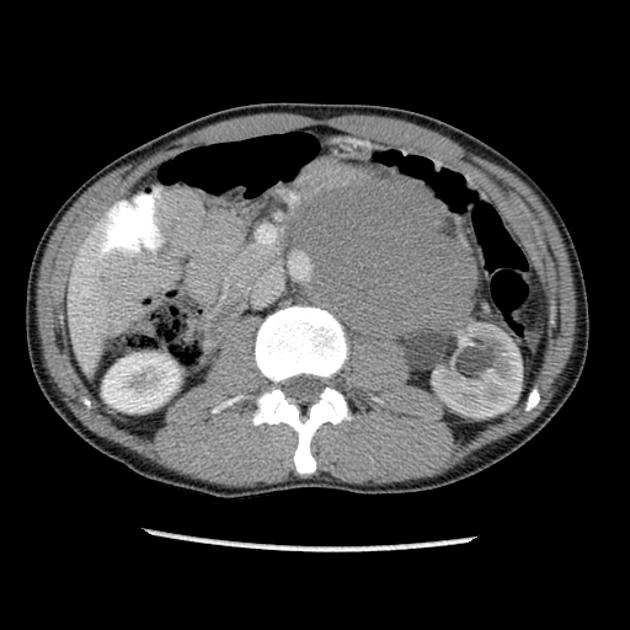
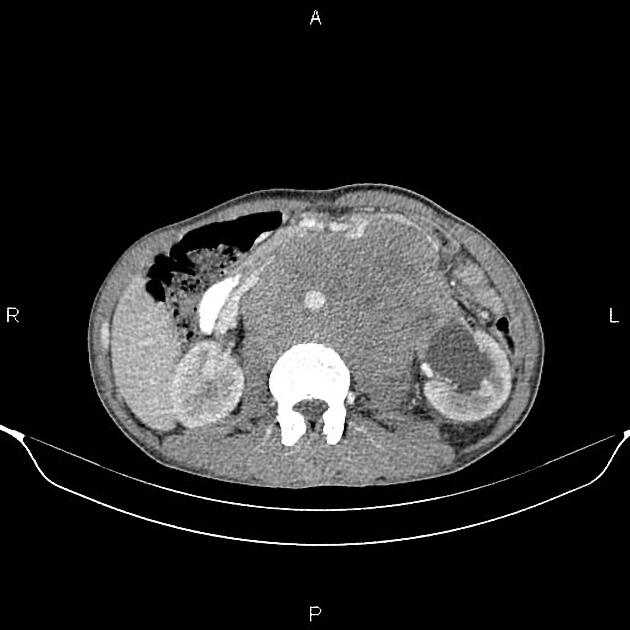
Abdominal and pelvic CT is important in visualizing metastases, both as a part of primary staging seminoma and in primary diagnosis when a testicular mass is unknown.
Metastases to the para-aortic lymph nodes at the level of the renal vessels are the typical first site of spread, owing to the lymphatic drainage of the testes relating to embryological testicular descent. The nodal metastases are often bulky and of homogeneous density and tend to encase surrounding vessels.
Inguinal or iliac lymph node metastases suggest lymphatic spread via the scrotum and, therefore, local tumor extension beyond the tunica vaginalis.
Visceral metastases are seen in ~5% of patients at presentation (lung, liver, bone, brain). Staging CT of the chest is only indicated when regional para-aortic lymph node spread is present or if there is an abnormal chest x-ray.
Following therapy, lymph node metastases reduce markedly in size, but some inactive abnormal tissue persists, which can be difficult to distinguish from residual disease, wherefore follow-up monitoring is required.
MRI
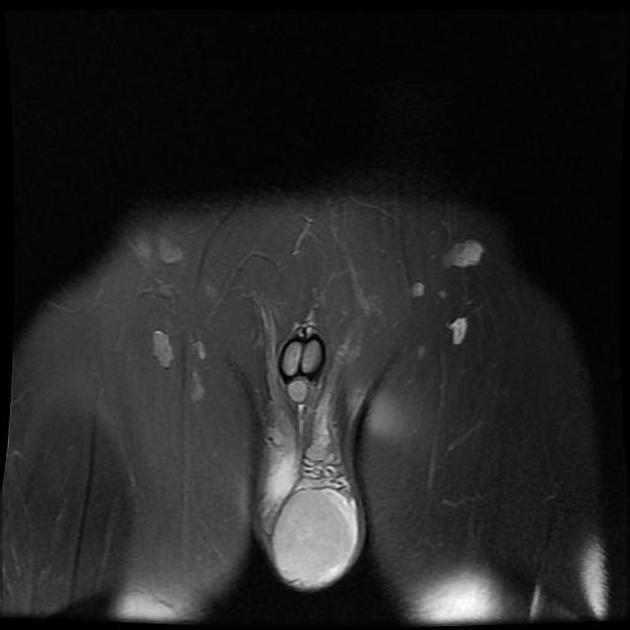
In some cases, ultrasound cannot confidently characterize the nature of a testicular lesion, and MRI may be a valuable alternative tool for problem-solving. MRI is highly accurate and superior to ultrasound in local staging of testicular tumors. MRI has a sensitivity of nearly 100%, specificity of 88%, PPV of 96.5%, NPV of 100%, and accuracy of 96.4% in differentiating benign tumors from malignant tumors.
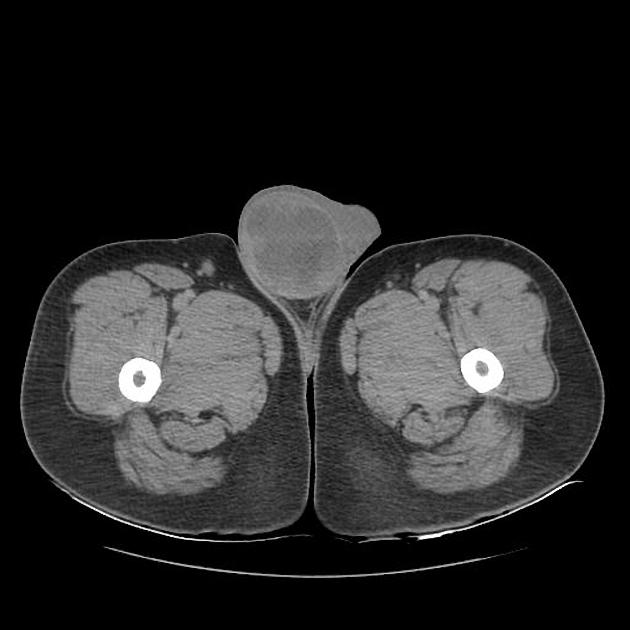
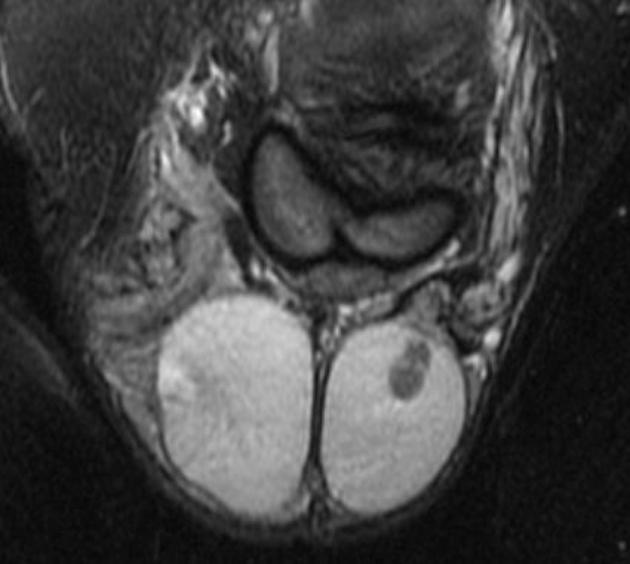
A preoperative diagnosis of a benign testicular lesion by MRI can reduce the number of diagnostic surgical scrotal explorations and can also help avoid unnecessary radical orchiectomies. MRI can also play a key role in the presurgical local staging of malignant testicular lesions in patients undergoing testis-sparing surgery (patients with bilateral testicular tumors or a solitary testis with a tumor). Usually, seminomas appear as multinodular tumors of uniform signal intensity 3,4:
T2: hypointense to normal testicular tissue
T1 C+ (Gd): heterogeneous enhancement
DWI/ADC: restricted diffusion is seen (feature of malignant tumors)
Nuclear medicine
FDG-PET is less sensitive in initial staging than CT. Usually only used following treatment of seminoma to look for metabolic activity within residual lymphadenopathy or extranodal metastases.
Treatment and prognosis
Treatment involves surgical removal of the testicular primary (orchiectomy).
Radiotherapy to regional nodes if there is local disease (stage I) or limited nodal para-aortic metastases (non-bulky stage II) may be necessary (seminomas are exquisitely radiosensitive, whereas non-seminomatous tumors are not radiosensitive).
Chemotherapy may be necessary if there is bulky para-aortic lymph node involvement or more distant disease. Four cycles of bleomycin, etoposide, and cisplatin (BEP) are the standard of care.
Prognosis is good for all stages with greater than 90% cure rate. Survivors have an increased lifetime risk of developing other malignancies, including testicular cancer in the remaining testis, mesothelioma, and cancer of the lung, colon, bladder, pancreas, and stomach.
Differential diagnosis
The main differential for testicular mass in young adults is non-seminomatous germ cell tumor (NGCT), which usually appears more heterogeneous, often with cysts and calcification. Lymphadenopathy of NGCT may also be more heterogeneous.
Testicular lymphoma is the main differential diagnosis to consider when para-aortic lymphadenopathy is the presenting finding or in the setting of bilateral testicular lesions.
see differential for unilateral testicular mass
see differential for bilateral testicular masses







 Unable to process the form. Check for errors and try again.
Unable to process the form. Check for errors and try again.