The central nervous system (CNS) is an uncommonly involved organ system in Langerhans cell histiocytosis (LCH). Involvement of the CNS is related but distinct from involvement of the skull or craniofacial structures, which are discussed separately in the article skeletal manifestations of LCH. For a general discussion of this disease please refer to the article on Langerhans cell histiocytosis (LCH).
On this page:
Epidemiology
The CNS is rarely involved in LCH, being present in only 6% of cases at the time of diagnosis 1. Patients with multisystem LCH or specific involvement of craniofacial structures have an increased risk of developing CNS involvement 1.
Clinical presentation
There are generally three distinct clinical presentations that may or may not all be present in any given patient:
neurogenic (central) diabetes insipidus 2,3
neurodegeneration characterized by psychomotor deterioration and ataxia 2,3
CNS involvement rarely occurs in isolation, thus, patients tend to present with other manifestations of LCH as well 2-4.
Pathology
There is proliferation of CD1a and CD207 Langerhans cells with an abundance of eosinophils, lymphocytes and neutrophils, typically affecting the hypothalamic-pituitary axis, cerebellum, and basal ganglia, accounting for the clinical presentation and radiographic features 2,4.
Radiographic features
MRI
MRI is the most useful imaging modality for evaluating LCH CNS involvement 2-6. Similar to the clinical presentations, three groups of neuroimaging features may be appreciated, which again may or may not all be present in any given patient 2-6.
Diabetes insipidus
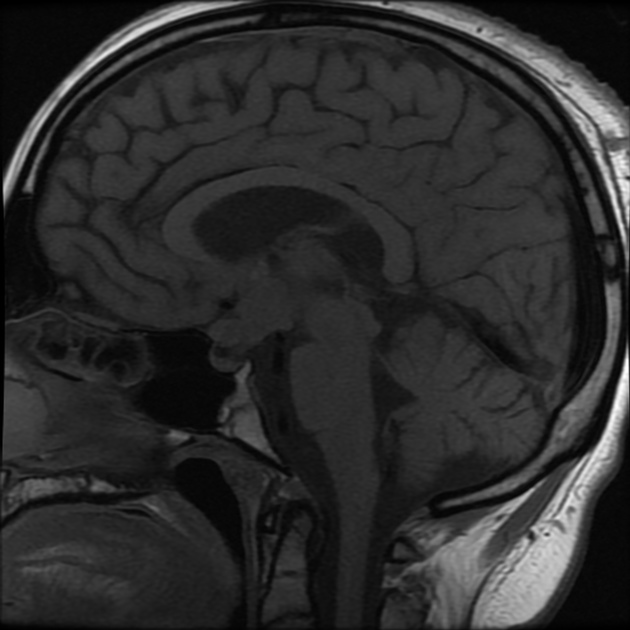
This is the most common CNS manifestation of LCH 2-6. Patients with diabetes insipidus in LCH demonstrate a lack of T1-weighted high intensity of the posterior pituitary with associated enhancement and thickening (>3 mm) of the infundibulum 2-6. This loss of the normal posterior pituitary bright spot is thought to be a result of the loss of vasopressin stores and is characteristic of CNS involvement in LCH, but is also non-specific 3.
Neurodegenerative changes
These are the second most common CNS manifestation of LCH 2-6. Patients with neurodegeneration demonstrate bilateral symmetric parenchymal lesions of the cerebellum (especially the dentate nuclei) and basal ganglia, however uncommonly other regions of the brain and brainstem (especially the pons) may be affected and cerebral atrophy may also be noted 2-6. These affected regions often initially demonstrate T1-weighted high intensity with variable T2-weighted intensity, however progress to marked T2-weighted high intensity 2-6. Contrast enhancement can also be variable throughout the temporal progression of the disease 2-6.
Mass lesions
Mass lesions are the least common CNS manifestation of LCH 2,3. Masses have a variable MR appearance and can arise from the meninges (especially the dura mater), pineal gland, choroid plexus, hypothalamus, ependyma, or can rarely be intraparenchymal 2,3. These lesions, depending on size and location, may cause obstructive hydrocephalus 2,3.
Treatment and prognosis
Treatment is complex, but often involves prednisolone and chemotherapy 4. However this treatment is not effective in improving the diabetes insipidus or neurodegenerative symptoms 1,7,8. Hence, many patients additionally require desmopressin for diabetes insipidus 1. Treatment of neurodegeneration in LCH may include immunochemotherapy (e.g. BRAF V600E inhibitor therapy) 7-9.





 Unable to process the form. Check for errors and try again.
Unable to process the form. Check for errors and try again.