Renal angiomyolipomas (AML) are a type of benign renal neoplasm encountered both sporadically and as part of a phakomatosis, most commonly tuberous sclerosis. They are considered one of a number of tumors with perivascular epithelioid cellular differentiation (PEComas) and are composed of vascular, smooth muscle and fat elements. They can spontaneously hemorrhage, which may be fatal. Angiomyolipomas usually have characteristic radiographic appearances.
On this page:
Epidemiology
Angiomyolipomas are the most common benign solid renal lesion and also the most common fat-containing lesions of the kidneys. The majority of angiomyolipomas are sporadic (80%) and are typically identified in adults (mean age of presentation 43 years), with a female predilection (F:M of 2-4:1) 7,9.
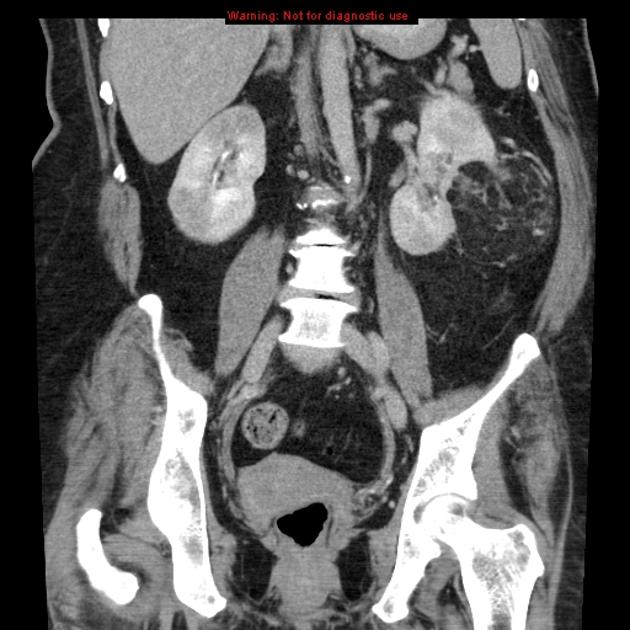
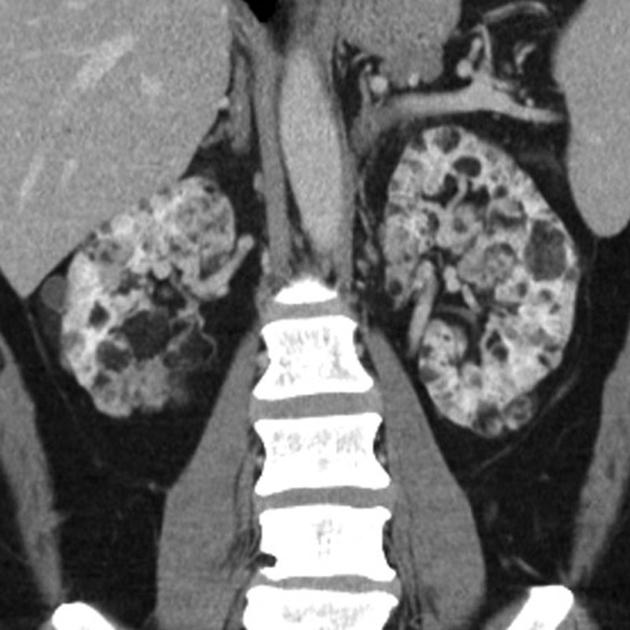
The remaining 20% are associated with phakomatoses, the vast majority in the setting of tuberous sclerosis. Angiomyolipomas are found in 25-50% of tuberous sclerosis patients 23. They have also been described in the setting of von Hippel-Lindau syndrome (vHL) and neurofibromatosis type 1 (NF1) 5,7. In these cases, they present earlier (usually identified by the age of 10 years), are larger, and are far more numerous. They are more likely to be fat-poor, which accounts for their earlier presentation 2,6,7. They have also been associated with lymphangioleiomyomatosis (LAM) 20.
Clinical presentation
Angiomyolipomas are often found incidentally when the kidneys are imaged for other reasons, or as part of screening in patients with tuberous sclerosis.
Symptomatic presentation is most frequently with spontaneous retroperitoneal hemorrhage due to microaneurysm rupture within the vascular component, and the risk of bleeding is proportional to the size of the lesion (>4 cm diameter). Shock due to severe hemorrhage from rupture is described as Wunderlich syndrome 4,5,7.
Patients may present with numerous other symptoms and signs 2, e.g. palpable mass, flank pain, urinary tract infections, hematuria, renal failure, or hypertension 3.
Pathology
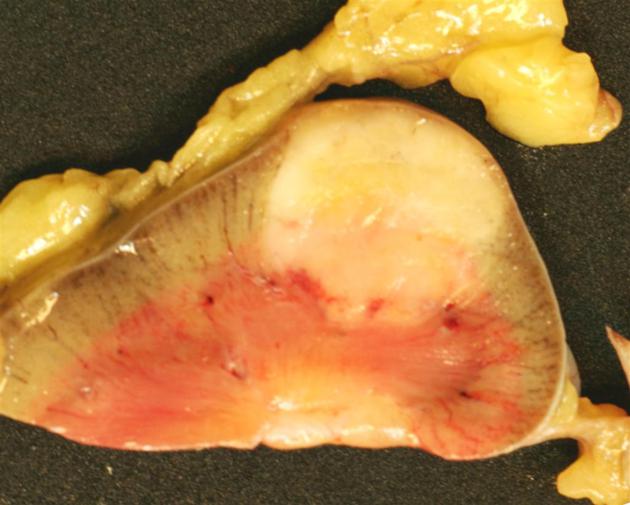
Angiomyolipomas are members of the perivascular epithelioid cells tumor group (PEComas) and are composed of variable amounts of three components; blood vessels (-angio) lacking elastic tissue, plump spindle cells (-myo), and adipose tissue (-lipo).
Almost all classic angiomyolipomas are benign, but they do have the risk of rupture with bleeding or secondary damage/destruction of surrounding structures as they grow.
Variants
Two histological types have been described
typical (triphasic)
atypical (monophasic or epithelioid)
A special variant called an epithelioid angiomyolipoma is composed of more plump, epithelial-looking cells, often with nuclear atypia, that have a described risk of malignant behavior. This variant, unlike conventional AMLs, may mimic renal cell carcinoma 10. Metastases have also been described 9.
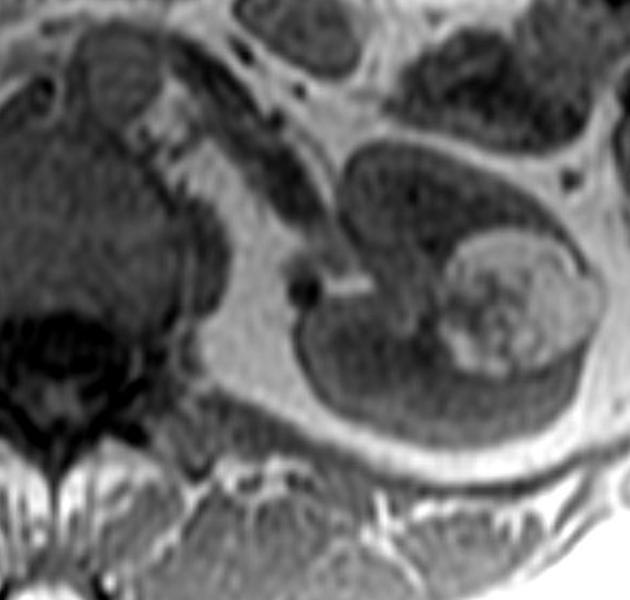
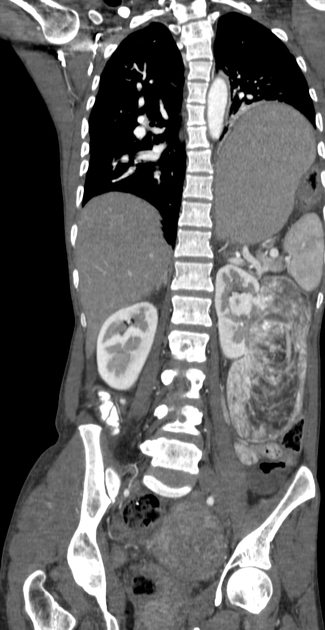
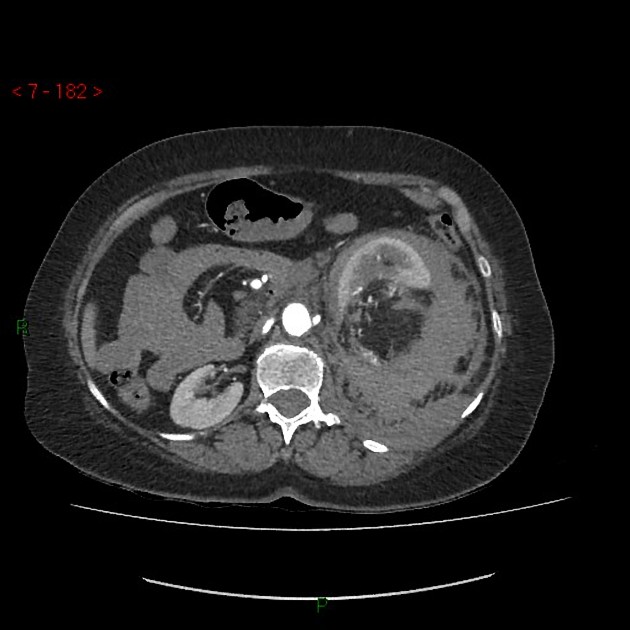
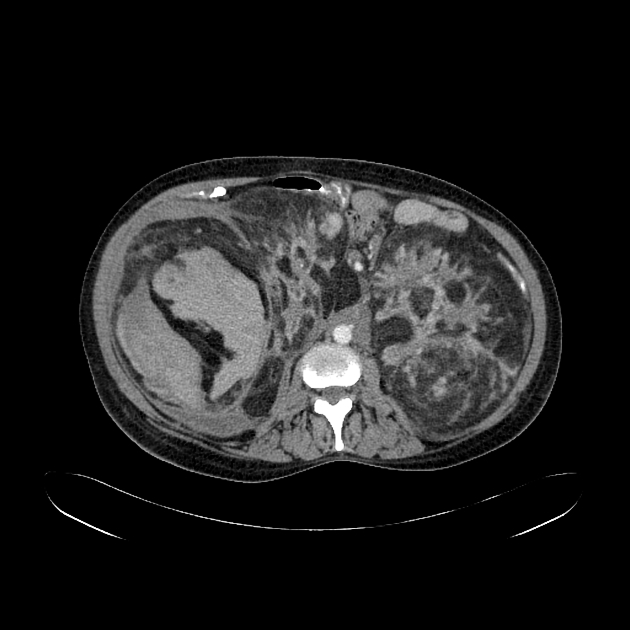
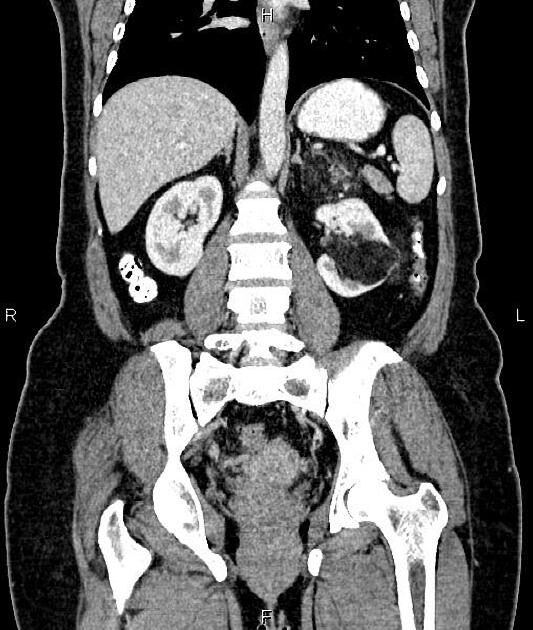
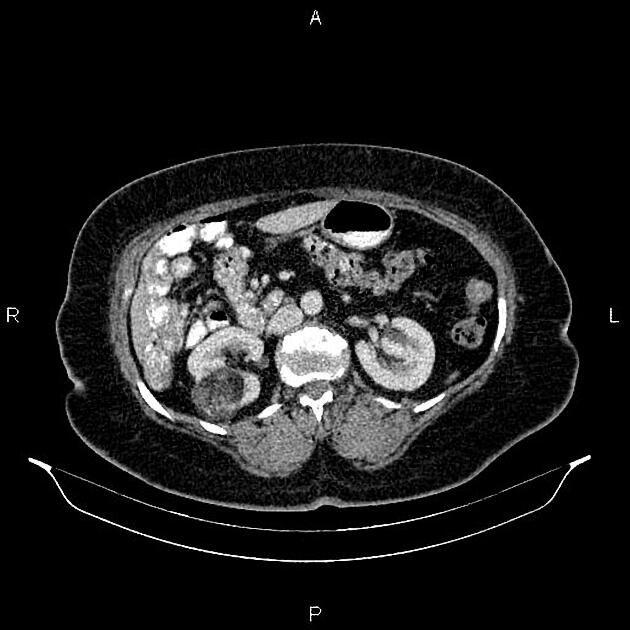
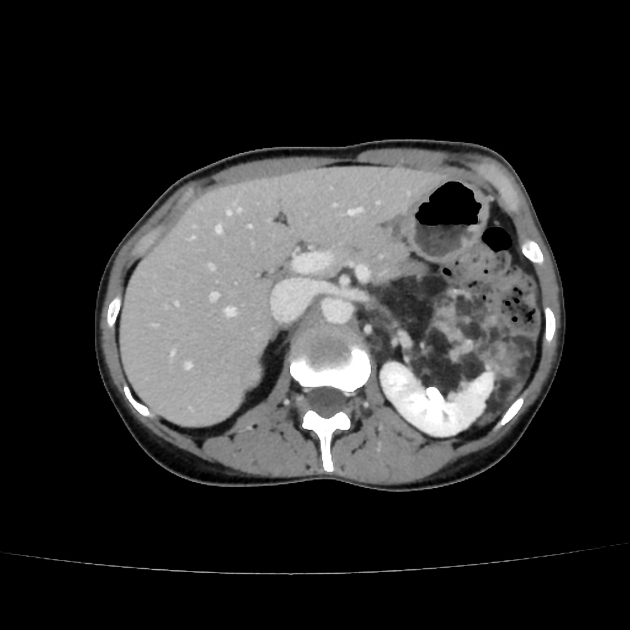
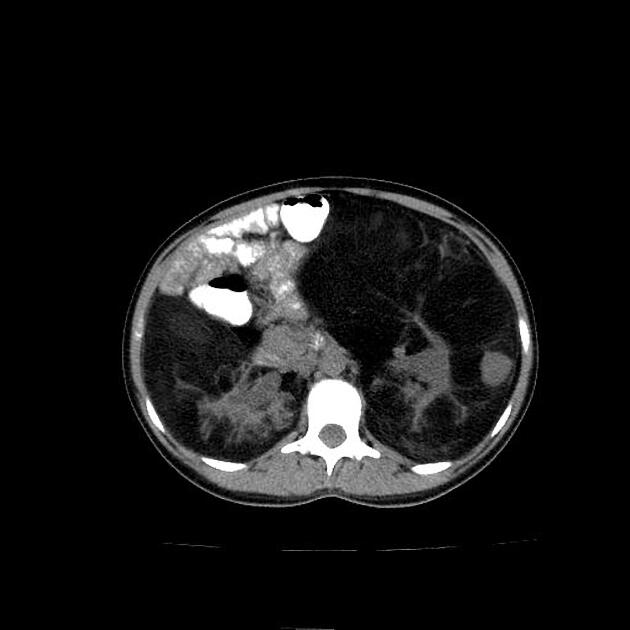
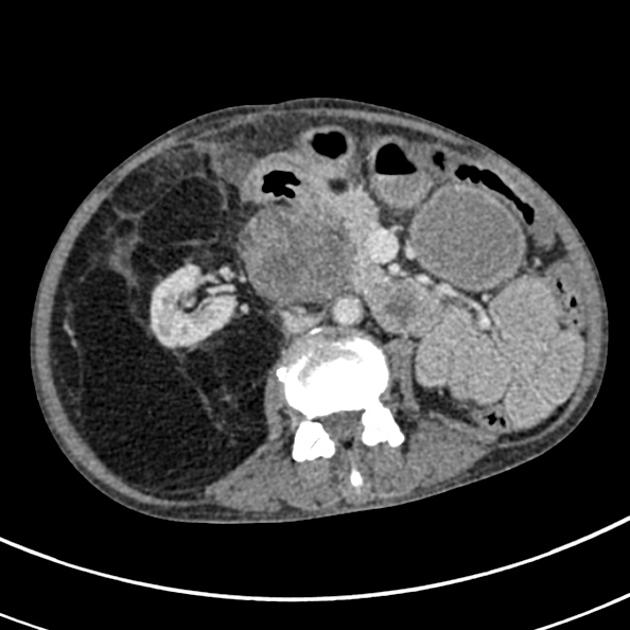
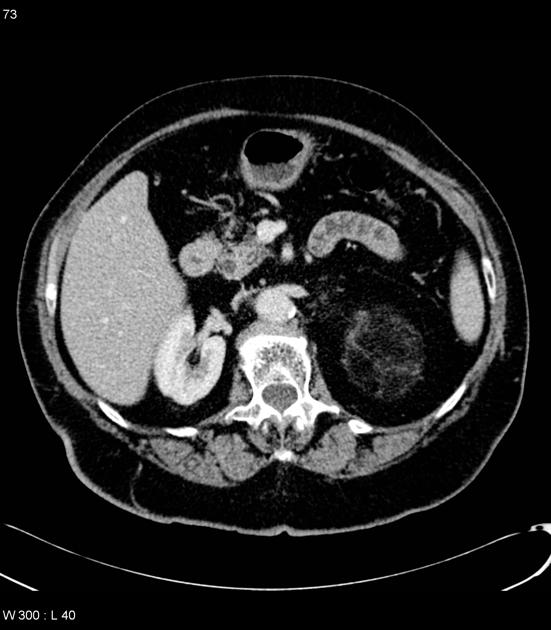
Radiographic features
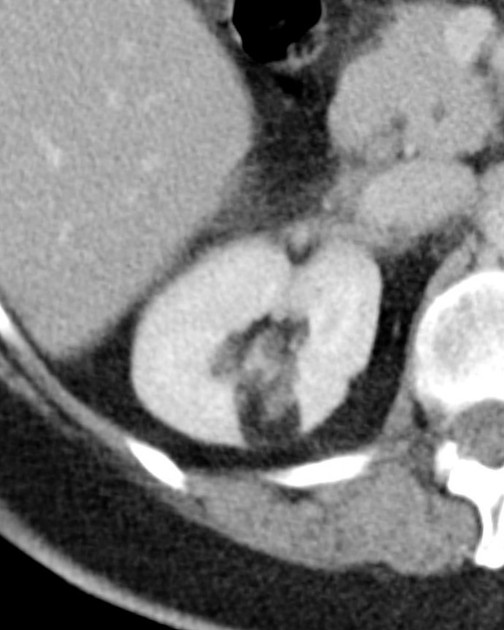
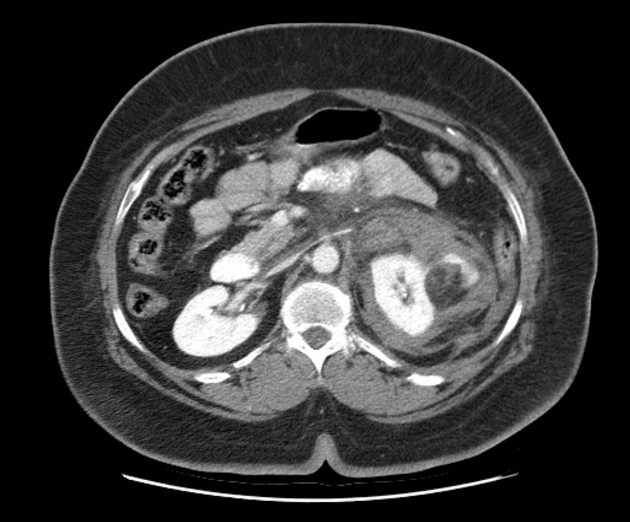
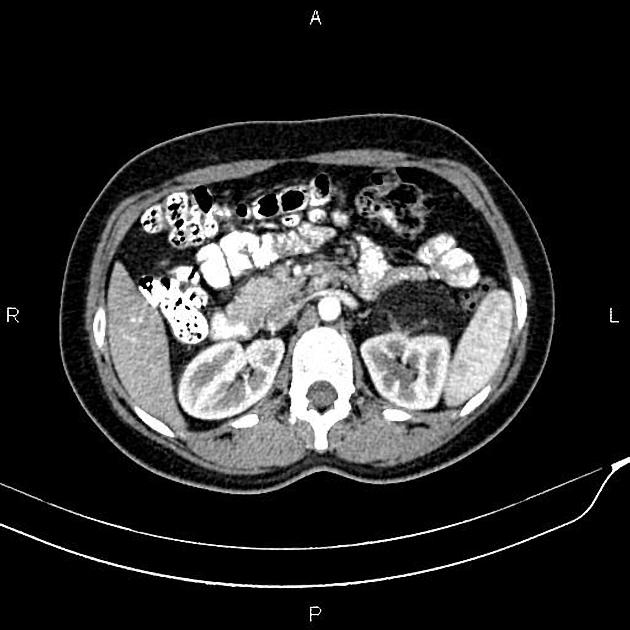
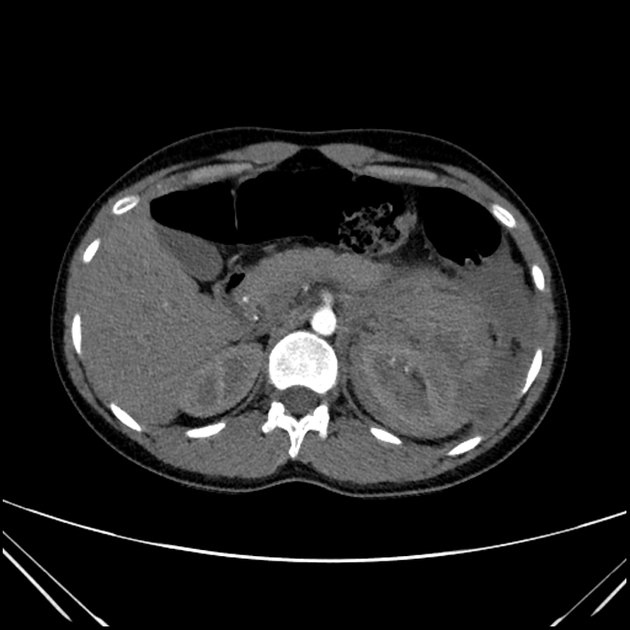
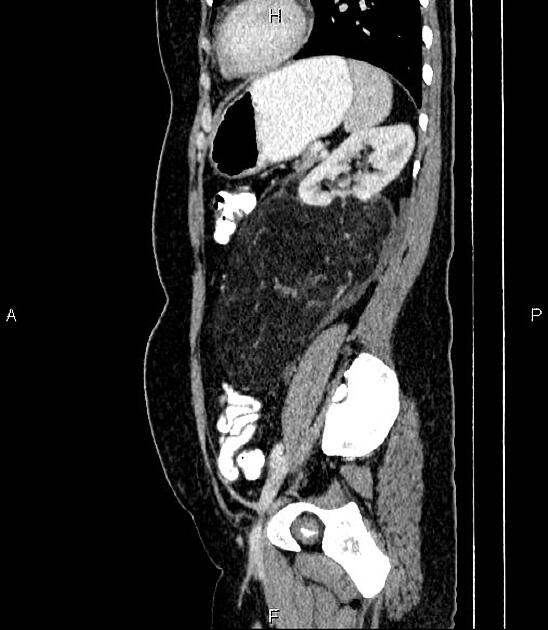
The cornerstone of diagnosis in all modalities is the demonstration of macroscopic fat; however, in the setting of hemorrhage, or when lesions contain little fat, it may be difficult to distinguish an angiomyolipoma from a renal cell carcinoma.
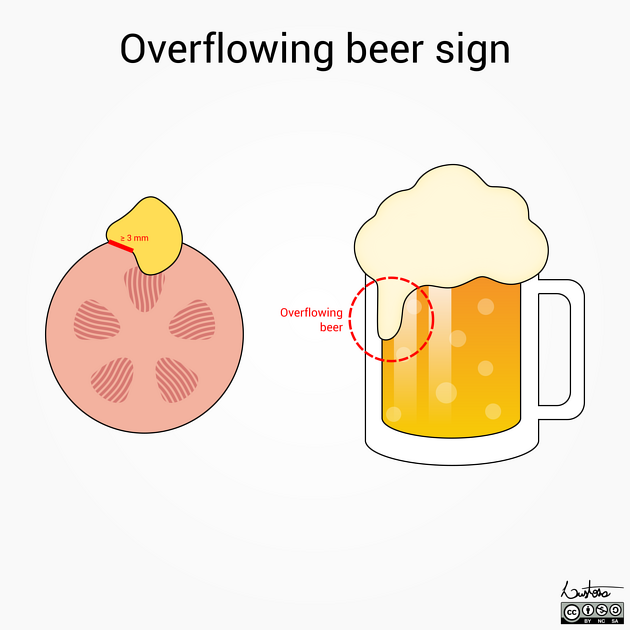
The overflowing beer sign has been described as a useful indicator of AML and the angular interface sign with a pointed projection into the renal substance has been described as an indicator of a benign lesion. These features are also applicable to MRI.
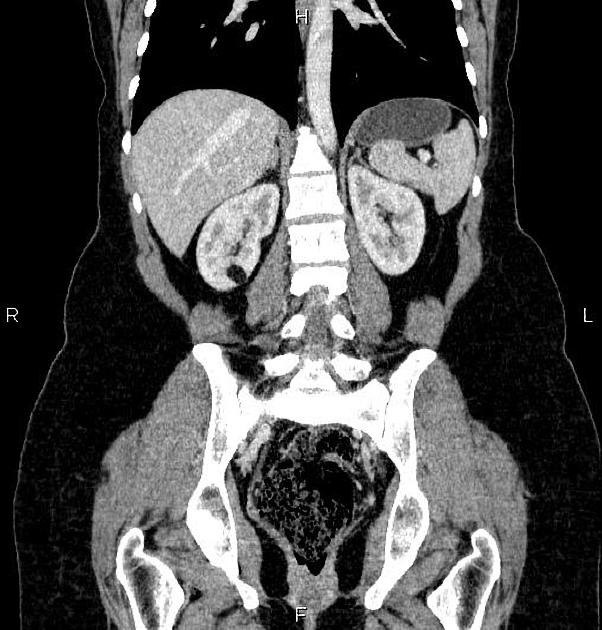
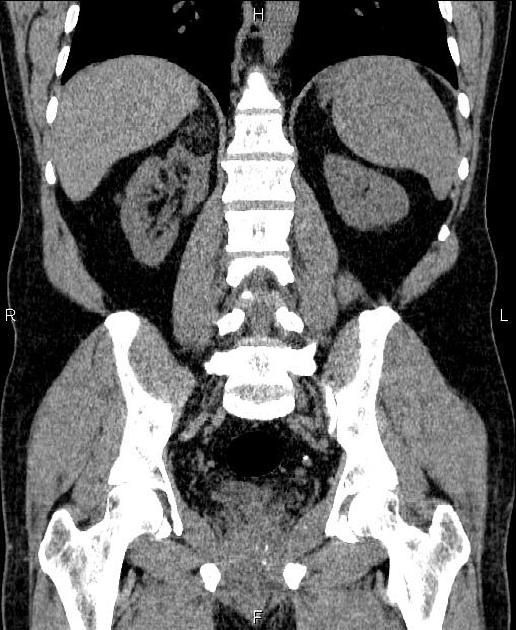
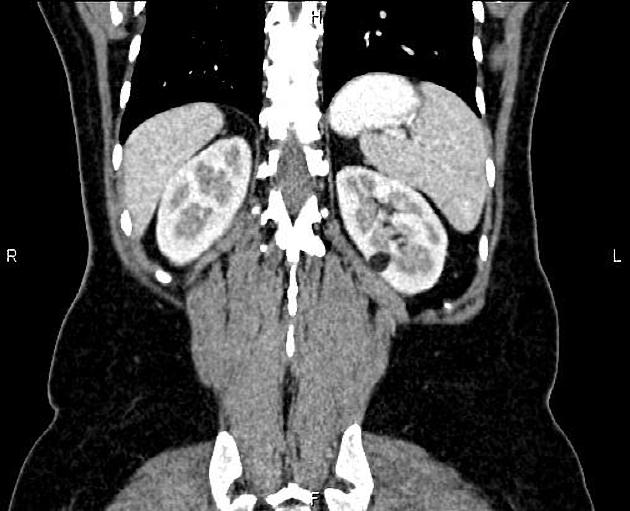
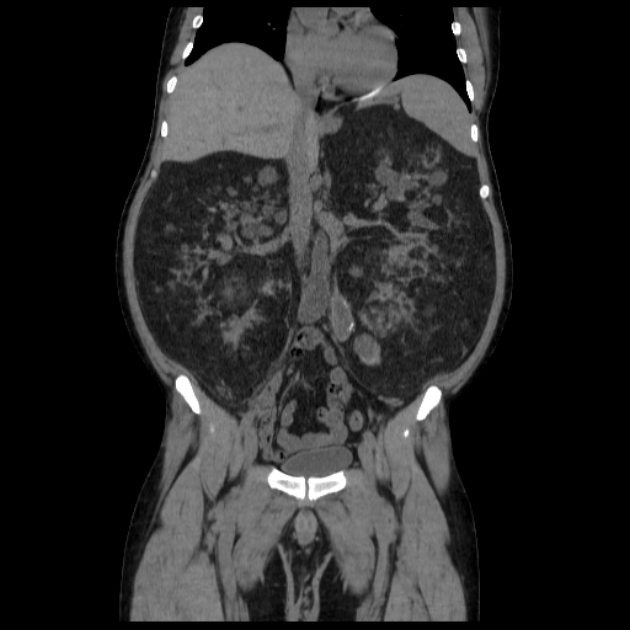
Tuberous sclerosis lesions are often multiple and bilateral and may be very large.
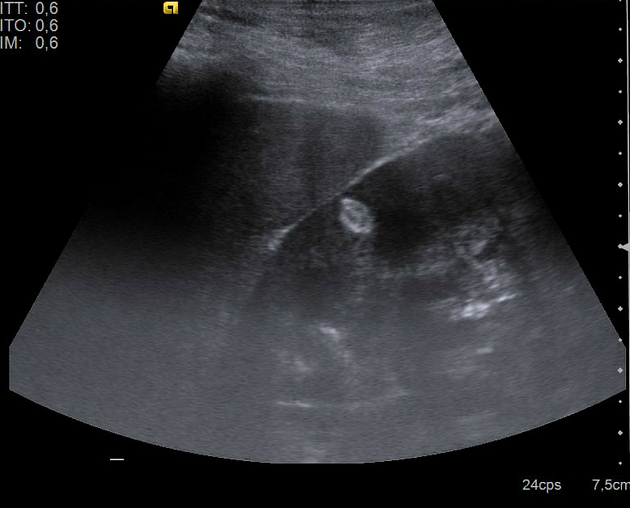
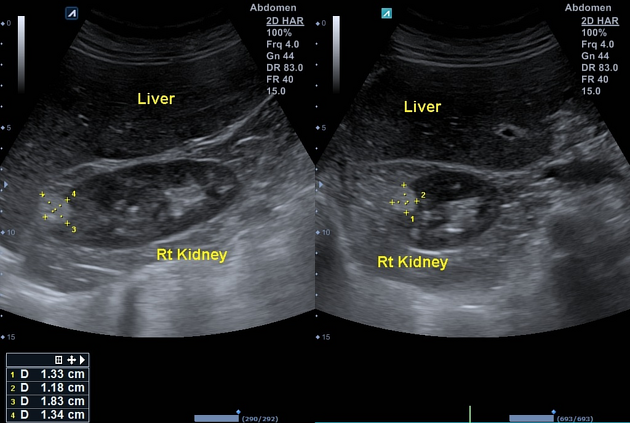
Ultrasound
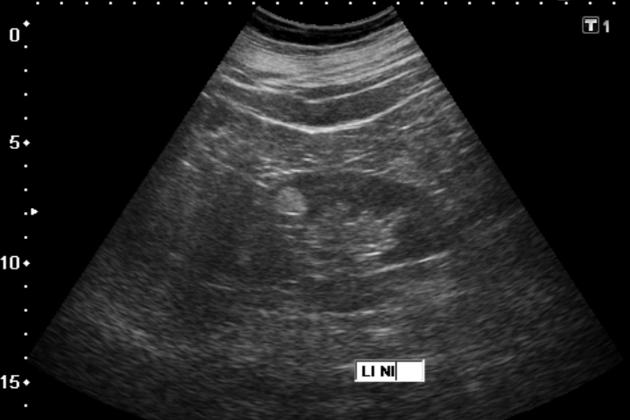
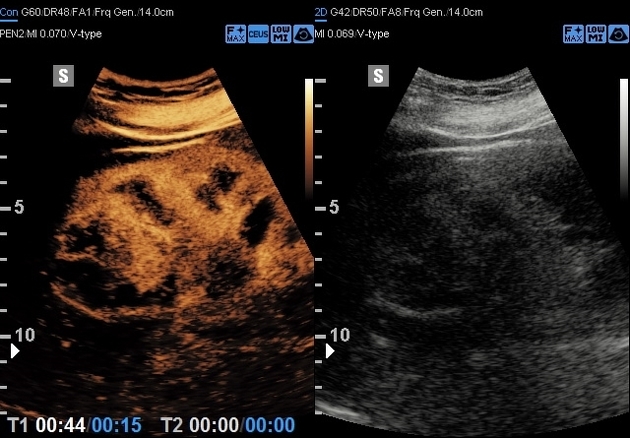
tend to appear as hyperechoic lesions on ultrasound, located in the cortex and with beam attenuation posteriorly (in the same manner as the beam is attenuated by a fatty liver)
in the setting of tuberous sclerosis, they may be so numerous that the entire kidney is affected, appearing echogenic with the loss of normal corticomedullary differentiation 7
-
contrast-enhanced ultrasound 12
tend to enhance peripherally
decreased central enhancement, compared with normal cortex
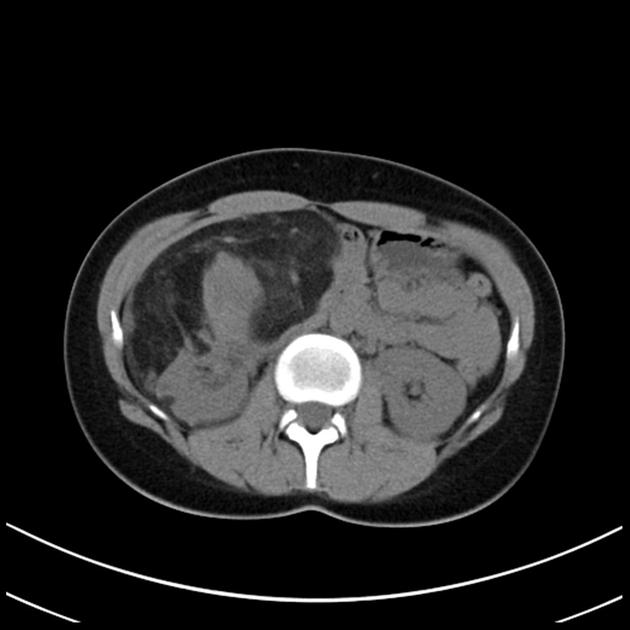
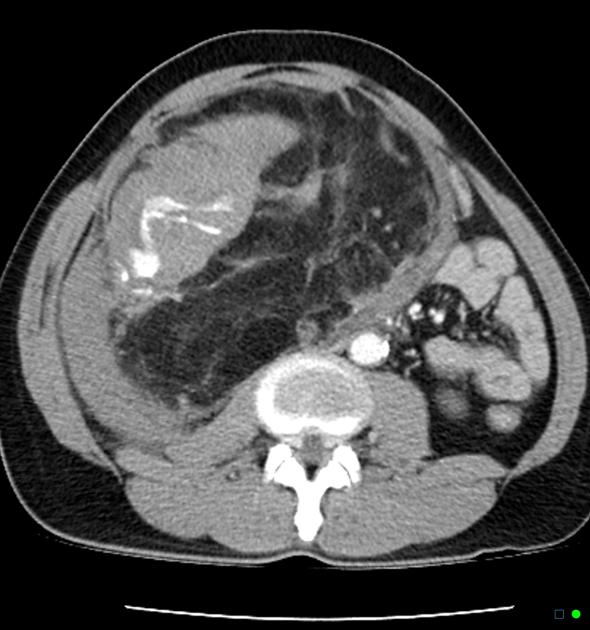
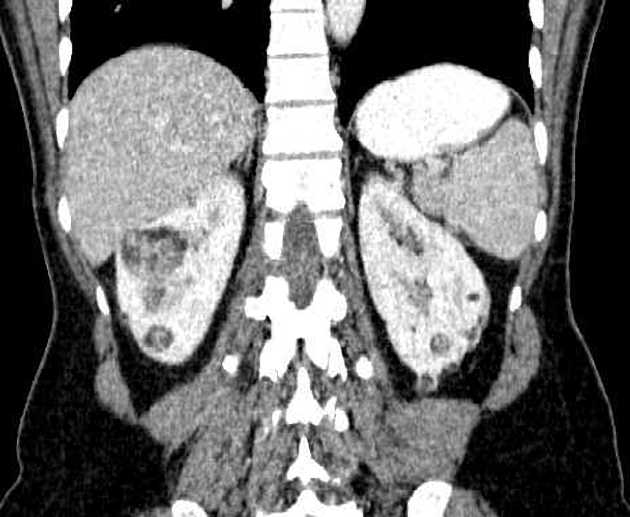
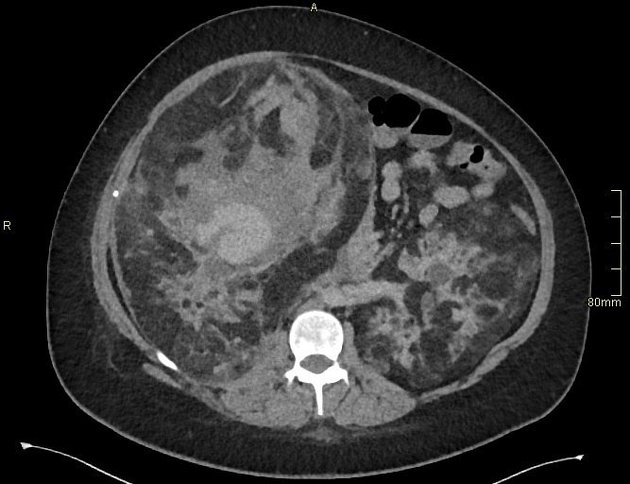
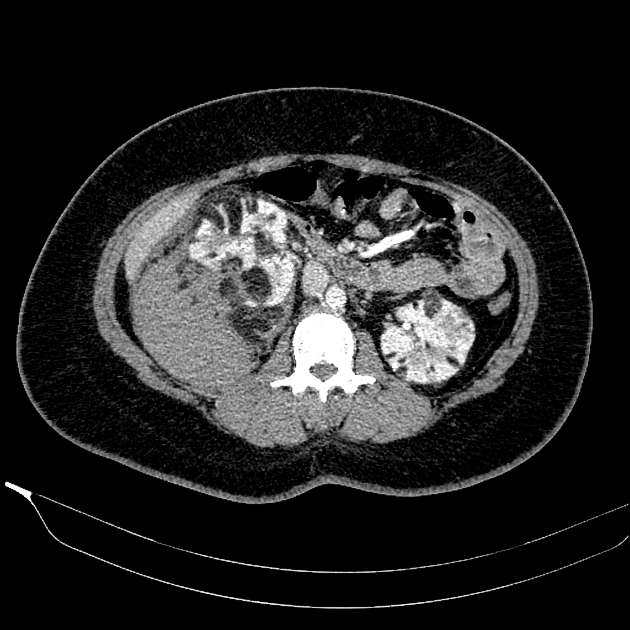
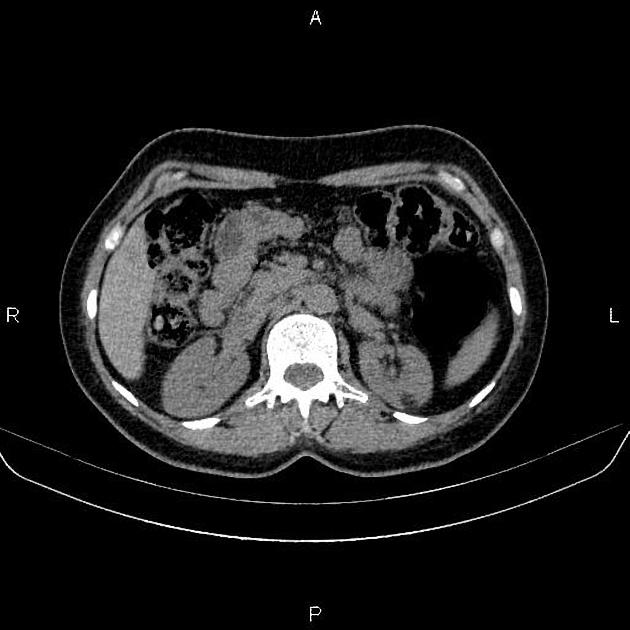
CT
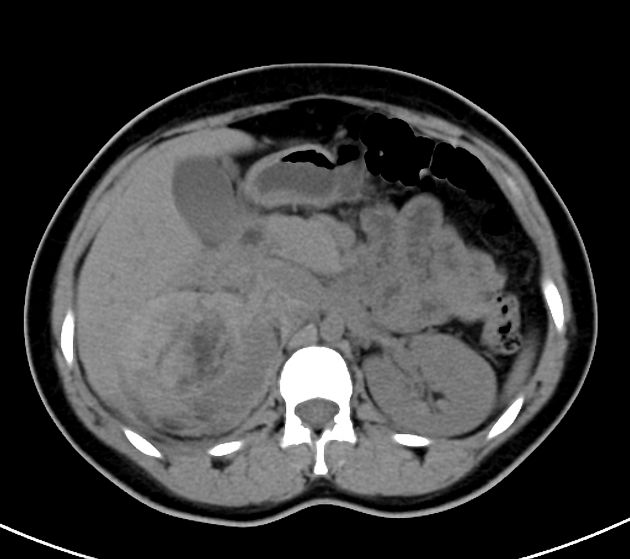
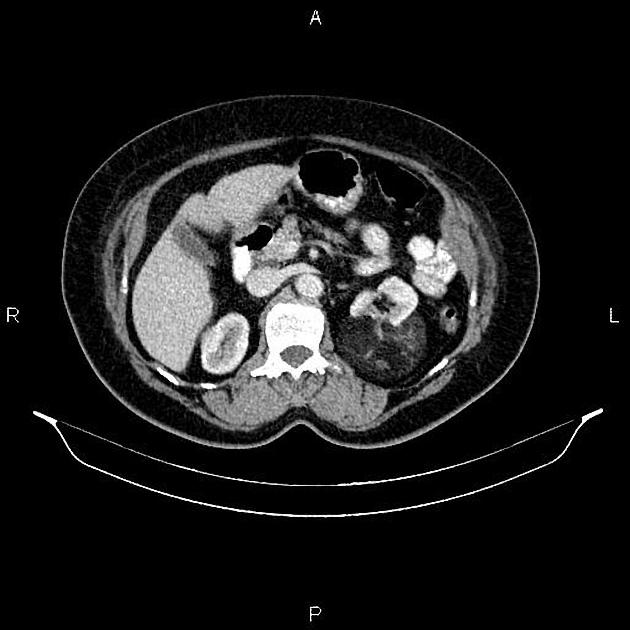
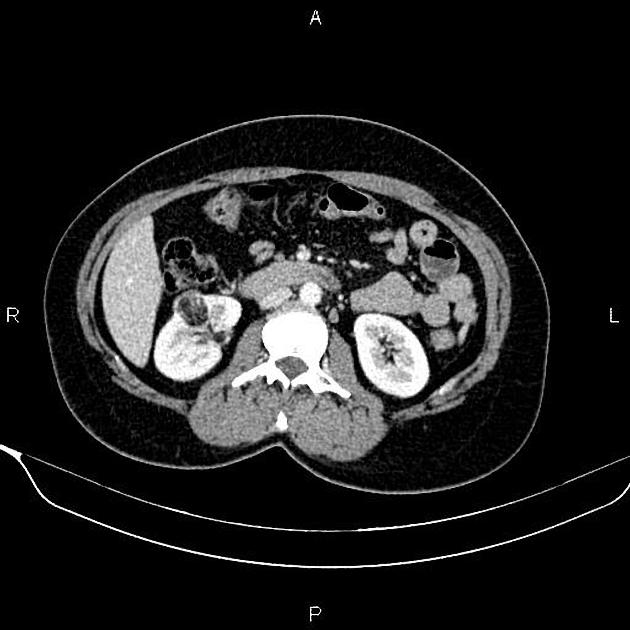
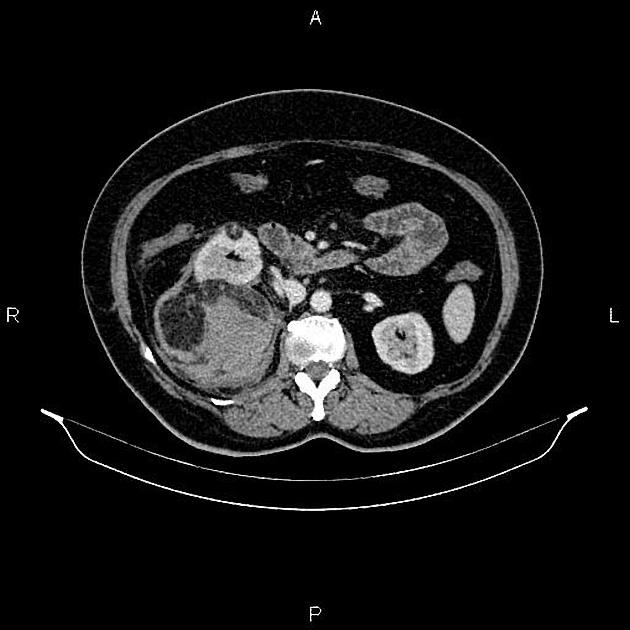
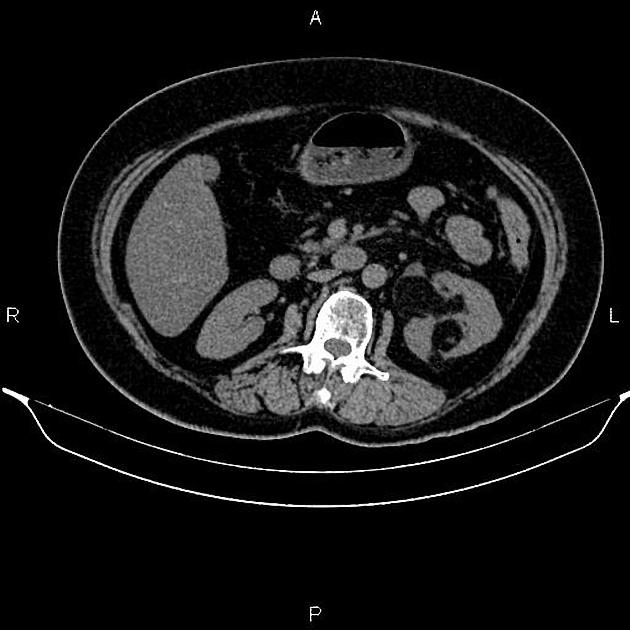
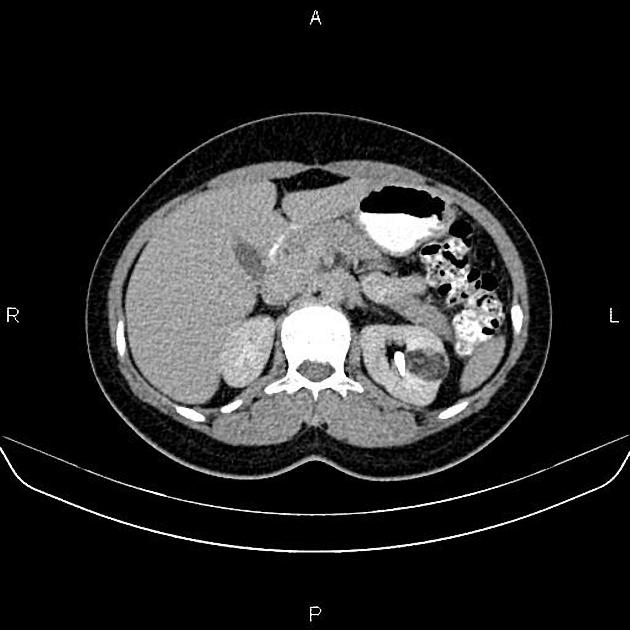
Most lesions involve the cortex and demonstrate macroscopic fat (less than -20 HU). When small, volume averaging may make differentiation from a small cyst difficult. It is essential to remember that rarely renal cell carcinomas (RCC) may have macroscopic fat components, and as such, the presence of fat is strongly indicative of an angiomyolipoma but not pathognomonic.
It is important to realize that ~5% of angiomyolipomas are fat-poor 15. This is especially the case in the setting of tuberous sclerosis, where up to a third do not demonstrate macroscopic fat on CT 6. Absence of ossification/calcification on imaging is in favor of AML 22.
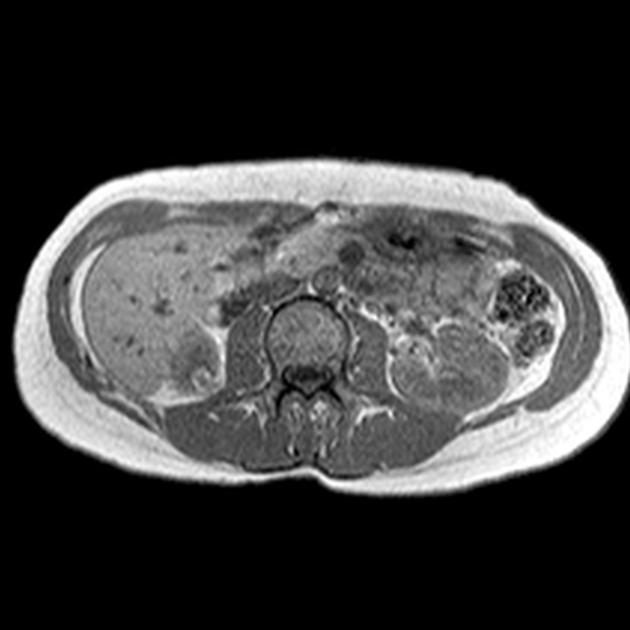
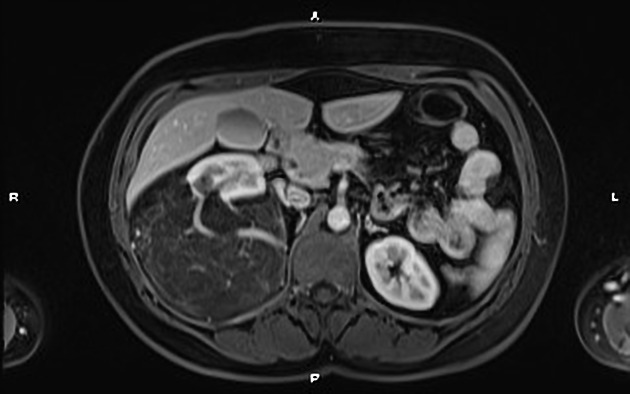
MRI
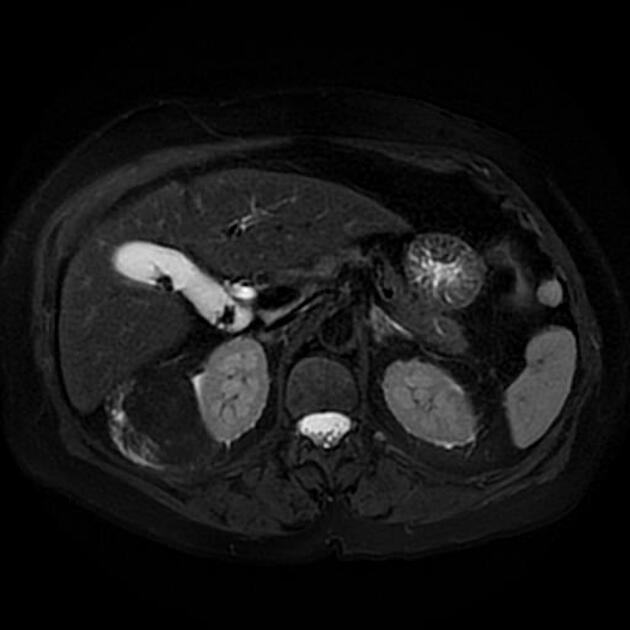
MRI is excellent at evaluating fat-containing lesions, and two main sets of sequences are employed. Firstly, angiomyolipomas demonstrate high signal intensity on non-fat-saturated sequences and loss of signal following fat saturation.
The second method is to use in-phase and out-of-phase imaging, which generates India ink artifact at the interface between fat and non-fat components. This can occur either at the interface between the angiomyolipoma and surrounding kidney or between fat and non-fat components of the mass 8. Chemical shift signal intensity loss and other features may suggest a fat-poor AML 15. Low T2 signal intensity is typical in fat-poor AMLs 28.
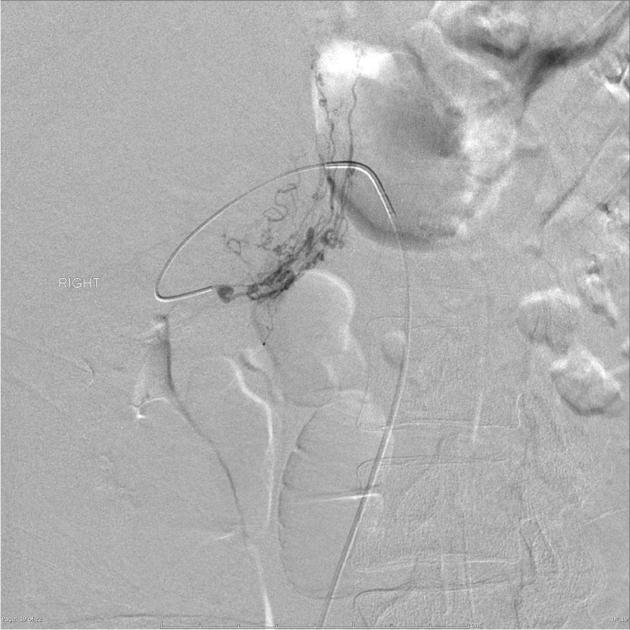
Digital subtraction angiography (DSA)
Angiomyolipomas are hypervascular lesions often demonstrating characteristic features:
arterial phase: a sharply marginated hypervascular mass with a dense early arterial network and tortuous vessels, giving the "sunburst" appearance
venous phase: whorled "onion peel" appearance of peripheral vessels
micro- or macro-aneurysms 2
absent arteriovenous shunting
Treatment and prognosis
Angiomyolipomas found incidentally often require no therapy, although follow-up has been recommended in the past to assess for growth. Size was considered an important risk factor for hemorrhage, however vascularity, the presence of aneurysms >5 mm and microaneurysms with incomplete arterial walls are now thought to be more important 29. Symptomatic AMLs may be more likely to bleed and women of child-bearing age should be carefully evaluated 29.
AMLs demonstrating risk factors can be electively embolized and/or resected with a partial nephrectomy.
Lesions that present with retroperitoneal hemorrhage often require emergency angioembolisation as a life-saving measure.
mTOR inhibitors (e.g., everolimus) have been shown to significantly decrease AML size in both TSC and sporadic cases, particularly the highly vascularized fat-poor component, with dramatic reduction in the size of the arteries and aneurysms thereby reducing the risk of hemorrhage and renal impairment 30. Side effects can be significant.
Differential diagnosis
When an AML has typical appearances, there is essentially no differential. If atypical, especially when fat-poor, other lesions to consider include:
-
macroscopic fat in RCC occurs with setting ossification/calcification, large irregular tumors invading the renal sinus and/or large necrotic tumors 11
focal or diffuse signal loss in opposed phase suggests microscopic fat (in contrast to the curvilinear India ink artifact seen with macroscopic fat), which is very common in clear cell RCC and less common in fat-poor AML 19
-
retroperitoneal liposarcoma invading the kidney
presence of a large vessel extending into the renal cortex suggestive of AML; liposarcoma is hypovascular
renal parenchymal defect at the site of tumor contact favors exophytic angiomyolipoma (claw sign)
calcifications can occur with liposarcoma
oncocytoma: may contain fat
Wilms tumor: may contain fat
perirenal fat entrapment / renal junctional parenchymal defect 11





 Unable to process the form. Check for errors and try again.
Unable to process the form. Check for errors and try again.