Ischemic stroke is an episode of neurological dysfunction due to focal infarction in the central nervous system attributed to arterial thrombosis, embolization, or critical hypoperfusion. While ischemic stroke is formally defined to include brain, spinal cord, and retinal infarcts 1, in common usage, it mainly refers to cerebral infarction, which is the focus of this article.
On this page:
Terminology
The term "stroke" is a clinical determination, whereas "infarction" is fundamentally a pathologic term 1. Bridging these terms, ischemic stroke is the subtype of stroke that requires both a clinical neurologic deficit and evidence of CNS infarction (cell death attributable to ischemia). The evidence of infarction may be based on imaging, pathology, and/or persistent neurologic symptoms, with other causes excluded. If there is imaging or pathologic evidence of an infarct but no attributable clinical symptoms, then it is called a "silent CNS infarction."
Epidemiology
Stroke is the second most common cause of morbidity worldwide (after myocardial infarction) and is the leading cause of acquired disability 2.
Risk factors for ischemic stroke largely mirror the risk factors for atherosclerosis and include age, gender, family history, smoking, hypertension, hypercholesterolemia, and diabetes mellitus.
Clinical presentation
An ischemic stroke typically presents with rapid onset neurological deficit, which is determined by the area of the brain that is involved. The symptoms often evolve over hours and may worsen or improve, depending on the fate of the ischemic penumbra.
The vascular territory affected will determine the exact symptoms and clinical behavior of the lesion:
Pathology
Interruption of blood flow through an intracranial artery leads to deprivation of oxygen and glucose in the supplied vascular territory. This initiates a cascade of events at a cellular level which, if circulation is not re-established in time, will lead to cell death, mostly through liquefactive necrosis.
The mechanism of vessel obstruction is important in addressing therapeutic maneuvers to both attempt to reverse or minimize the effects and to prevent future infarcts. Popular and simple etiological classifications of ischemic stroke include the TOAST classification and ASCOD classification 20.
Examples of etiologies include:
-
embolism
cardiac embolism (e.g. atrial fibrillation, ventricular aneurysm, endocarditis)
artery-to-artery (atherosclerotic) embolism (e.g. internal carotid artery stenosis, intracranial atherosclerotic disease)
tumor embolism 25
-
thrombosis
perforator thrombosis: lacunar infarct
acute plaque rupture with overlying thrombosis
Global cerebral hypoxia (e.g. as is seen in drowning or asphyxiation) is usually considered separately.
Etiologies more commonly seen in the pediatric or young adult demographic are discussed separately: see stroke in children and young adults.
Radiographic features
In many institutions with active stroke services which provide reperfusion therapies, a so-called code stroke aimed at expediting diagnosis and treatment of patients will include a non-contrast CT brain, CT perfusion and CT angiography.
Aging ischemic strokes can be important in a number of clinical and medicolegal settings. Both CT and MRI can help in determining when a stroke occurred as imaging features evolve in a reasonably predictable fashion. There is substantial heterogeneity in the terminology denoting time from onset. For the purposes of this article, the following definitions are used 10:
early hyperacute: 0 to 6 hours
late hyperacute: 6 to 24 hours
acute: 24 hours to 1 week
subacute: 1 to 3 weeks
chronic: more than 3 weeks
The above definition of hyperacute as 0-24 hours and acute as 1-7 days was affirmed by the international Stroke Recovery and Rehabilitation Roundtable 19. However, this group defined subacute as 1 week to 6 months (with 3 months dividing early and late subacute phases) and chronic as older than 6 months 19.
Video - acute infarction
CT
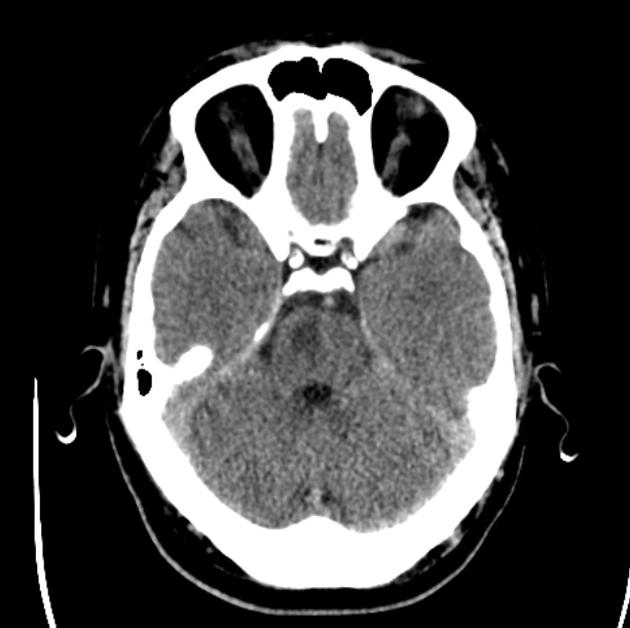
Non-contrast CT of the brain remains the mainstay of imaging in the setting of an acute stroke. It is fast, inexpensive and readily available. Its main limitation, however, is the limited sensitivity in the acute setting. Detection depends on the territory, the experience of the interpreting radiologist and of course the time of the scan from the onset of symptoms. Whether tissue is supplied by end arteries (e.g. lenticulostriate arteries) or has collateral supply (much of the cerebral cortex) will influence how quickly cytotoxic edema develops 6. For example, detection of MCA territory infarct has been shown to be approximately 60-70% in the first 6 hours 3, although changes in the deep grey matter nuclei (especially lentiform nucleus) can be visible within 1 hour of occlusion in up to 60% of patients 6.
The goals of CT in the acute setting are:
exclude intracranial hemorrhage, which would preclude thrombolysis
look for any "early" features of ischemia
exclude other intracranial pathologies that may mimic a stroke, such as a tumor
Non-contrast CT has also been used historically to exclude patients from receiving thrombolysis based on the extent of hypoattenuation at presentation. This criterion has, however, been removed from the 2018 American Heart Association guidelines 18. Nonetheless, finding large areas of established infarction on acute non-contrast CT continues to play an important role in patient selection and management.
Immediate
The earliest CT sign visible is the hyperdense vessel sign, representing direct visualization of the intravascular thrombus/embolus and as such is visible immediately 7,21. Although this can be seen in any vessel, it is most often observed in the middle cerebral artery (see hyperdense middle cerebral artery sign and middle cerebral artery dot sign) 21. It may be of therapeutic and prognostic value to differentiate this hyperdense 'regular' thromboembolic focus from a calcified cerebral embolus. In very rare instances of fat macroembolism, a hypodense vessel sign may be seen instead 22.
Early hyperacute
Within the first few hours, a number of signs are visible depending on the site of occlusion and the presence of collateral flow. Early features include:
-
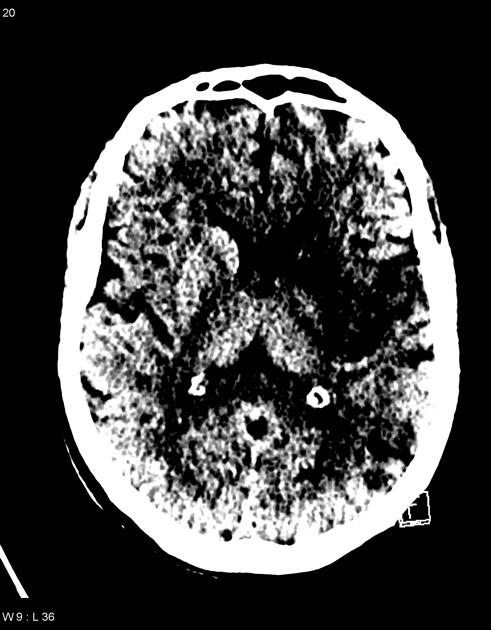
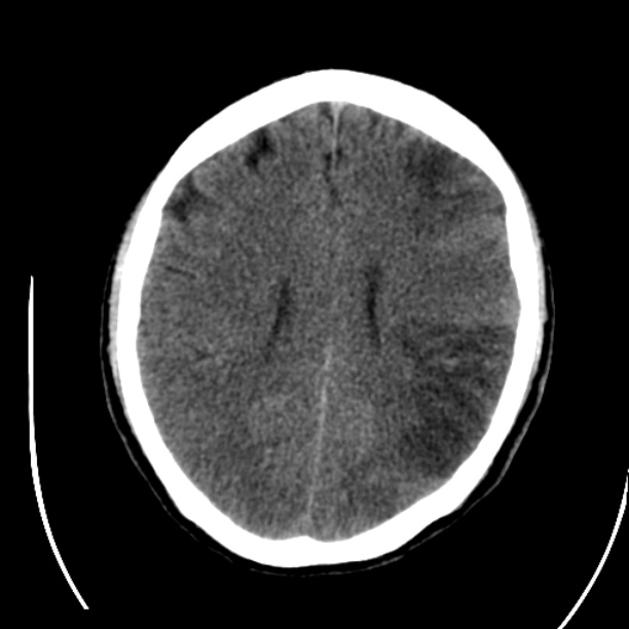
loss of grey-white matter differentiation, and hypoattenuation of deep nuclei:
lentiform nucleus changes are seen as early as 1 hour after occlusion, visible in 75% of patients at 3 hours 6
-
cortical hypodensity with associated parenchymal swelling with resultant gyral effacement
cortex which has poor collateral supply (e.g. insular ribbon) is more vulnerable 6
Visualization of loss of grey-white matter differentiation is aided by the use of a stroke window which has a narrow width and slightly lower center than routine brain window (width = 8, center = 32 HU) 18.
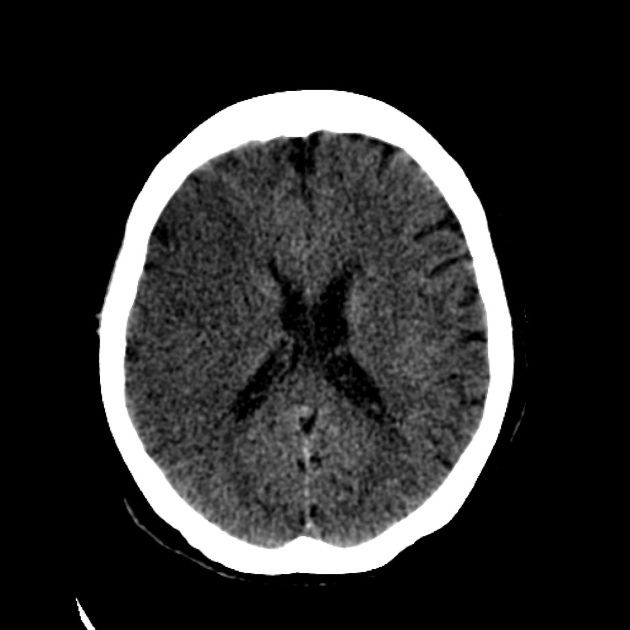
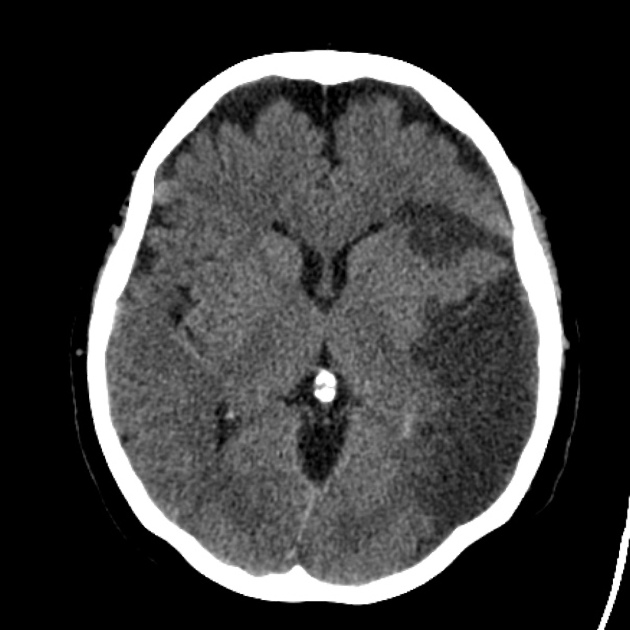
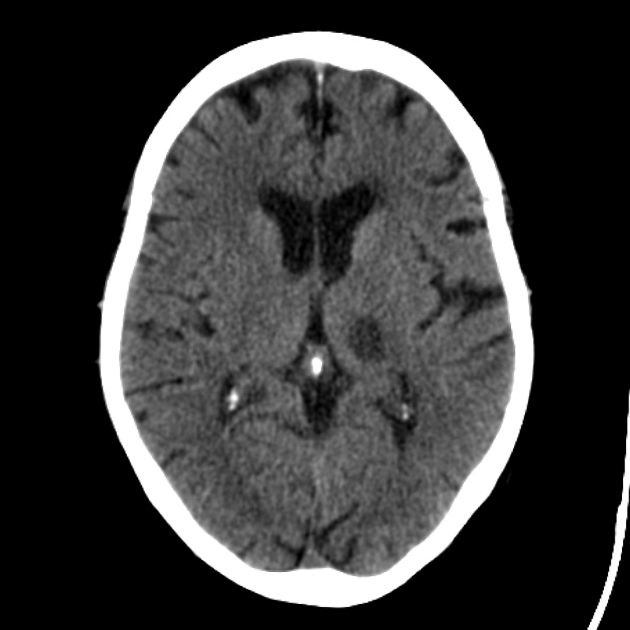
Acute
The hypoattenuation and swelling become more marked with time, resulting in a significant mass effect. This is a major cause of secondary damage in large infarcts.
Subacute
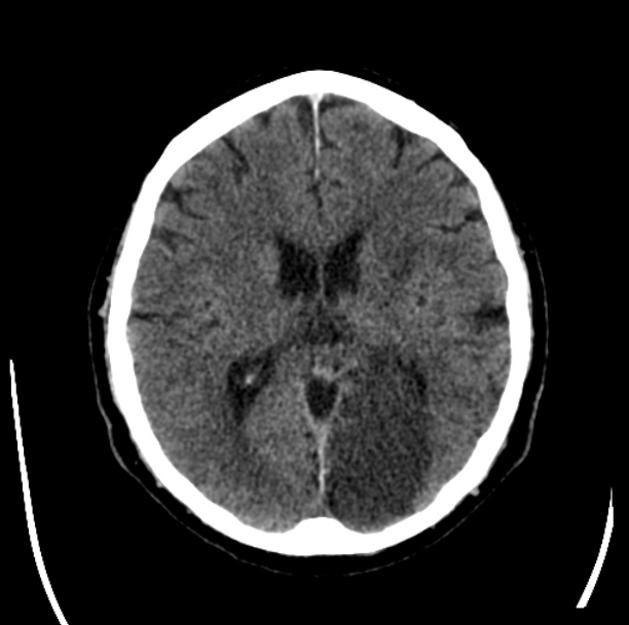
As time goes on, the swelling starts to subside and small amounts of cortical petechial hemorrhages (not to be confused with hemorrhagic transformation) result in elevation of the attenuation of the cortex. This is known as the CT fogging phenomenon 5. Imaging a stroke at this time can be misleading as the affected cortex will appear near normal.
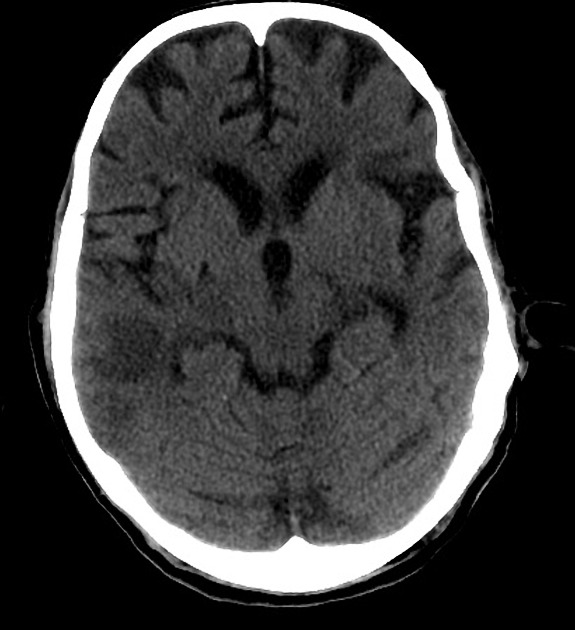
Chronic
Later still the residual swelling passes, and gliosis sets in eventually appearing as a region of low density with a negative mass effect. Cortical mineralization can also sometimes be seen appearing hyperdense.
Video - stroke evolution
CT perfusion
CT perfusion has emerged as a critical tool in selecting patients for reperfusion therapy as well as increasing the accurate diagnosis of ischemic stroke among non-expert readers four-fold compared to routine non-contrast CT 9.
It allows both the core of the infarct (that part destined to never recover regardless of reperfusion) to be identified as well as the surrounding penumbra (the region which although ischemic has yet to go on to infarct and can be potentially salvaged). CT perfusion may also demonstrate early evidence of associated crossed cerebellar diaschisis.
The key to interpretation is understanding a number of perfusion parameters:
Areas that demonstrate matched defects in CBV and MTT represent the unsalvageable infarct core, whereas areas that have prolonged MTT but preserved CBV are considered to be the ischemic penumbra 9.
These factors will be discussed further separately. See CT perfusion.
CT angiography
-
may identify thrombus within an intracranial vessel, and may guide endovascular clot retrieval
it is useful to determine whether the affected occluded vessel constitutes a large vessel occlusion (LVO) or middle vessel occlusion (MeVO)
-
evaluation of the carotid and vertebral arteries in the neck
establishing stroke etiology (e.g. atherosclerosis, dissection, carotid web, vertebral web)
assess endovascular access and potential limitation for endovascular treatment (e.g. tortuosity, stenosis, anatomical variants)
-
may be necessary prior to thrombolysis in pediatric stroke cases
some guidelines only advise that children with an arterial thrombus benefit from thrombolysis ref
assess collateral vessels using single-phase CTA
Multiphase or delayed CT angiography
Multiphase or delayed CT angiography is showing benefit either replacing CT perfusion or as an additional 4th step in the stroke CT protocol as it guides patient selection for endovascular therapy by assessing collateral blood flow in ischemic and infarct tissue 24.
MRI
MRI is more time consuming and less available than CT but has significantly higher sensitivity and specificity in the diagnosis of acute ischemic infarction in the first few hours after onset.
Early hyperacute
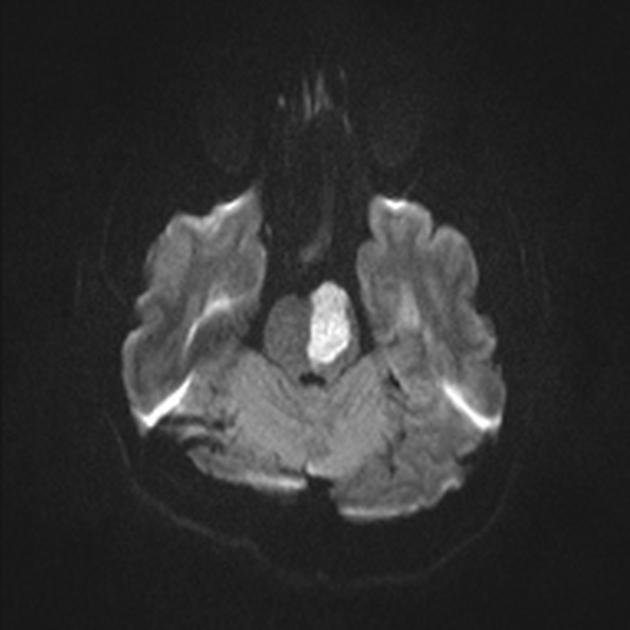
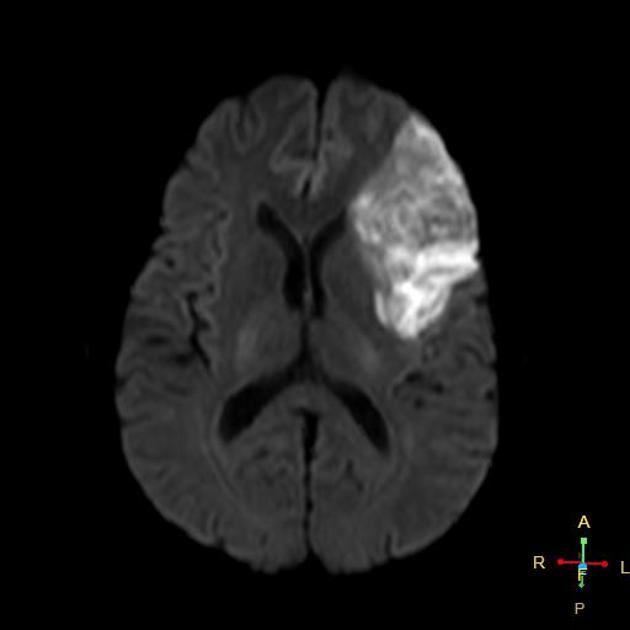
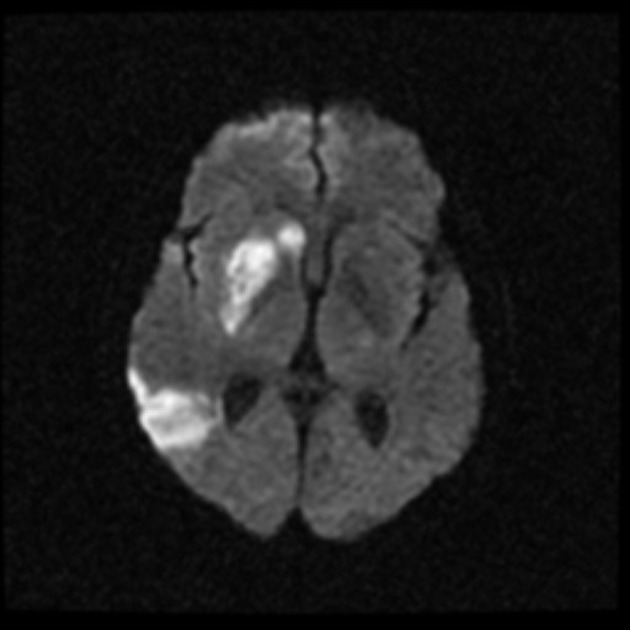
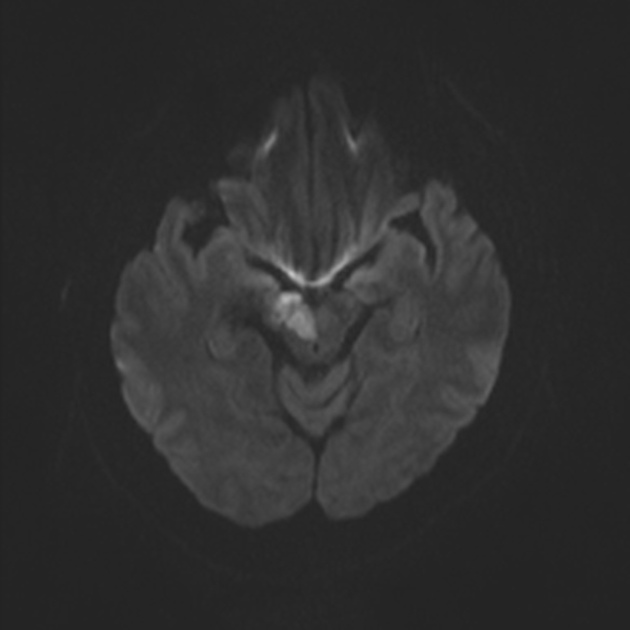
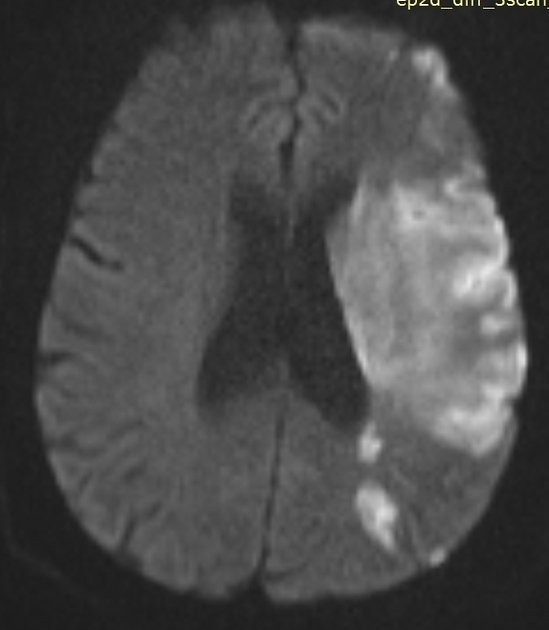
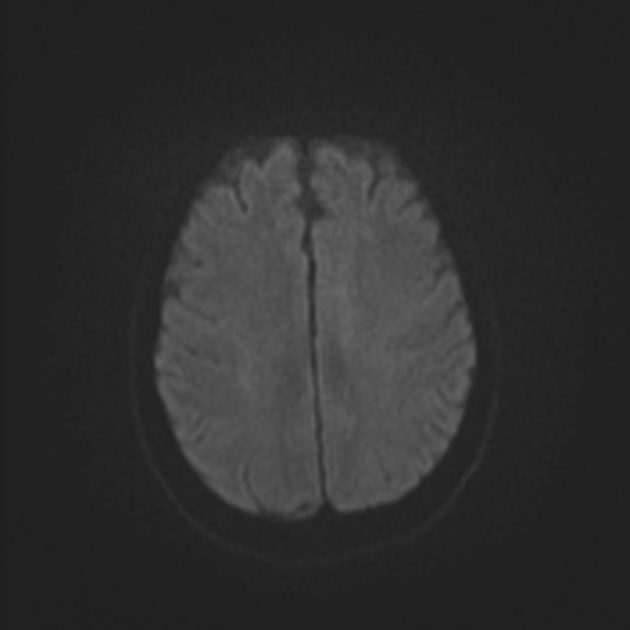
Within minutes of arterial occlusion, DWI demonstrates increased signal and reduced ADC values 4,10. This correlates well with infarct core (for a detailed discussion of DWI and ADC in stroke see diffusion-weighted MRI in acute stroke). At this stage, the affected parenchyma appears normal on other sequences, although changes in flow will be detected (occlusion on MRA) and the thromboembolism may be detected (e.g. the susceptibility vessel sign on SWI). Slow or stagnant flow in vessels may also be detected as a loss of normal flow void and high signal on T2/FLAIR and T1 C+ (intravascular enhancement), and presence of a the prominent vessel sign on SWI may indicate poor collateralisation 23.
If infarction is incomplete then cortical contrast enhancement may be seen as early as 2 to 4 hours 10.
In a minority of cases, DWI may be normal (please refer to DWI-negative acute ischemic stroke for more details).
Late hyperacute
Generally, after 6 hours, high T2 signal will be detected, initially more easily seen on FLAIR than conventional fast spin-echo T2 10. This change continues to increase over the next day or two.
T1 hypointensity is only seen after 16 hours 10 and persists.
Acute
During the first week, the infarcted parenchyma continues to demonstrate high DWI signal and low ADC signal, although by the end of the first week ADC values have started to increase. The infarct remains hyperintense on T2 and FLAIR, with T2 signal progressively increasing during the first 4 days. T1 signal remains low, although some cortical intrinsic high T1 signal may be seen as early as 3 days after infarction 10. After day 5 the cortex usually demonstrates contrast enhancement on T1 C+ 10. Less common patterns of enhancement include arterial enhancement, encountered in approximately half of strokes and becomes evident after 3 days, and meningeal enhancement which is uncommon and is usually seen between 2 and 6 days 10.
Hemorrhage, most easily seen on susceptibility-weighted imaging (SWI), is not a good indicator of age. Although most commonly seen after 12 hours and within the first few days, it may occur earlier or as late as 5 days 10.
Subacute
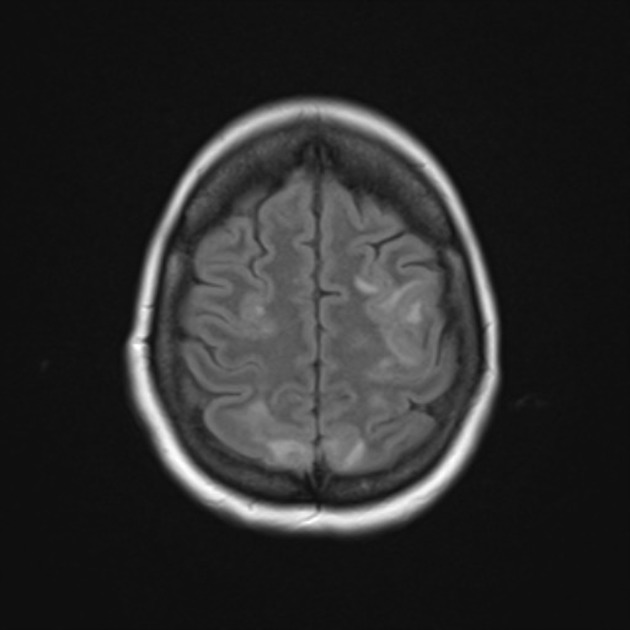
ADC demonstrates pseudonormalization typically occurring between 10-15 days 10. As ADC values continue to rise, infarcted tissue progressively gets brighter than normal parenchyma. In contrast, DWI remains elevated due to persistent high T2/FLAIR signal (T2 shine through), unless hemorrhage (T2 blackout) or cystic encephalomalacia 10. T2 fogging is also encountered typically between 1 and 5 weeks, most commonly around week 2 10,11. Cortical enhancement is usually present throughout the subacute period.
T1 weighted sequences continue to show hypointensity throughout the area of infarct with cortical intrinsic high T1 signal due to the liquefactive necrosis and influx of monocytes as a response. The terms "cortical laminar necrosis" or "pseudolaminar necrosis" are occasionally, but incorrectly, used to describe this appearance in the context of thromboembolic stroke, but should be restricted to use in cases of isolated cortical necrosis. See the article on cortical laminar necrosis for a fuller discussion of this.
Chronic
T1 signal remains low with intrinsic high T1 in the cortex if cortical necrosis is present 10. T2 signal is high. Cortical contrast enhancement usually persists for 2 to 4 months 10. Importantly if parenchymal enhancement persists for more than 12 weeks the presence of an underlying lesion should be considered 10.
ADC values are high. DWI signal is variable, but as time goes on signal progressively decreases.
Transcranial Doppler ultrasound
Often described as an emerging application of point-of-care ultrasonography, use of transcranial Doppler (TCD) sonography has been utilized for the diagnosis of intracranial vessel occlusion, as well as the differentiation between ischemic and hemorrhagic stroke 14.
In the context of a CT negative for intracerebral hemorrhage and a clinically suspicious patient presentation, diagnostic criteria for occlusion of an isolated vessel are as follows 12;
complete absence of color flow Doppler signals
absence of pulsed-wave Doppler signals
-
concurrent adequate visualization of surrounding parenchyma and vessels
color flow and pulsed wave Doppler signals must be demonstrated adequately in the remainder of the circle of Willis 16
Sonographic monitoring of the complications of ischemic stroke is also possible, including the detection of;
hemorrhagic transformation 16
-
elevated intracranial pressure (ICP)
measurement of the optic nerve sheath diameter (ONSD) in common use as an intracranial pressure surrogate
Treatment and prognosis
Acute treatment focuses on prompt application of reperfusion therapies:
intravenous or (rarely) intra-arterial thrombolysis (e.g. alteplase, tenecteplase)
-
endovascular clot retrieval for large vessel occlusions - response graded with TICI
trials have been negative for medium vessel occlusions 26,27
Neurosurgical intervention can also be pursued in certain cases, to allow patients to survive the period of maximal swelling by performing decompressive craniectomies (with or without duroplasty), particularly in younger patients with either large MCA infarcts or posterior fossa infarcts.
Additionally, supportive care should be provided, including caring for patients in dedicated inpatient stroke units and attempting to prevent the numerous complications which are encountered by patients with neurological impairment from stroke.
Complications
non-neurological complications: e.g. aspiration pneumonia, pressure ulcers, venous thromboembolism, etc.





 Unable to process the form. Check for errors and try again.
Unable to process the form. Check for errors and try again.