Autoimmune pancreatitis is a form of chronic pancreatitis associated with autoimmune manifestations on clinical, histological, and laboratory grounds 1.
Distinguishing this entity from other forms of chronic pancreatitis (such as alcohol-induced) is important as steroid treatment is effective both in reversing morphologic changes and also to return pancreatic function to normal 2.
On this page:
Epidemiology
Autoimmune pancreatitis is rare, with the exact incidence unknown but with an increasing number of cases reported in the literature 3. It is thought to account for 5-11% of chronic pancreatitis cases.
Age of presentation is variable, and there appears to be a male predilection (M:F 2:1) 3.
Associations
IgG4-related sclerosing cholangitis (up to 39%) 16
in Japanese patients: haplotypes DRB1 and DQB1
Diagnosis
Diagnostic criteria have been established, see the article diagnostic criteria for autoimmune pancreatitis for further details 3,8.
Clinical presentation
Clinical presentation is also variable, including 3,7:
new-onset diabetes mellitus
-
extrapancreatic manifestations, with lymphocytic infiltration
pulmonary involvement
renal involvement
elevated IgG and antinuclear antibody levels >50% of cases
abdominal pain and acute pancreatitis are unusual
Sometimes serum amylase and lipase are raised as well as a raised alkaline phosphatase ref.
Serum Ca 19-9 levels may be elevated in autoimmune pancreatitis, usually Ca 19-9 level <74 U/m are seen in autoimmune pancreatitis but higher in pancreatic cancer 20.
Pathology
Autoimmune pancreatitis is a type of chronic pancreatitis characterized by a heterogeneous autoimmune inflammatory process in which prominent lymphocytic infiltration with associated fibrosis of the pancreas causes organ dysfunction.
The cause is unknown though there is a strong basis for an autoimmune process where antibody reaction against carbonic anhydrase and lactoferrin has been postulated 9.
Subtypes
Macroscopic appearance
Diffusely indurated and firm pancreas, with some patients demonstrating a focal mass.
Microscopic appearance
Collar-like periductal infiltrates composed of lymphocytes and plasma cells 4, with the lymphocytes being CD8+ and CD4+ T lymphocytes.
Clusters of inflammatory cells are also seen in the walls of small veins and nerves as well as medium and large-sized vessels.
Interlobular septa are thickened by a proliferation of myofibroblasts and infiltrated by lymphocytes and plasma cells.
Other organs can also be involved with a lymphocytic infiltrate: gallbladder, biliary tree, kidney, lung and salivary glands more commonly.
Radiographic features
CT
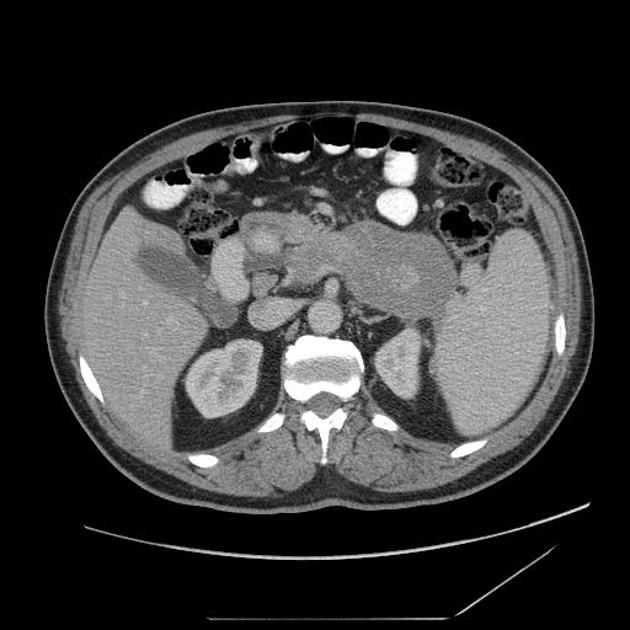
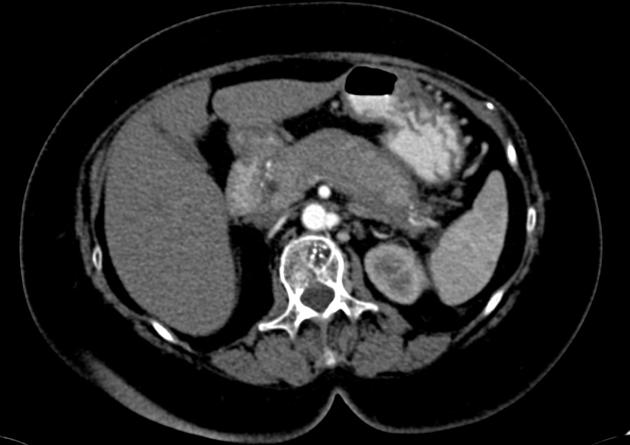
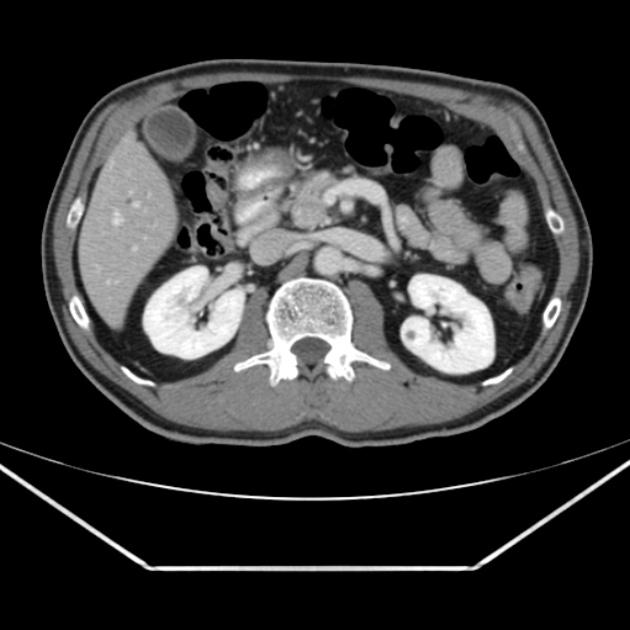
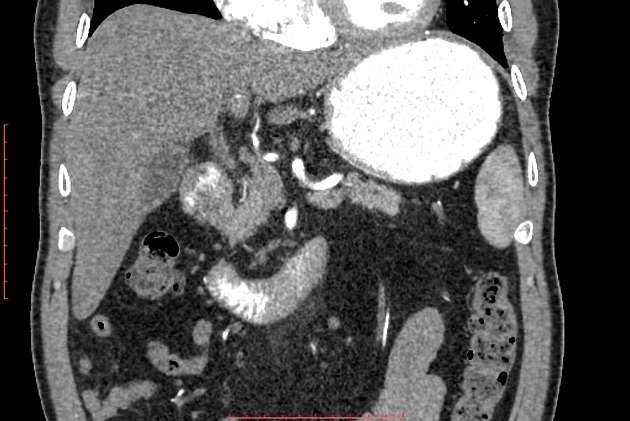
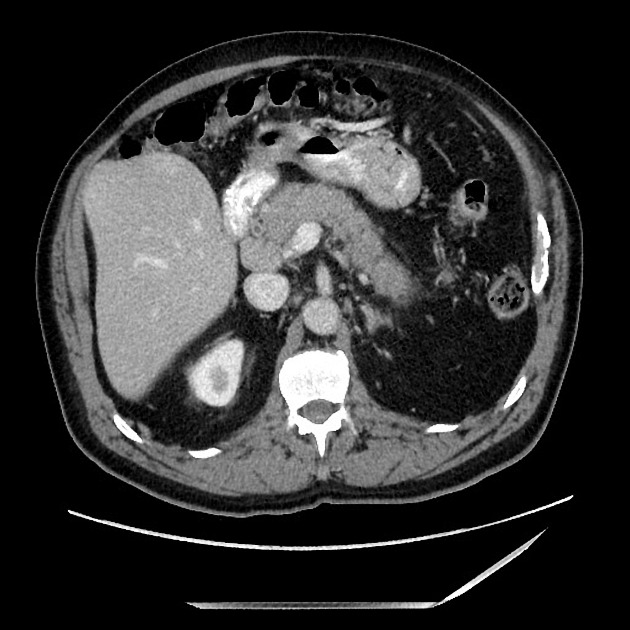
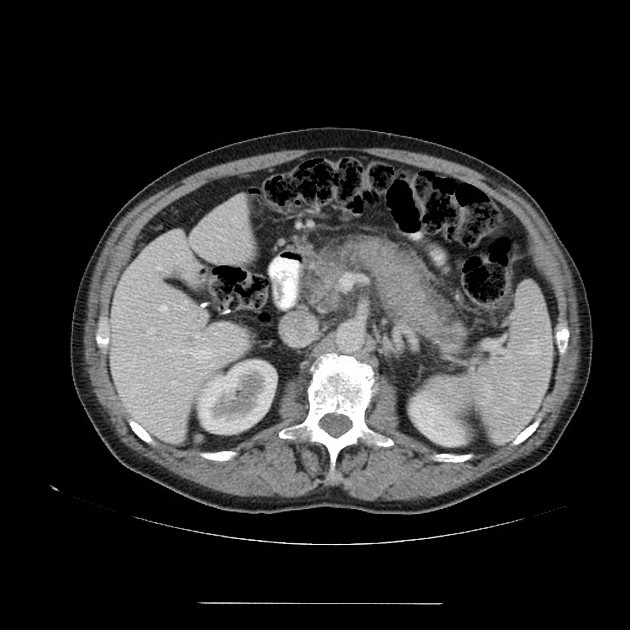
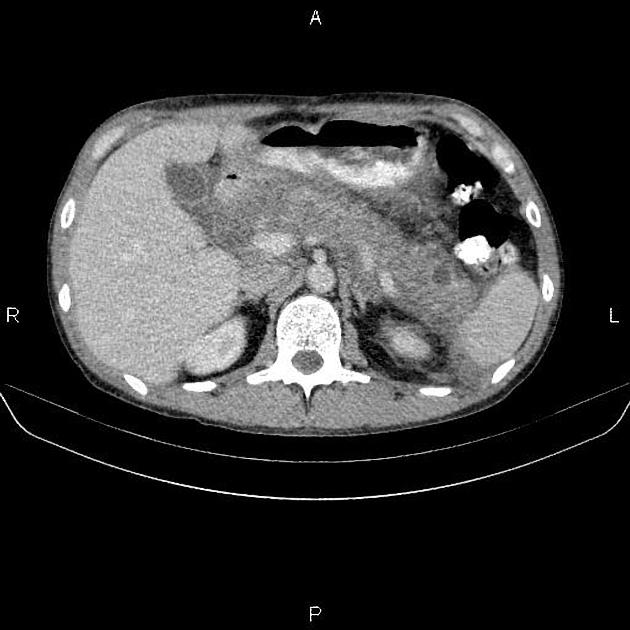
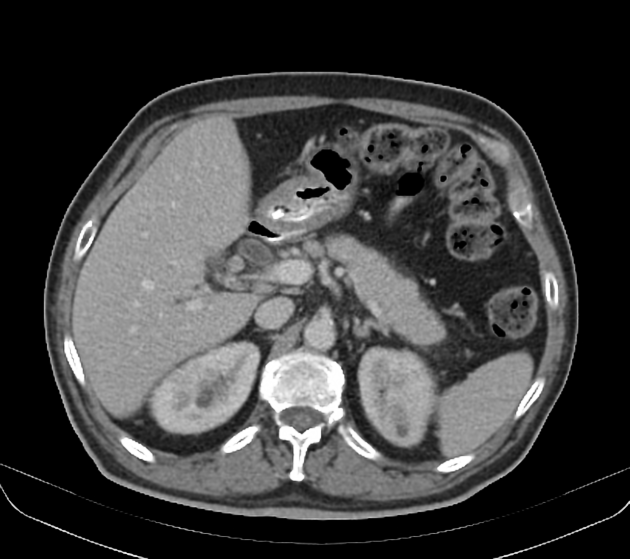
diffuse or focal enlargement of the pancreas with loss of definition of the pancreatic clefts; diffuse involvement results in "sausage-shaped pancreas" 17
minimal peripancreatic fat stranding, with inflammatory thickening confined to the peripancreatic region not extending into the mesentery, anterior pararenal fascia or lateroconal fascia
peripancreatic rim of low attenuation "halo" 5
when focal involvement, typically head and uncinate process and maybe iso- or hypo-dense to pancreatic tissue
the pancreas may appear normal
pancreatic dilatation and/or biliary dilatation may be seen
Contrast enhancement is helpful as autoimmune pancreatitis demonstrates reduced enhancement during the pancreatic phase (~40 seconds) but near normal enhancement on portal venous phase (70 seconds) 6.
Other sites also seem to be involved:
peripancreatic lymph nodes >1 cm
kidney: inflammatory pseudotumors; hypoattenuating lesions
retroperitoneal fibrosis: with periaortic soft tissue
pleural effusions
common bile duct: strictures
mediastinal nodes
MRI
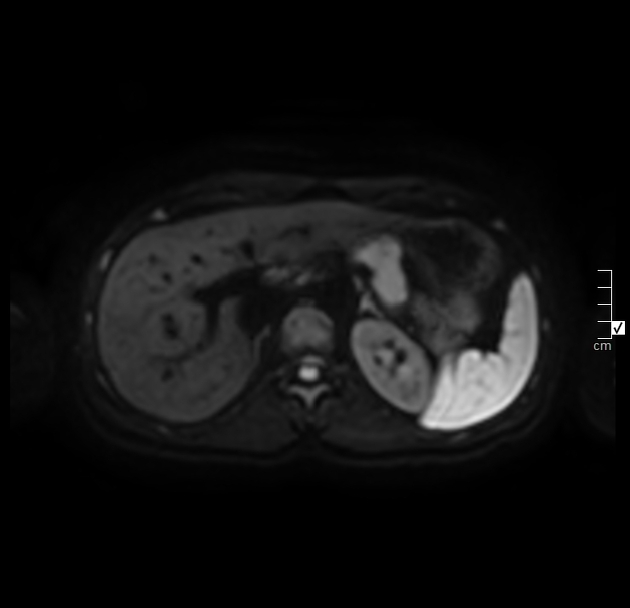
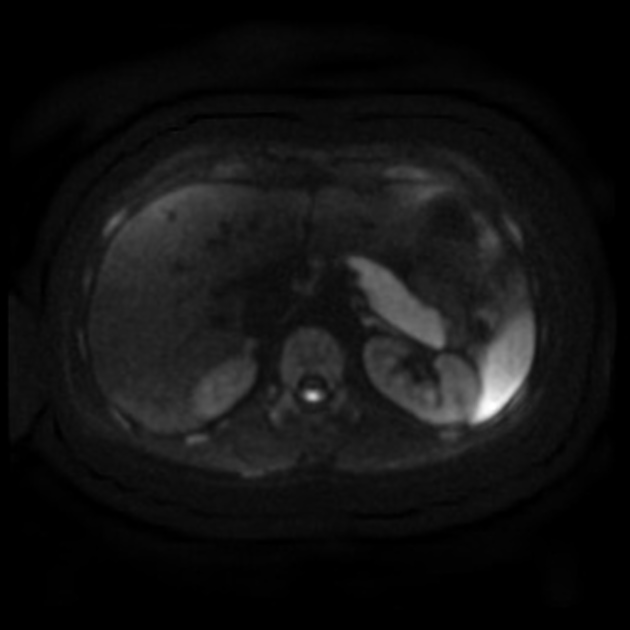
MRI demonstrates diffuse pancreatic enlargement 5,10.
T1: decreased signal intensity
T2: minimal increased signal intensity
T1 C+ (Gd): delayed parenchymal enhancement on dynamic imaging
-
DWI/ADC:
shows diffusion restriction with high DWI signal and low ADC signal 13,14
the reduction in ADC values can be lower than with pancreatic cancer 13
DWI signal may be useful as a marker for determining the indication for steroid therapy 15
MRCP: multiple intrahepatic duct strictures and common bile duct; diffuse narrowing of the main pancreatic duct
ERCP
ERCP can be normal. Biliary duct abnormalities are common with strictures of the common bile duct and short intrahepatic duct strictures representing primary sclerosing cholangitis.
Typically shows diffuse irregularity and narrowing of the main pancreatic duct (distinct from pancreatic carcinoma).
Treatment and prognosis
Treatment with steroids usually leads to the resolution both of morphological changes and a return to normal pancreatic function, with remission seen in 98% of cases 7. The condition is however also frequently self-limiting, with a spontaneous remission rate of up to 74% 7.
Relapse rates are high, seen in 24% of patients treated with steroids. The rate is higher in untreated patients 7.
Autoimmune pancreatitis accounts for 2-6% of patients who undergo pancreatic resection because they are suspected of having pancreatic cancer 5,10.
History and etymology
It was first described in 1995 by Yoshida et al 1.
Differential diagnosis
For the diffuse form consider:
lymphoma: pancreatic lymphoma
diffuse infiltrative pancreatic ductal adenocarcinoma
In these conditions, the pancreas is heterogeneously enhancing and an irregular pancreatic contour is seen.
For the focal form consider:
pancreatic ductal adenocarcinoma: adenocarcinomas tend to have minimal enhancement both on pancreatic (late arterial, ~40 seconds) and hepatic phase imaging, whereas autoimmune pancreatitis demonstrates near-normal enhancement during hepatic phase (portal venous, ~70 seconds) 6
combination of Ca 19-9 <74 U/ml and IgG4 >1.0 g/l distinguishes patients with autoimmune pancreatitis from those patients with pancreatic carcinoma with 94% sensitivity and 100% specificity 20.




 Unable to process the form. Check for errors and try again.
Unable to process the form. Check for errors and try again.