Intrauterine contraceptive devices (IUCD), also known as intrauterine devices (IUD) and colloquially commonly as the coil, are one of the most frequently used methods of contraception throughout the world. It prevents pregnancy by:
thinning the endometrial lining
preventing sperm motility
preventing implantation
There are two main types of intrauterine contraceptive devices:
-
non-hormonal
copper: pure copper or alloyed with gold/silver
stainless steel: also known as Chinese ring (manufacture ceased in 2000)
polyethylene plastic: Lippes loop (discontinued in 1980s) 14
hormonal, e.g. Mirena, Kyleena, Liletta, Skyla
On this page:
Terminology
The preferred abbreviation for an intrauterine contraceptive device is IUCD. Occasionally IUD is employed instead, however, this shortening is disliked by some specialists as it is also used as an abbreviation for intrauterine death.
Complications
-
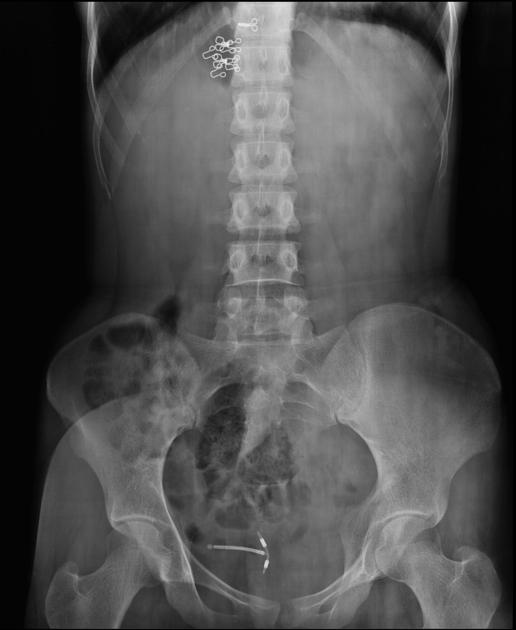
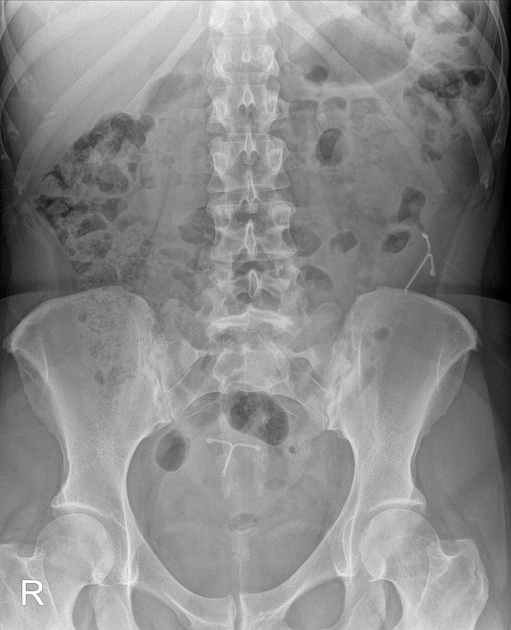
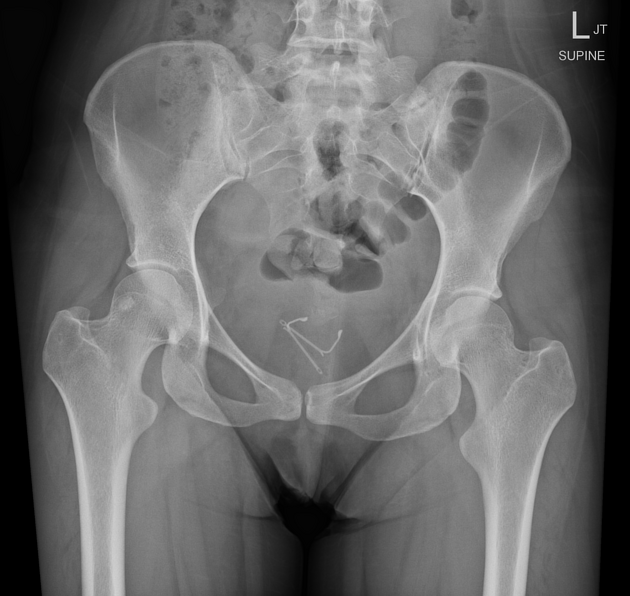
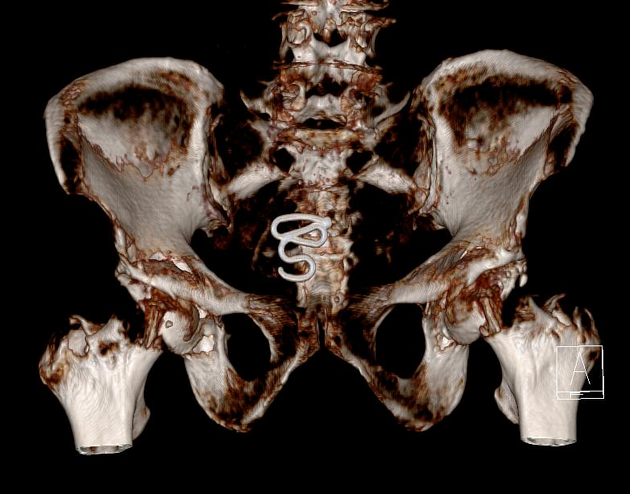
IUCD migration can take many forms 13
spontaneous IUCD expulsion: passage in or through the external cervical os
-
abnormal rotation or inferior position in the lower uterine segment or cervix
IUCD position >3-4 mm has been associated with an increased likelihood of IUCD related symptoms, such as pain and bleeding 2,9 as well as expulsion 10 - although further studies have shown that most low IUCDs migrate to the fundus after a few months 6
asymptomatic displacement of a hormone-releasing IUCD (e.g. Mirena®) may not affect its efficacy in prevention of pregnancy, according to some studies 6-8; in contrast, for copper IUCDs, displacement from the fundal position is associated with higher rates of inadvertent pregnancy 7
IUCD embedment: penetration into the myometrium, but not through the serosa
IUCD perforation: penetration through myometrium and serosa
-
three-fold increased risk of generalized pelvic inflammatory disease (PID)
associated pregnancy with spontaneous miscarriage
Radiographic features
Plain radiograph
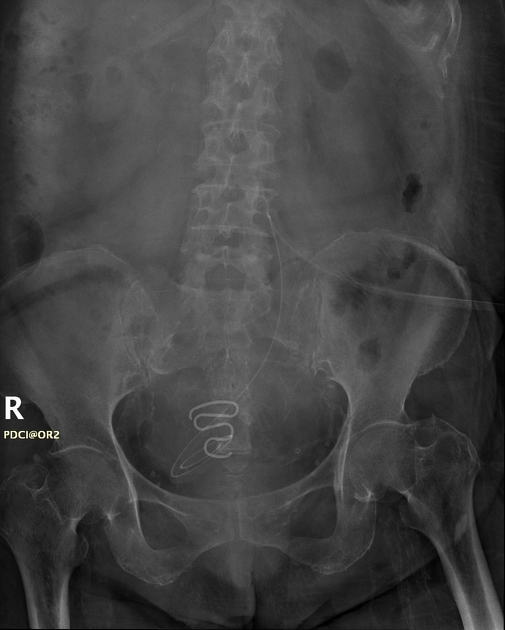
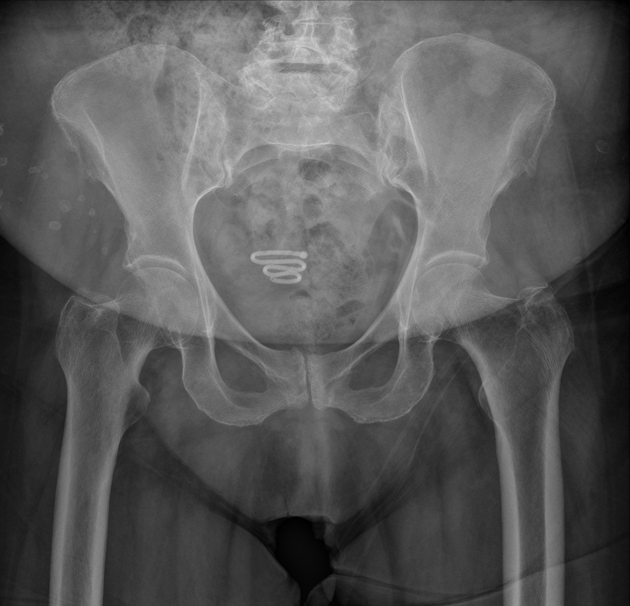
all intrauterine contraceptive devices are radiopaque (by design) 6
most often 'T-shaped' or at times seen as a serpiginous structure, as in the case of a Lippes loop 14
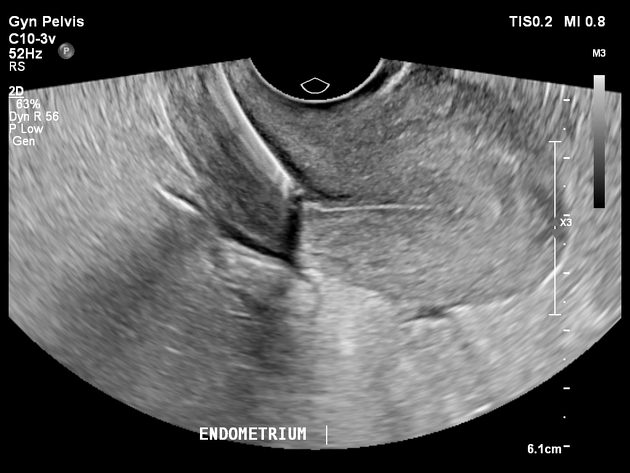
Ultrasound
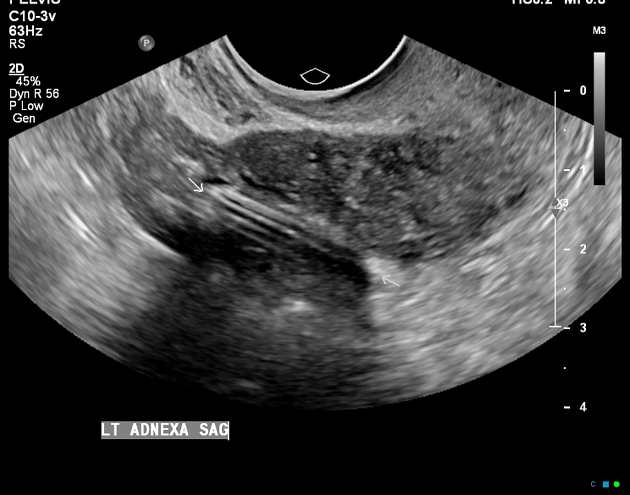
preferred modality for assessing an intrauterine contraceptive device
properly-placed IUCD may be visualized as a straight hyperechoic structure in the endometrial canal of the uterus and the arms of the IUCD extending laterally at the uterine fundus
often causes posterior acoustic shadowing
at evaluation of fundal placement of the IUCD, the distance from the top of the uterine cavity to the IUD should be ≤3 mm, a distance >4 mm is more often associated with symptoms such as bleeding and pain, as well as with a higher risk of expulsion or displacement 2 although most low IUCDs migrate to the fundus in a few months 6
in cases where it becomes embedded, a part of it may be visualized within the myometrium
3D ultrasound may be useful to help visualize the IUCD location (especially with serpiginous IUCDs) 2.
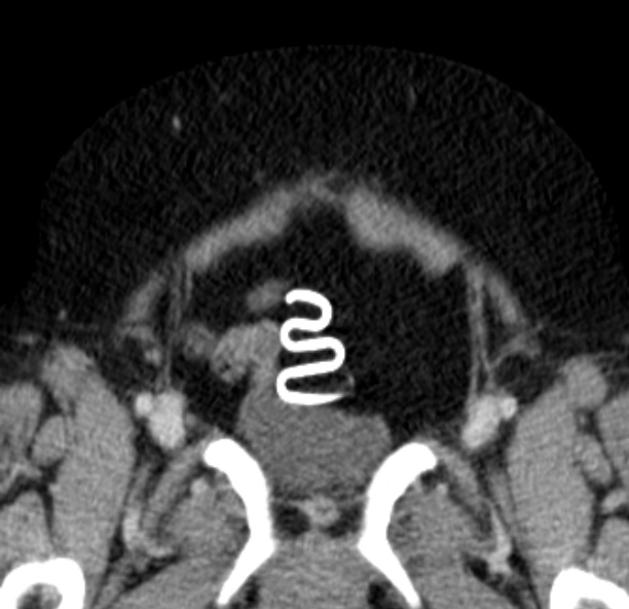
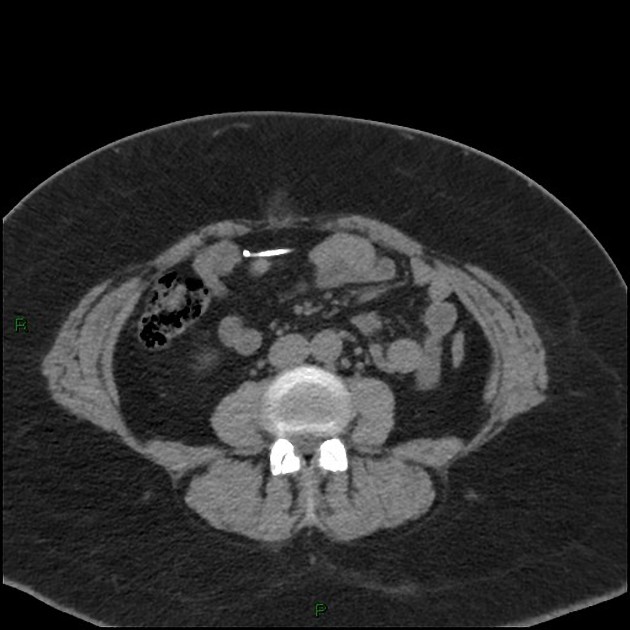
CT
hyperattenuating structures with metallic density
MRI
The MRI compatibility of intrauterine contraceptive devices may be of concern to women undergoing MRI examinations. Generally speaking, stainless steel devices are unsafe and non-metallic devices are considered safe. Copper devices have been found to be "conditionally safe" at 1.5 and 3.0 T 11.
History and etymology
The forerunner to the intrauterine contraceptive device was first introduced by the German physician, Richard Richter of Waldenburg, in 1909 4,5. His device comprised a loop of silkworm gut placed into the endometrial cavity 5.
Practical points
The contraceptive efficacy of non-fundal placement of an IUCD cannot be guaranteed. Measuring the distance between the fundal end of the cavity and the superior end of the IUCD is important when assessing placement. Low-lying IUCDs (those that are over 2 cm from the fundal end of the cavity) should be reported and the measurement included in the report 12.












 Unable to process the form. Check for errors and try again.
Unable to process the form. Check for errors and try again.