Lymphomas of the central nervous system, the most common of which is primary diffuse large B-cell lymphoma of the CNS, are the second most common primary brain tumor after gliomas 17. By definition, there is no co-existing systemic disease at the time of diagnosis, distinguishing it from CNS involvement from systemic lymphoma (secondary CNS lymphoma).
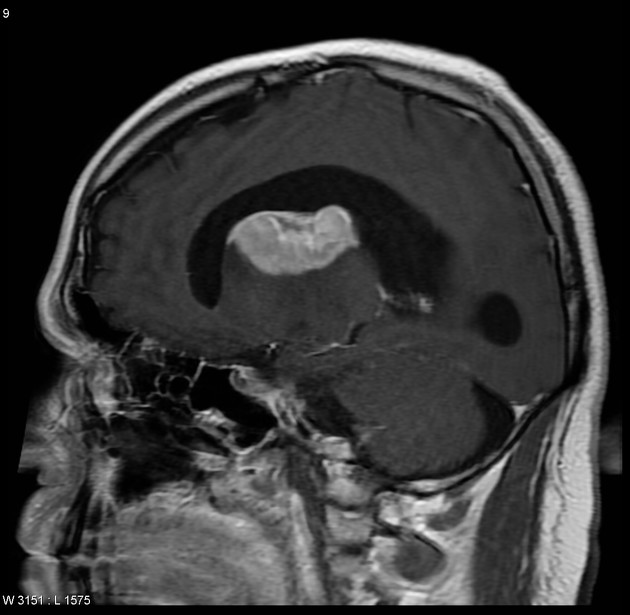
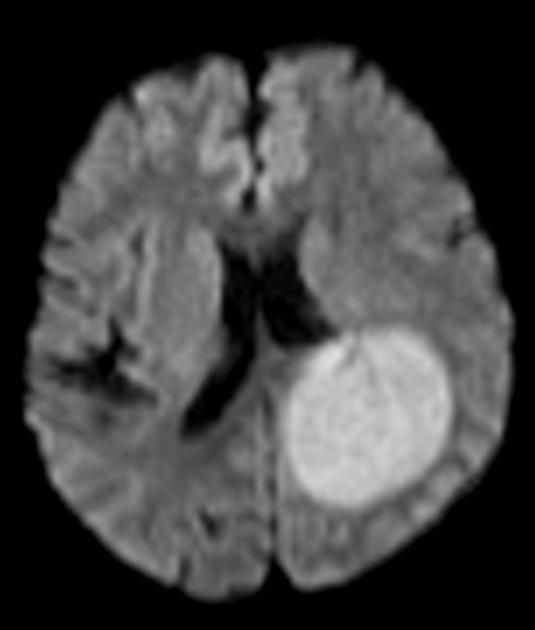
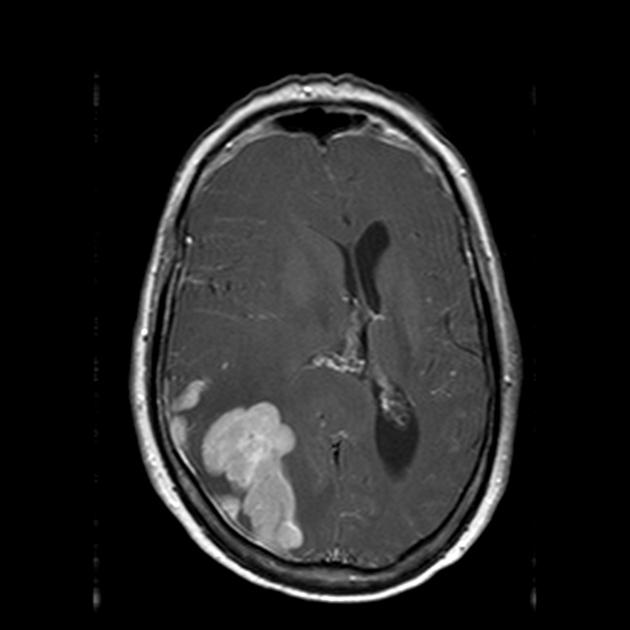
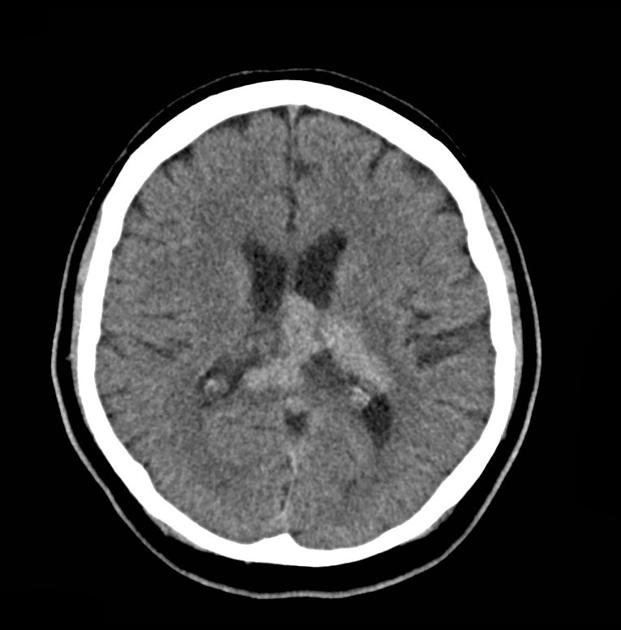
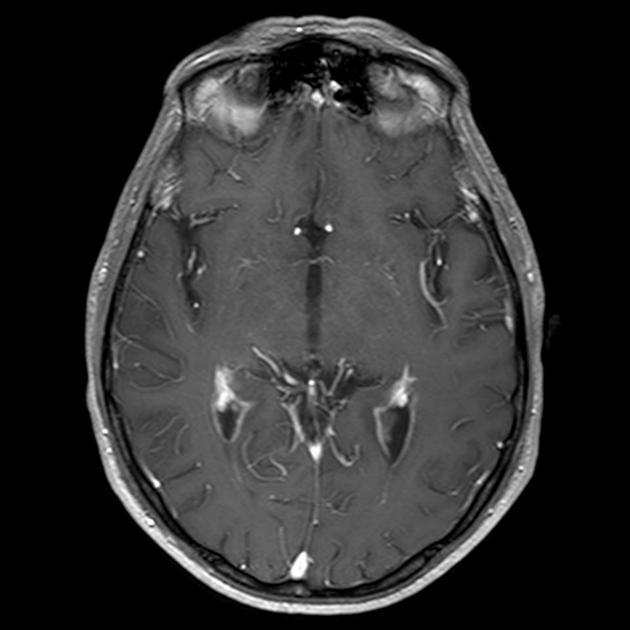
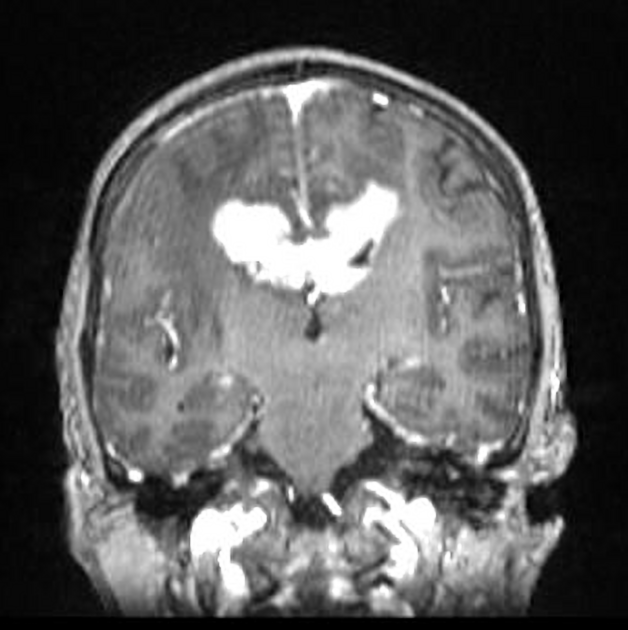
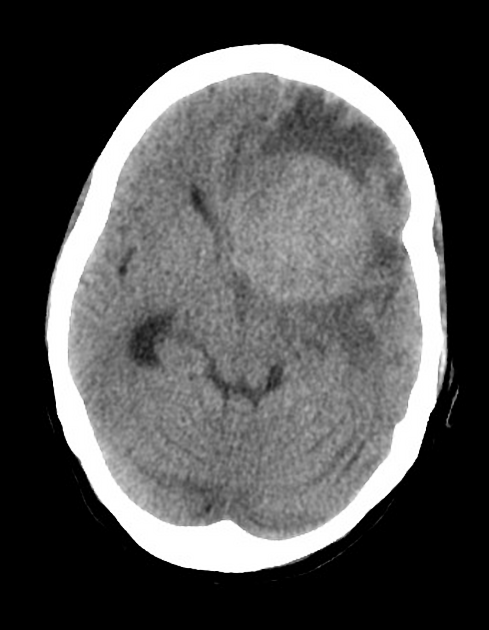
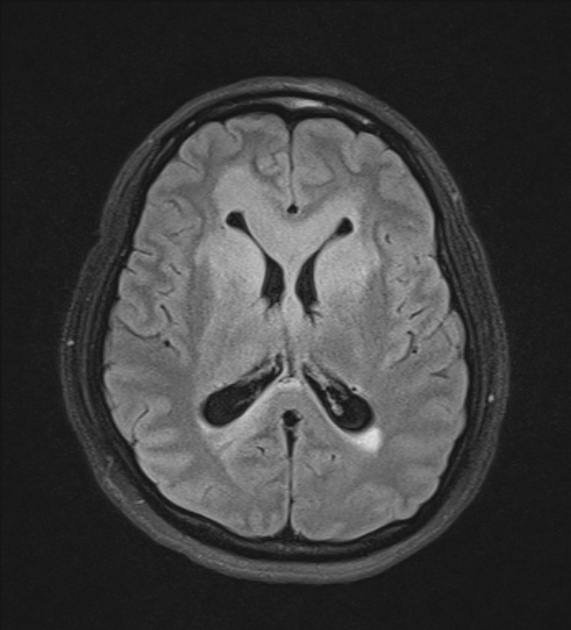
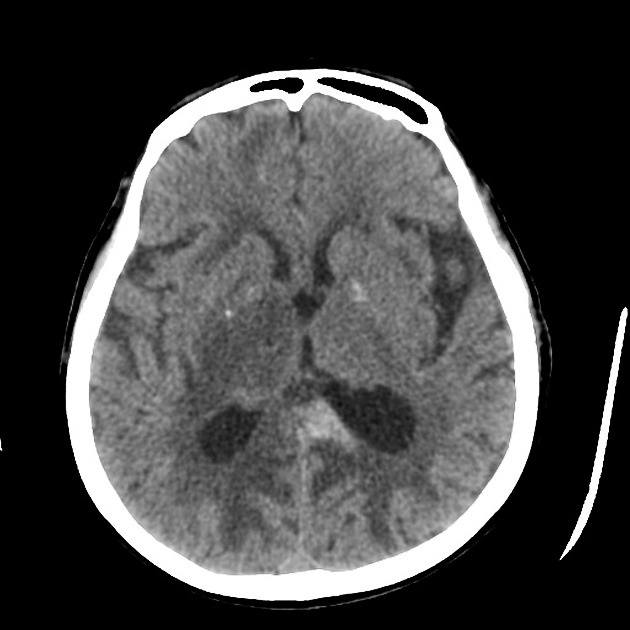
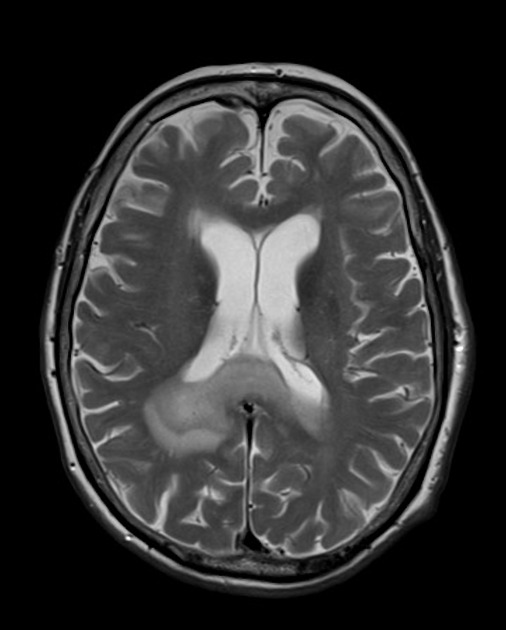
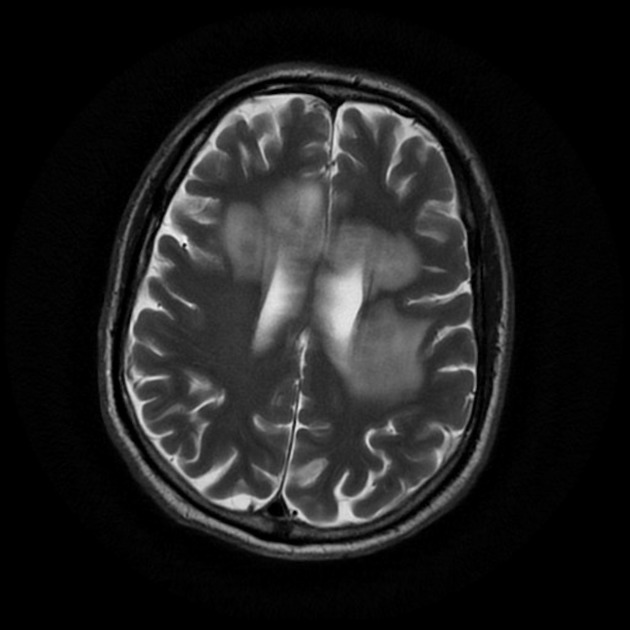
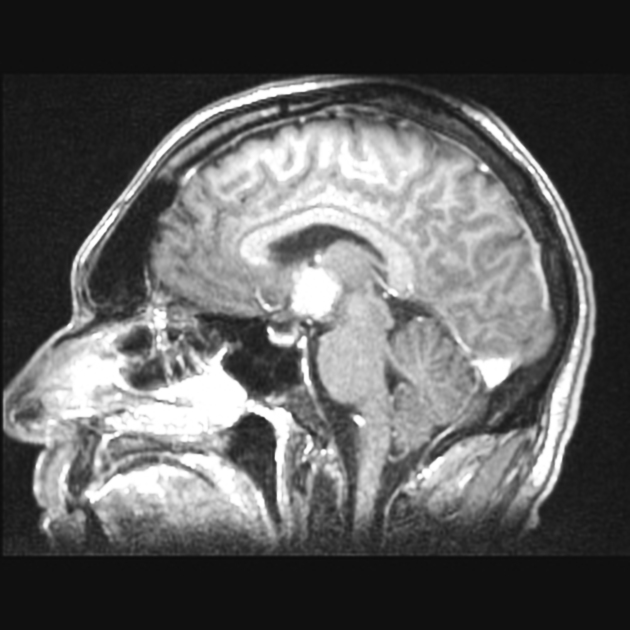
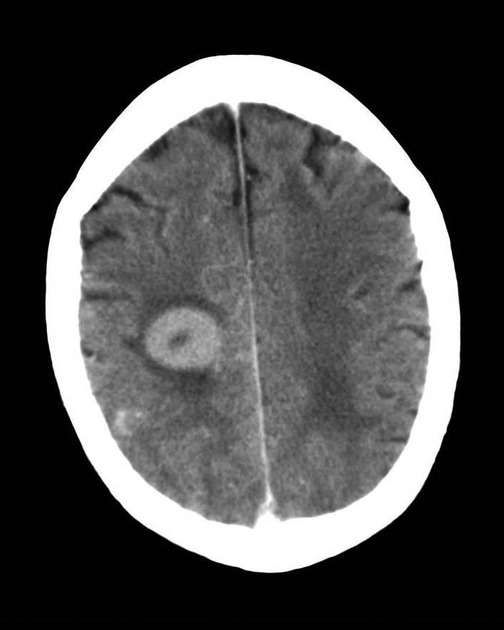
On imaging, primary diffuse large B-cell lymphoma of the CNS typically appears as a CT hyperdense enhancing supratentorial mass, with MRI T1 hypointense, T2 iso- to hypointense, vivid homogeneous enhancement and restricted diffusion. Usually, there is relatively little associated vasogenic edema and no central necrosis.
It is important to note that in immunocompromised individuals or other lymphoma subtypes, appearances can be different and more heterogeneous.
On this page:
Terminology
The WHO classification of CNS tumors divides CNS lymphomas into a number of subtypes, based on the cell of origin, and molecular and histological features.
-
CNS lymphomas
primary diffuse large B-cell lymphoma of the CNS
-
miscellaneous rare lymphomas in the CNS
other low-grade B-cell lymphomas of the CNS
It is worth noting that the WHO classification of hematolymphoid tumors (5th edition, 2022) overlaps the CNS classification and groups primary diffuse large B-cell lymphoma of the CNS in with tumors of the vitreous and testes under the primary DLBCL of immune-privileged sites 17.
The remainder of this article presents a general discussion of primary diffuse large B-cell lymphoma of the CNS, which is by far the most common type of CNS lymphoma, accounting for 80-85% of cases 17. Other types are discussed separately.
Epidemiology
Typically patients diagnosed with primary CNS lymphoma are over the age of 50 years with a short duration of symptoms (at most a few months) 4. There is a male predominance of approximately 2:1 4.
Historically there has been a strong association with HIV/AIDS and other immunocompromised states and demographics in these patients reflect the underlying condition – see immunodeficiency-associated CNS lymphomas.
There has been an increase in the incidence of sporadic, non-EBV-associated primary CNS lymphomas in immunocompetent individuals, which is particularly seen in older patients (50-80 years of age) 14.
Clinical presentation
Patients with primary CNS lymphoma present similarly to patients with other central nervous system masses; symptoms and signs of raised intracranial pressure, focal neurological disorders and seizures. If occular involvement is present then changes in vision may be present 25.
Pathology
The vast majority (>90%) of primary CNS lymphomas are B-cell in origin: diffuse large B-cell lymphoma and high-grade Burkitt-like B-cell lymphoma 1. Malignant cells tend to accumulate around and within blood vessels. Low-grade tumors are more frequently T-cell in origin 1.
Effects of steroids
An important factor to be aware of is the transient but profound response of CNS lymphoma to the use of glucocorticoids (e.g. dexamethasone and prednisolone) which are routinely administered in patients with intracranial mass effect from a tumor and edema. Within a few days of administration of steroids, CNS lymphoma can shrink dramatically due to the combined effect of steroid as a cytotoxic agent (reducing the neoplastic B-cell population) and anti-edema agent (resulting in decreased permeability of capillaries via a variety of mechanisms) 12,16. Whatever the underlying mechanism, the result is that administration of steroid prior to biopsy prevents a diagnosis in as many as 50% of cases 14, although some studies have suggested that this effect is perhaps not as profound as initially suspected 16.
Nonetheless, if the diagnosis is suspected and the patient does not require steroids emergently to treat mass effect, then they are best avoided prior to biopsy to maximize the diagnostic yield 16.
Location
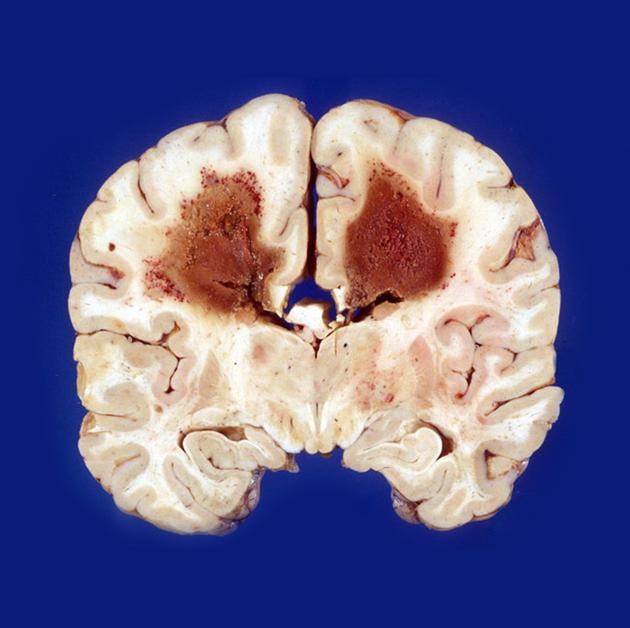
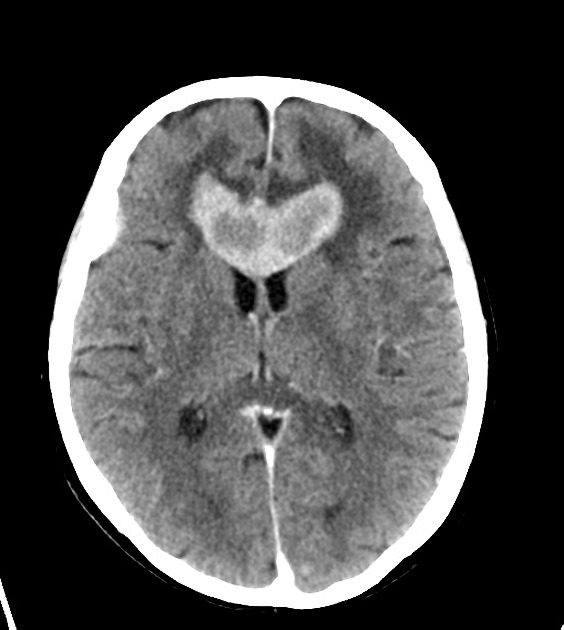
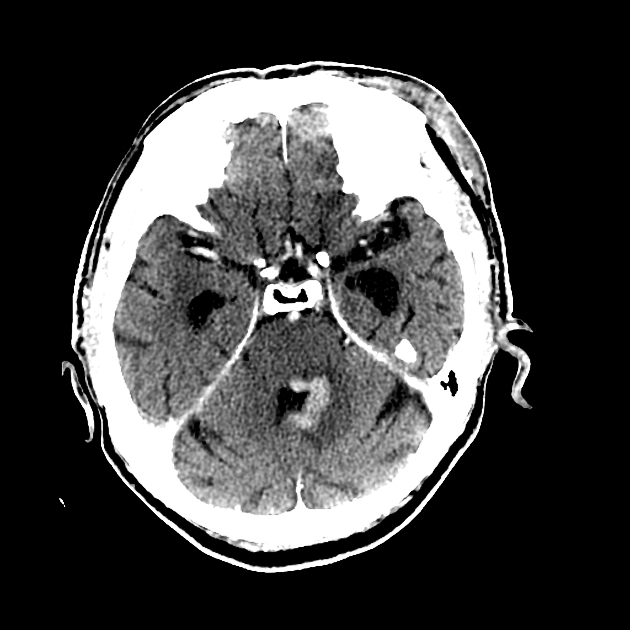
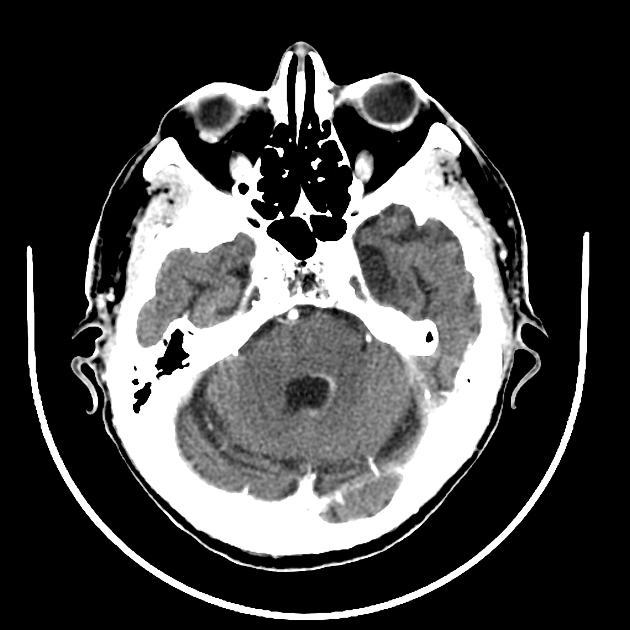
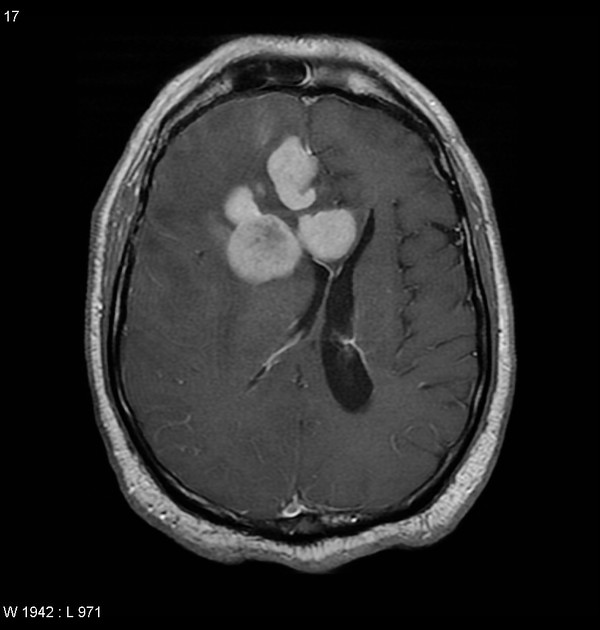
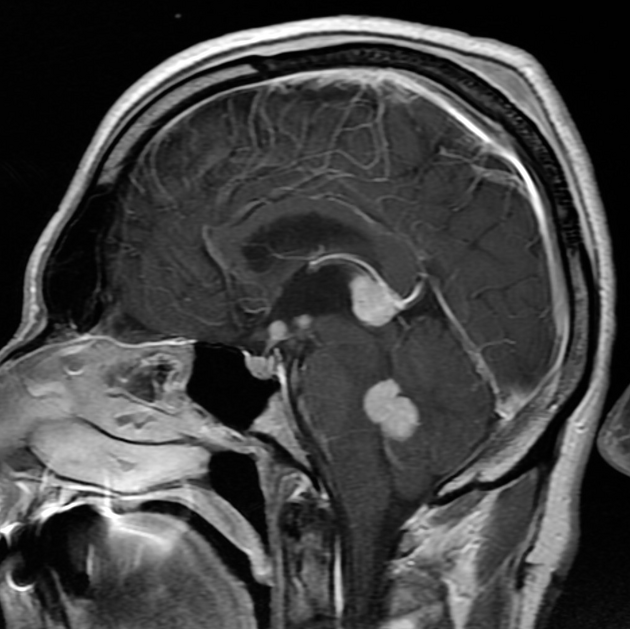
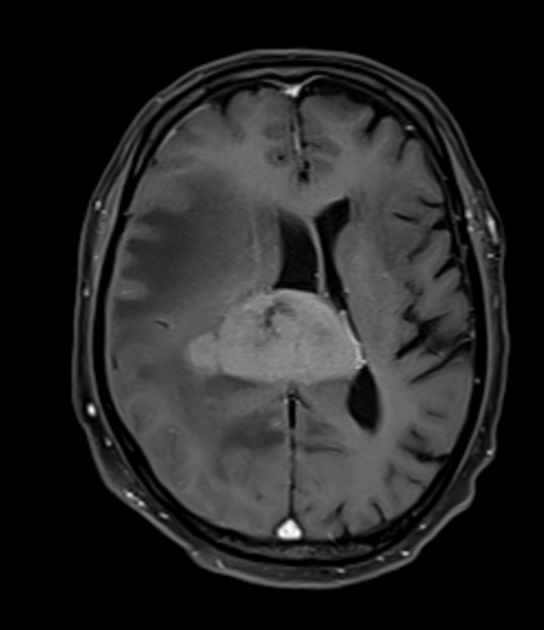
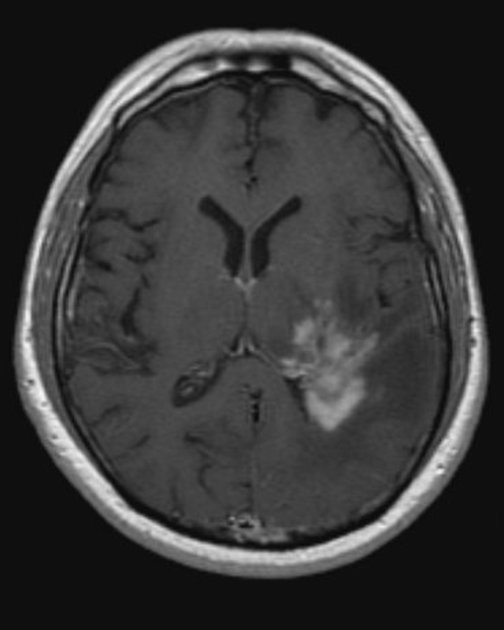
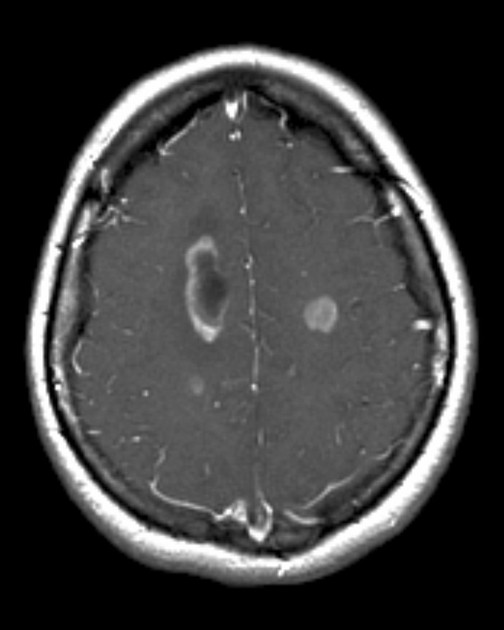
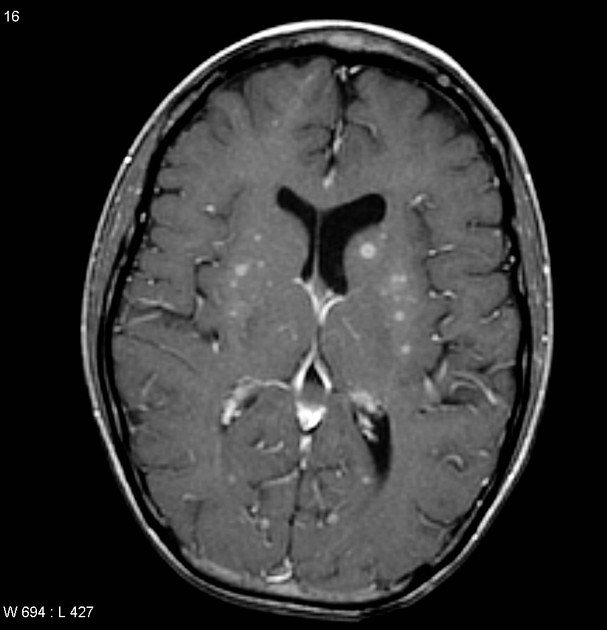
Primary CNS lymphomas present as solitary (60-70%) or multiple (30-40%) lesions with a predilection for the periventricular white matter, although they can also arise in the cortex or deep grey matter 3,5,17. Although leptomeningeal and ependymal deposits are common, if no substantial parenchymal mass is present, then secondary lymphoma should be favored 17.
They are most frequently found in the supratentorial brain (70-80%), uncommon in the posterior fossa and distinctly rare in the spinal cord 14,17. Rarely, a primary central nervous system lymphoma may presents with involvement of the ocular globe 25. This is most frequently bilateral (up to 90%) and often has concurrent involvement of other parts of the central nervous system (16-34%) 25.
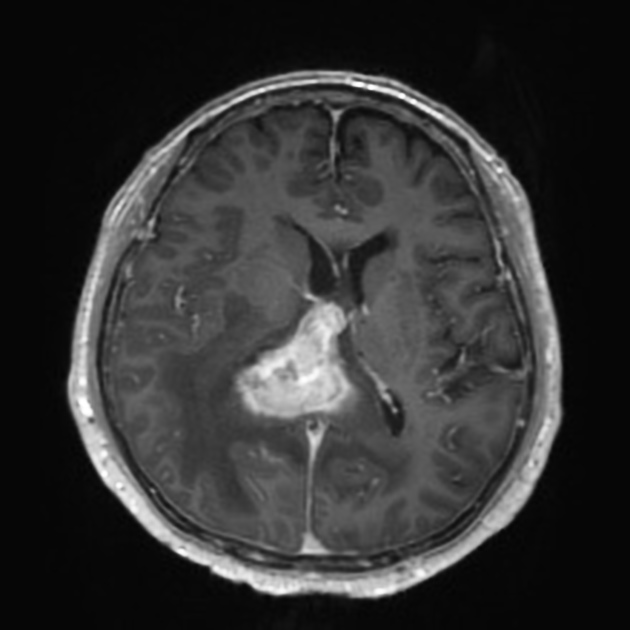
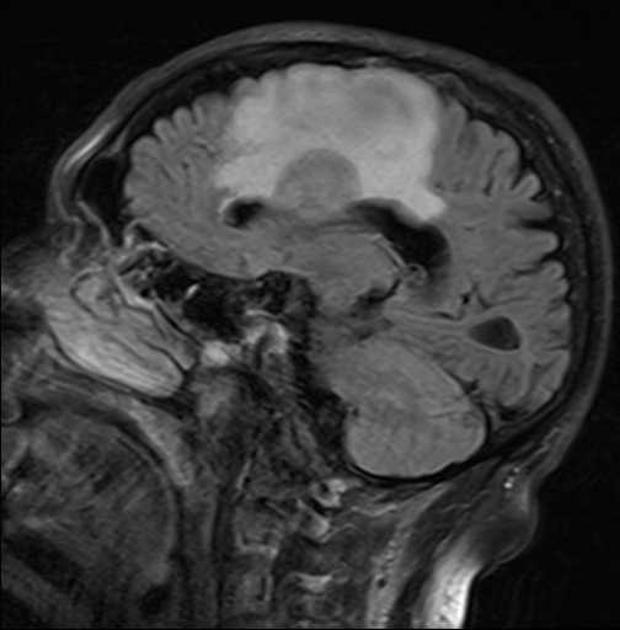
Crossing of the corpus callosum fairly common and a helpful feature.
Macroscopic appearance
Macroscopic appearance is variable, ranging from almost indistinguishable from the normal brain to well-circumscribed masses to heterogeneous ill-defined hemorrhagic or necrotic masses 14.
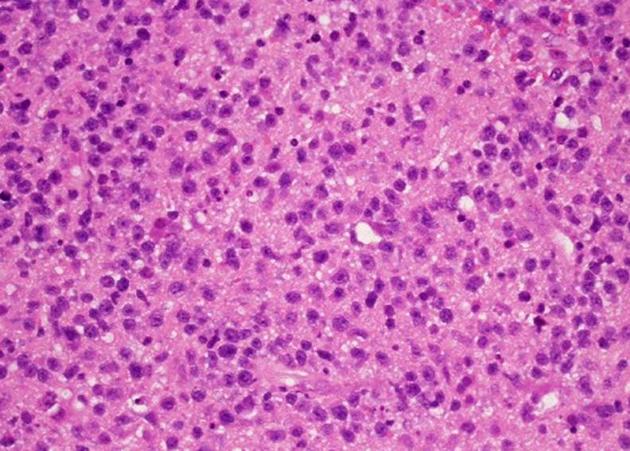
Microscopic appearance
Primary CNS lymphomas are composed of large quantities of lymphocytes without a particular growth pattern, although there is a predilection for a perivascular distribution, and in many instances infiltration within blood vessels 14. They may demonstrate areas of necrosis, especially in immunodeficient patients.
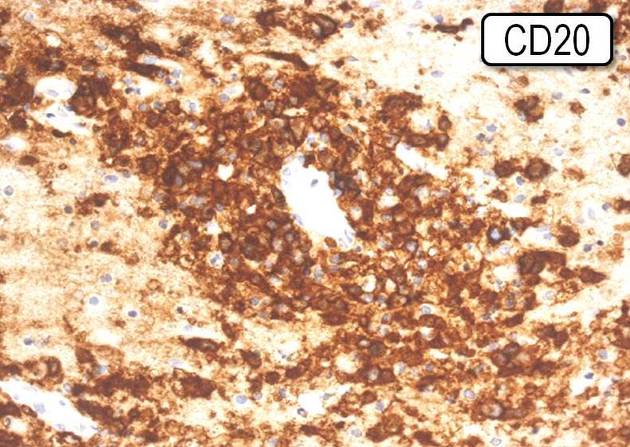
Immunophenotype
The exact immunophenotype depends on the tumor type.
Diffuse large B-cell lymphoma, which is the most common, is characterized by immunohistochemical reactivity for CD19, CD20, CD22, CD79a and PAX-5 14.
CSF
CSF examination demonstrates elevated protein and decreased glucose. Positive cytology is uncommon (~25%). Positive EBV DNA in CSF is helpful for the diagnosis of lymphoma, particularly in immunocompromised individuals.
Radiographic features
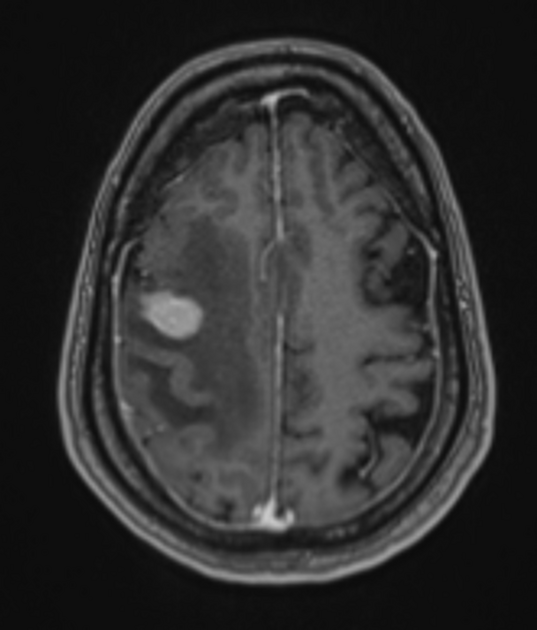
Classic imaging appearance for primary CNS lymphoma is of a CT hyperdense avidly enhancing mass, with T1 hypointense, T2 iso- to hypointense, vivid homogeneous gadolinium-enhancing lesion(s) with restricted diffusion on MRI, and exhibiting subependymal extension and crossing of the corpus callosum 17.
While this typical pattern is helpful in diagnosis, it is predominantly observed in untreated non-immunocompromised patients; diffuse large B-cell lymphoma, EBV negative 17.
Primary CNS lymphomas in immunocompromised patients (typically HIV/AIDS or post-transplant), or other types of lymphoma may be more heterogeneous in appearance. For example in immunocompromised individuals central non-enhancement/necrosis and hemorrhage is more common, although the latter is still uncommon – see immunodeficiency-associated CNS lymphomas 8.
Enhancement on both CT and MRI is pronounced and usually homogeneous. Even with larger lesions, there is little mass effect for size and limited surrounding vasogenic edema.
Low-grade tumors differ from the more common high-grade primary CNS lymphomas in several ways 1:
deep locations and spinal involvement is more common
contrast enhancement is absent, irregular or only mild
Disseminated meningeal or intraventricular disease is uncommon. It is seen in ~5% (range 1-7%) of cases at presentation and usually in high-grade cases 8.
CT
most primary CNS lymphomas are hyperattenuating (70%) 3
show enhancement
hemorrhage is distinctly uncommon 8
often multiple lesions in patients with HIV/AIDS
MRI
MRI signal characteristics of untreated primary CNS lymphoma presenting as masses in immunocompetent individuals are fairly stereotypical allowing a pre-operative diagnosis to be made with confidence in most cases.
T1: typically hypointense to grey matter 9
-
T1 C+ (Gd)
intense homogeneous enhancement is most common
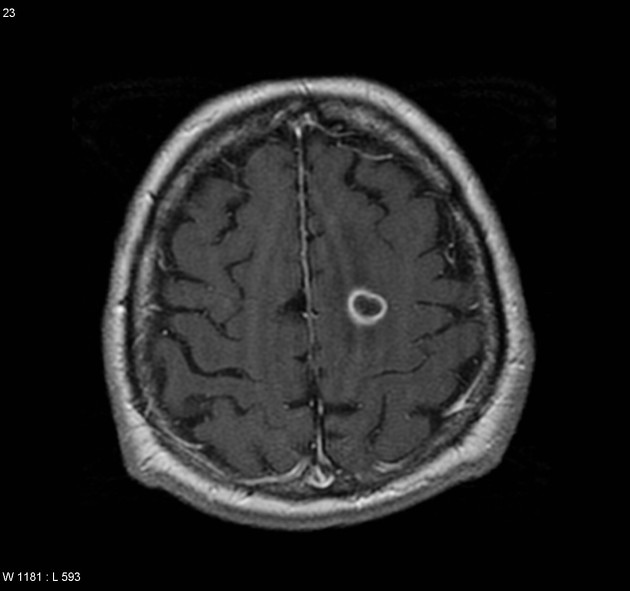
peripheral ring enhancement may be seen in 10-15% of cases 17 (especially in immunocompromised patients)
notch sign 15
-
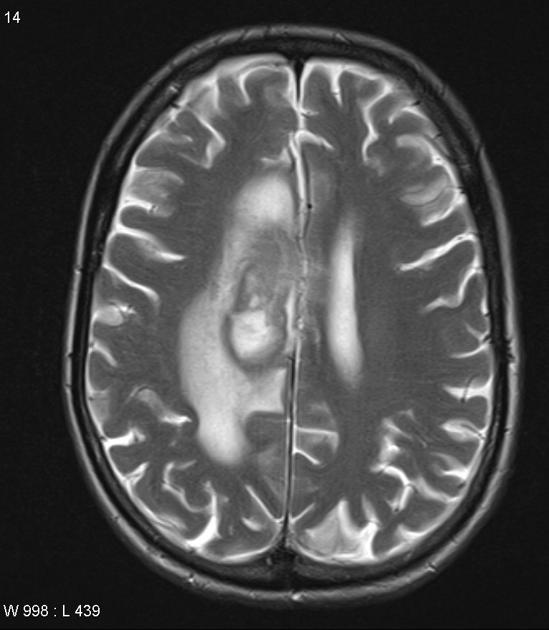
T2: variable
-
majority are iso to hypointense to grey matter
isointense: 33% 9
hypointense: 20% 9 - when present this is a helpful distinguishing feature
T2*/SWI
evidence of some foci of hemorrhage is common on SWI (up to 50%) 17
less commonly seen on less sensitive sequences
-
-
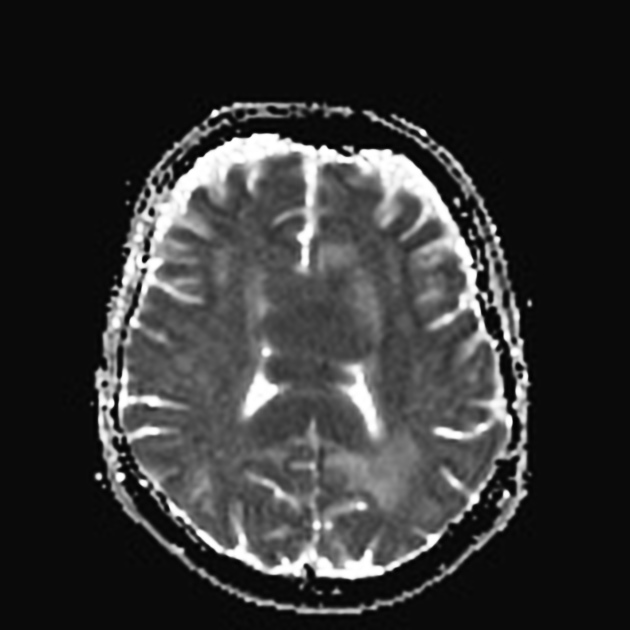
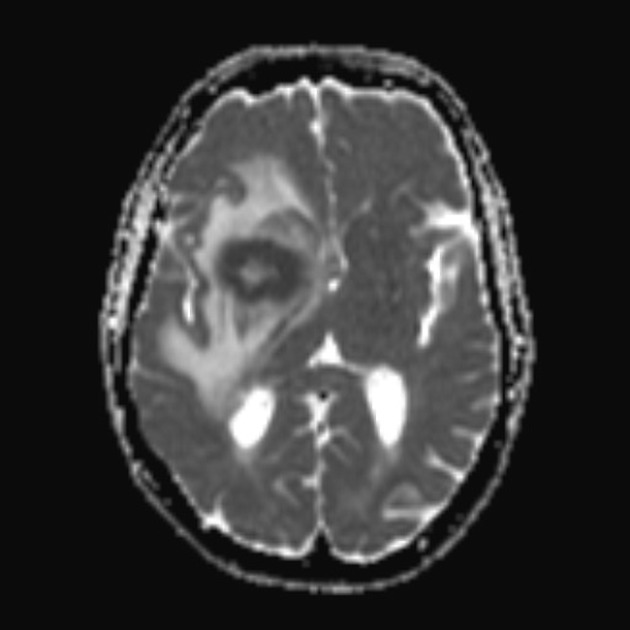
DWI/ADC
restricted diffusion with ADC values lower than a normal brain, typically between 400 and 600 x 10-6 mm2/s (lower than high-grade gliomas and metastases 8,13)
a number of studies have suggested that the lower the ADC values of a tumor the poorer the response to a tumor and higher likelihood of recurrence 13
ADC is particularly useful in assessing response to chemotherapy, with increases in ADC values to above those of normal brain predictive of complete response 13
note: low T2 signal may result in lower b=1000 signal despite low ADC values; blackout effect 17
-
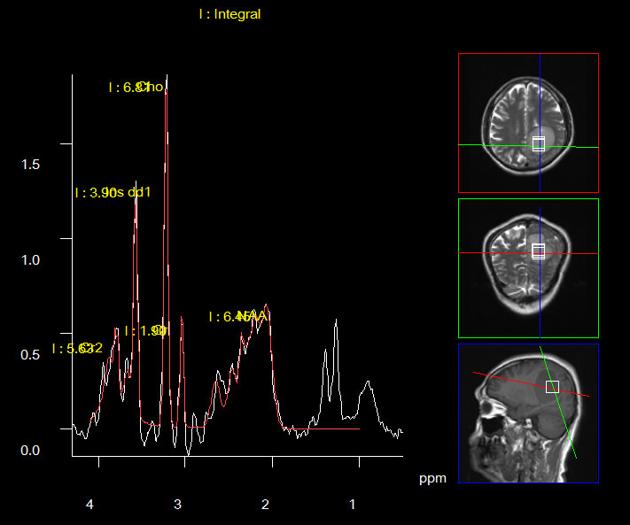
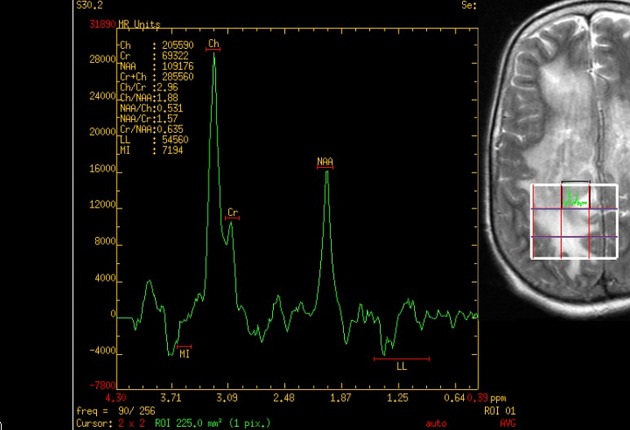
MR spectroscopy
large choline peak
reversed choline/creatinine ratio
markedly decreased NAA
lactate peak may also be seen 7
-
MR perfusion
only modest, if any, increase in rCBV 11,17
note: lower rCBV values have worse prognosis 17
higher percentage signal recovery (PSR) in DSC perfusion PSR-optimized protocol, with typical "overshoot" (PSR>100%)26
In a minority of patients, imaging features can diverge significantly from the aforementioned typical appearances.
This is particularly the case in patients who are immunocompromised, where peripheral ring enhancement is more common. These tumors will have central necrotic components with high T2 signal 1,9.
In a minority of cases, enhancement will be limited or even absent. Whether this represents lower-grade tumors 1,18,19 or intravascular lymphoma (lymphomoatosis) is unclear. What is important to remember is that enhancement does not directly indicate the tumor cells, but rather areas where the blood brain barrier has been disrupted.
Nuclear medicine
Thallium-201 scintigraphy
primary CNS lymphoma shows increased uptake
Fluorine-18 FDG PET
primary CNS lymphoma shows increased uptake
Carbon-11 methionine PET
primary CNS lymphoma shows increased uptake
Treatment and prognosis
Treatment of primary CNS lymphoma is predominantly with steroids (which can dramatically shrink a tumor due to combined anti-edema and cytotoxic effects) and methotrexate-based chemotherapy 4,13. Whole-brain irradiation can also be added, particularly in patients with high-grade tumors, or recurrence 4,13.
There is increasing evidence that in selected patients (those with accessible solitary lesions) local treatment with surgical resection can significantly improve survival 1,20-22.
The tumors are often high grade and despite treatment have a poor prognosis. If only surgical resection is performed, then death occurs within a few months. With high dose chemotherapy, the tumor can be significantly reduced in size; however, recurrence is common, with a median survival of around 30 months 1. Those who are immunocompromised (e.g. HIV positive) have worse outcomes.
Differential diagnosis
For general imaging appearances on CT and MRI consider:
secondary CNS lymphoma: indistinguishable on imaging, however, it tends to involve more leptomeninges (~2/3 of cases) 8
-
cerebral toxoplasmosis: see toxoplasmosis vs lymphoma
toxoplasmosis does not exhibit subependymal spread
more likely to lie in basal ganglia, corticomedullary junction
CNS lymphoma is thallium/PET avid, whereas toxoplasmosis is not
-
more commonly centrally necrotic
more commonly demonstrates evidence of hemorrhage
-
rarely demyelination may precede the diagnosis of primary CNS lymphoma 23,24
-
peripheral enhancement of primary CNS lymphoma is thicker 3
central restricted diffusion







 Unable to process the form. Check for errors and try again.
Unable to process the form. Check for errors and try again.