Amiodarone lung refers to the various manifestations of amiodarone toxicity in the lung including acute lung injury, fibrosis, nodules, hemorrhage and pleural disease.
On this page:
Epidemiology
Amiodarone is a vasodilator which was found to be an effective anti-arrhythmic agent and is consequently in common usage. The reported prevalence of pulmonary toxicity in patients receiving amiodarone is ~10% (range 2-18%) 8.
Toxicity is related to cumulative dose and typically occurs after at least two months on 400 mg/day or two years on 200 mg/day 11. The minimum effective dose should be used and baseline pulmonary function assessed.
Risk factors include 11:
dose and duration of treatment
increasing age
male
ethnicity, e.g. Japanese
pre-existing lung disease
cardiothoracic surgery, e.g. pneumonectomy
high-dose supplemental oxygen
pulmonary angiography
Clinical presentation
Patients typically present with progressive dyspnea as the dominant symptom. Low grade fever, dry cough, malaise, anorexia, pleurisy, thyroid dysfunction, photosensitivity and blurred vision due to corneal deposits have also been reported 2, 11. In approximately a third of patients, the presentation may mimic pulmonary infection 6.
Respiratory function tests are usually abnormal with a restrictive pattern on spirometry and decreased gas transfer 2,6. Hypoxemia is almost always present 6.
Open lung biopsy should be avoided as toxicity may be exacerbated and these patients may already have compromised cardiopulmonary function 11.
Pathology
Amiodarone hydrochloride is a tri-iodinated antiarrhythmic, composed of 37% iodine by weight and structurally similar to thyroxine, which is widely distributed in adipose tissue, liver, spleen and lung. Elimination half-life is 6 months and the metabolites are excreted in bile.
Amiodarone causes direct cytotoxic effects, indirect immunological damage and causes phospholipid accumulation. Bronchoalveolar lavage (BAL) fluid contains cytotoxic T-cells.
Amiodarone accumulates in type II pneumocytes 5,7 associated with hyperplasia and septal thickening. Foamy alveolar macrophages and cytoplasmic lamellar bodies containing surfactant-like material are typical of amiodarone exposure and do not necessarily indicate clinically significant toxicity.
Pathologic patterns include 6,7,11:
pneumonitis (most common) with inflammation and fibrosis
Radiographic features
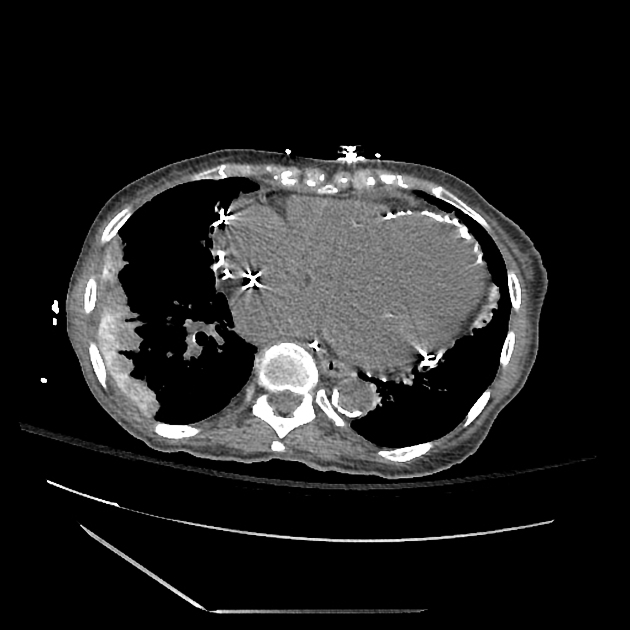
Amiodarone accumulation increases tissue attenuation due to the iodine content. This is simply a marker of exposure and is seen in liver, spleen and atelectatic lung.
Plain radiograph
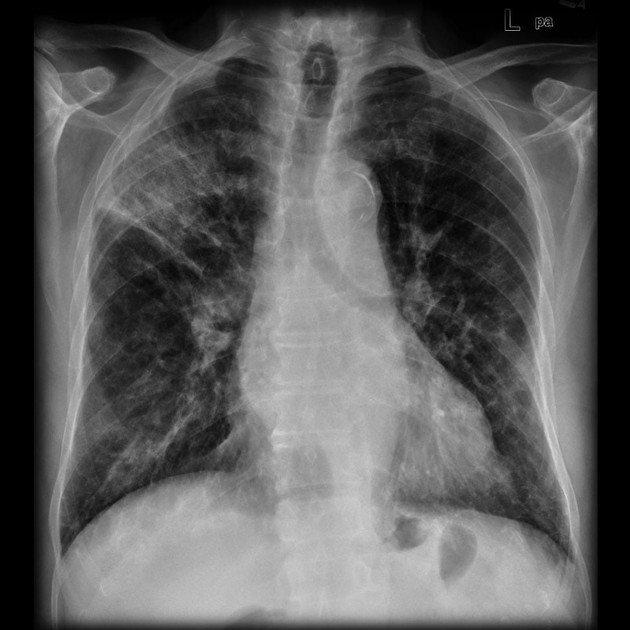
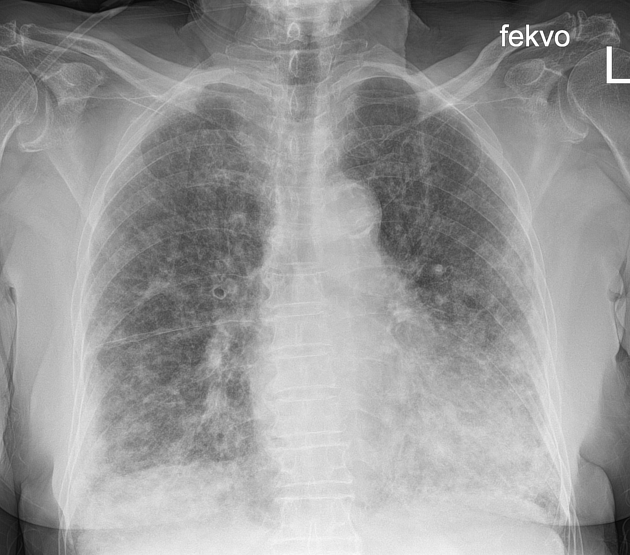
Appearances on chest radiography are variable and non-specific, such as:
peripheral opacities
right upper lobe predominance
CT
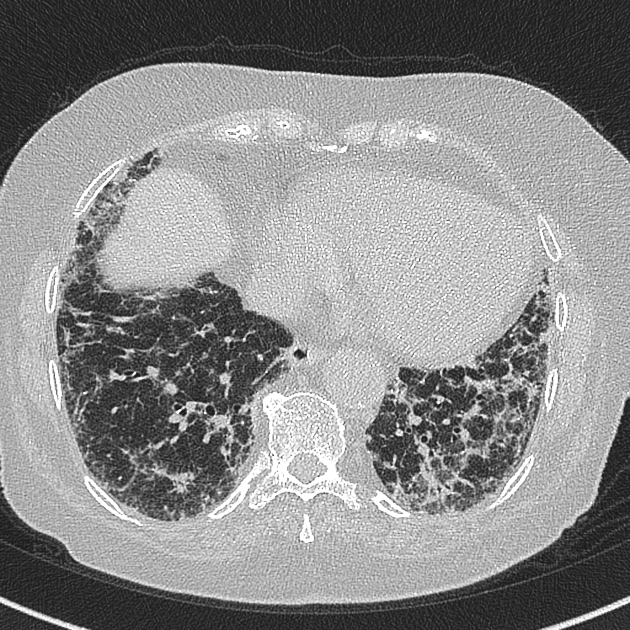
Drug history is helpful as the features are non-specific and variable:
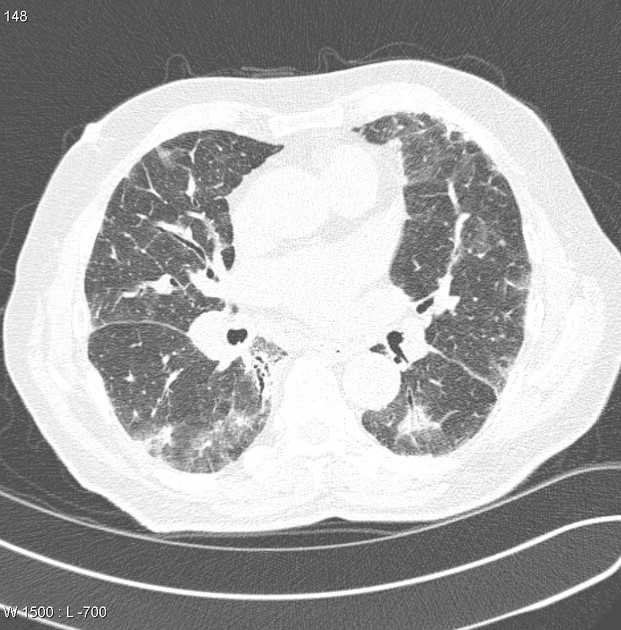
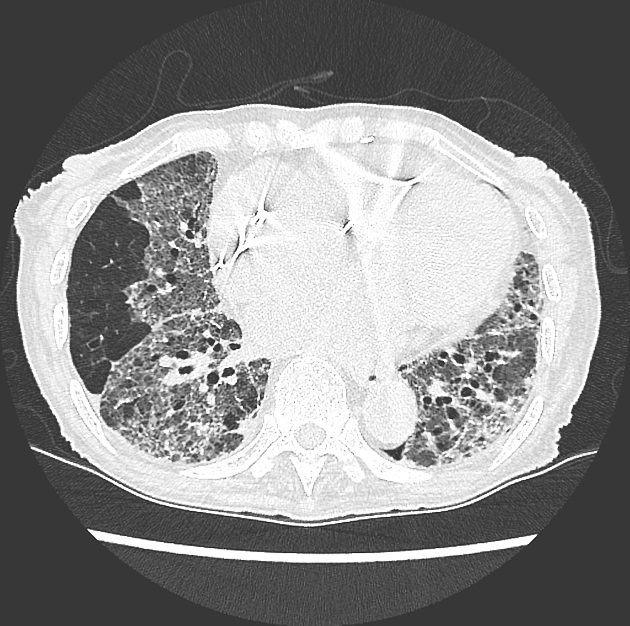
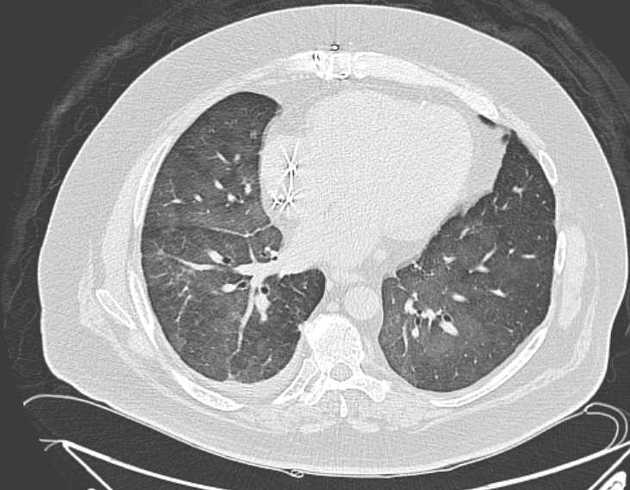
basal predominant fibrosis with reticulation and traction bronchiectasis
bilateral opacities, sometimes ground-glass and sometimes peripheral
peripheral nodule(s) or masses
high attenuation in atelectatic lung, in liver, spleen and heart
Features of heart disease or pre-existing lung disease may also be present.
Nuclear medicine
gallium-67 scan: sensitive but non-specific
FDG-avid, mimicking lung neoplasm
Treatment and prognosis
Cessation of amiodarone and four to twelve months treatment with corticosteroids can arrest progress and often results in resolution of imaging findings over time 3. Overall mortality from amiodarone lung is <10% 6.
Differential diagnosis
Imaging differential considerations include:
other causes of pulmonary fibrosis or diffuse alveolar damage
lung cancer
-
patients with amiodarone lung have normal blood and tissue eosinophil counts 2
also peripheral distribution




 Unable to process the form. Check for errors and try again.
Unable to process the form. Check for errors and try again.