Pulmonary manifestations of cystic fibrosis are some of the best known in cystic fibrosis (CF). This is partly because the lungs are often severely affected and the cause of significant morbidity and mortality.
For general discussion of cystic fibrosis, and a discussion of its other manifestations, please refer to:
-
cystic fibrosis (parent article)
On this page:
Clinical presentation
Clinical presentation is with the expected recurrent bacterial infections and hemoptysis. Patients have a chronic cough and expectorate copious quantities of sputum, frequently blood stained and containing mucous plugs 2,7.
Later in the disease, larger volume hemoptysis, which may be life-threatening, as well as pneumothoraces become more common 2.
Pathology
In the lung, the cystic fibrosis transmembrane regulator (CFTR) is a protein responsible for efflux of chloride and inhibition of the sodium channel's activity which controls the influx of sodium. Therefore, under normal circumstances, salt and chloride remain in the lumen and keep water there osmotically. In CF patients, too little chloride is pumped out, too much sodium is reabsorbed resulting in osmotic re-absorption of water from the lumen. The result is iso-osmotic, but low volume, secretions, which tend to dry out, or be thick as they still contain all the other constituents.
Abnormal CFTR function has other effects 1,3, e.g:
increased bacterial adhesion
disrupted epithelial tight junctions
altered bacterial killing
The organisms most responsible for pulmonary infections are:
Pseudomonas aeruginosa
Staphylococcus aureus: especially in the first 6 months of life 1
Haemophilus influenzae: especially in the first 6 months of life
Burkholderia cepacia
As a result of repeated and chronic infections, there is a marked increase in the number of polymorphonuclear (PMN) leukocytes and associated inflammatory agents, including elastase and collagenase. Over time, these weaken the bronchial walls, leading to bronchiectasis 3.
These chronic bronchial injuries also lead to bronchial angiogenesis and vascular hypertrophy of the bronchial arteries, sometimes with the recruitment of additional aortobronchial collaterals. The bronchial submucosal arteries then become hypertrophied and, given the chronic inflammation, also more fragile, making them more susceptible to bleeding in the airways 9.
Radiographic features
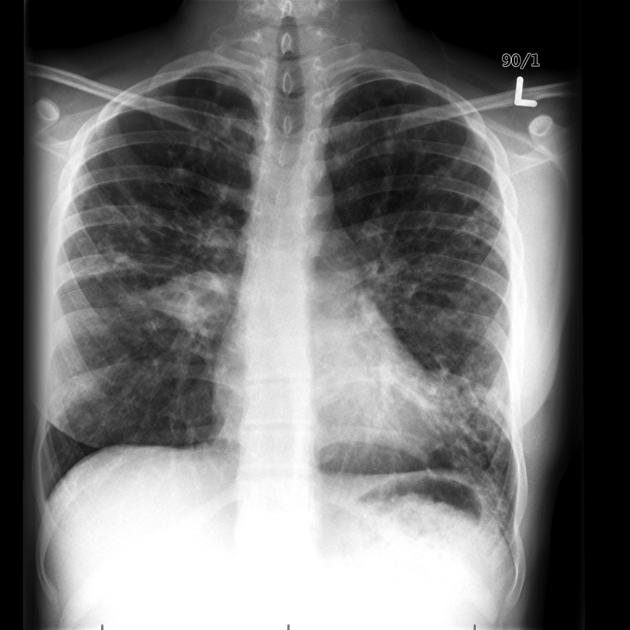
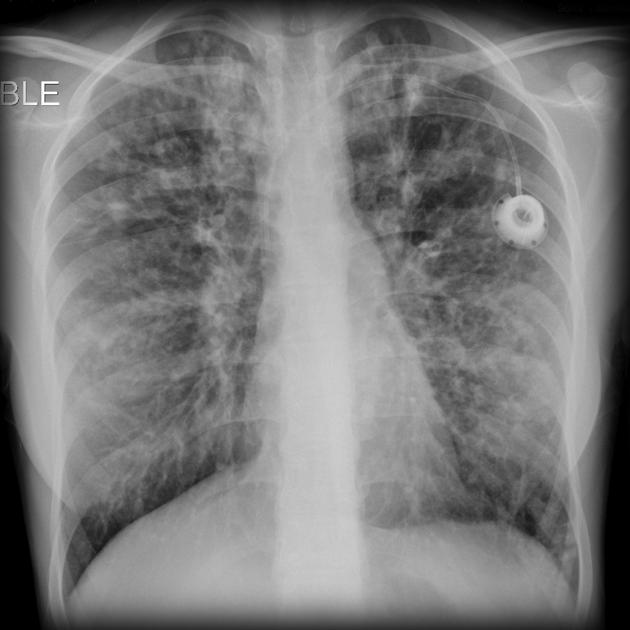
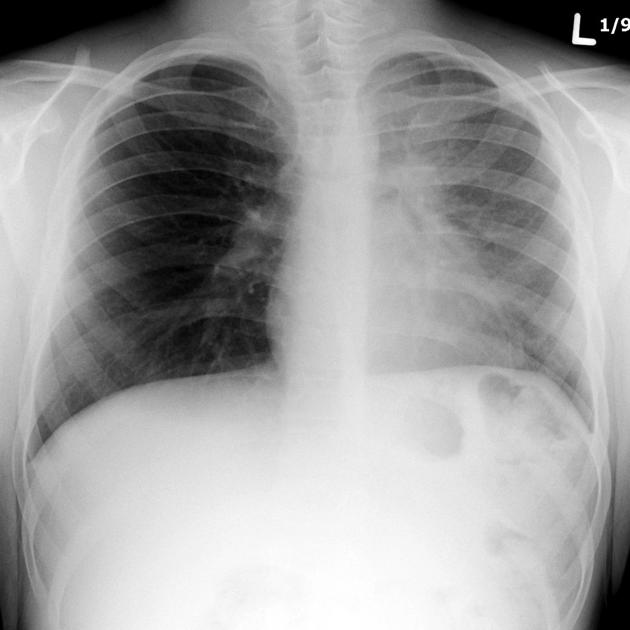
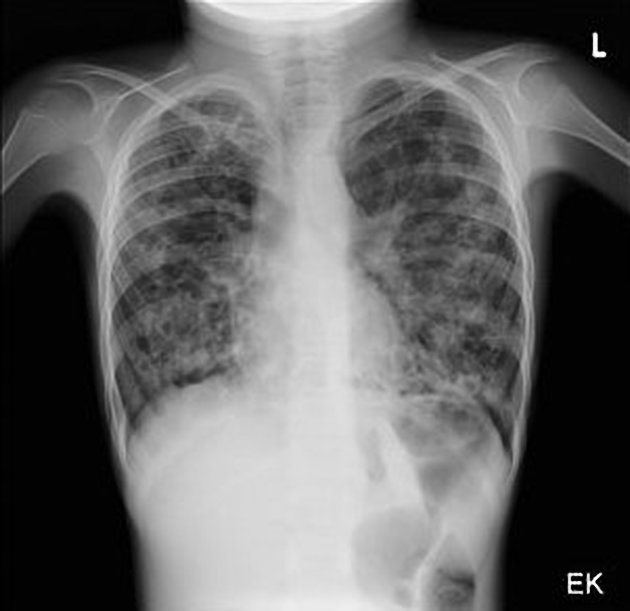
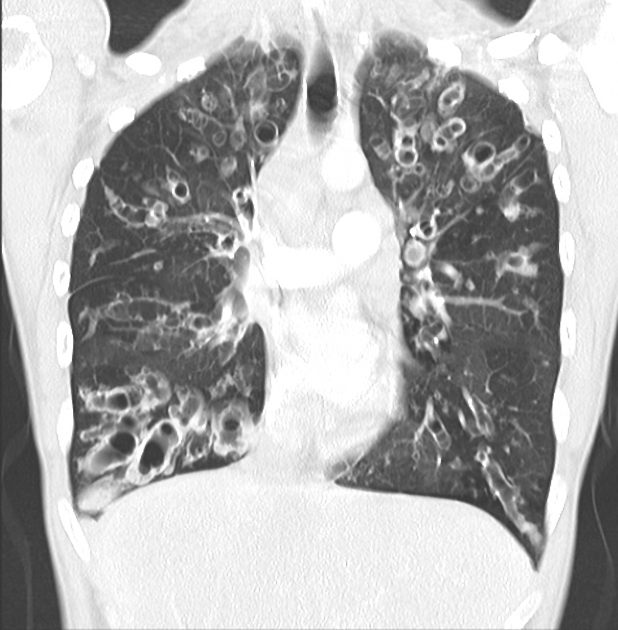
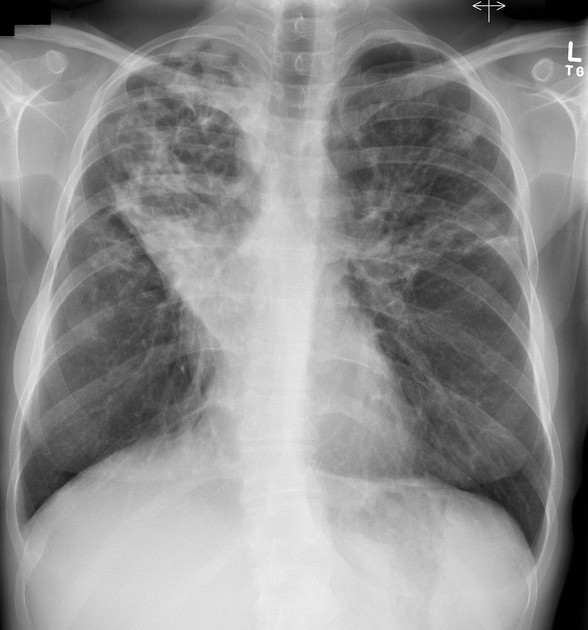
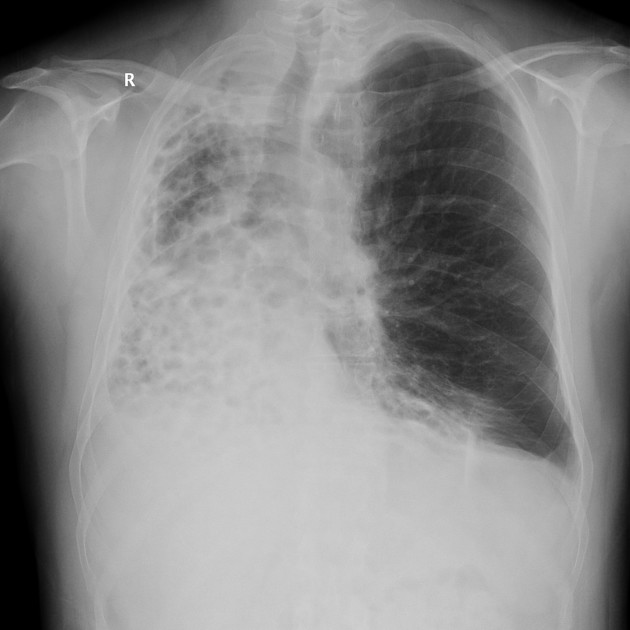
The cardinal finding of later stages of cystic fibrosis is the presence of thick-walled bronchiectasis. These begin as cylindrical and progress through varicoid to cystic forms. The intervening lung is often densely fibrotic and retracted 3.
Although the entire lung is affected, there is a predilection for:
central (perihilar) distribution
upper lobes
apical segment of lower lobes
Other features to be sought include hyperinflation, regions of consolidation, lymph node enlargement, pneumothorax and pulmonary arterial hypertension.
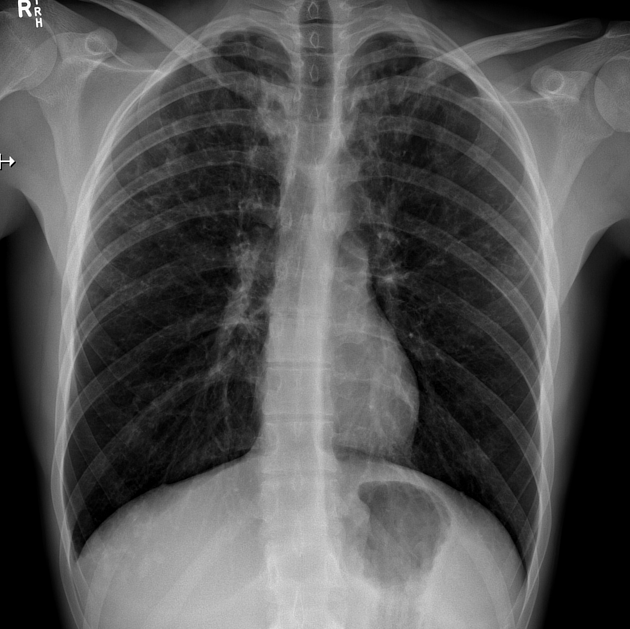
Plain radiograph
Chest radiographs are insensitive to the early changes of cystic fibrosis, with changes seen on HRCT in 65% of patients with CF and normal chest radiographs 6. Later changes include:
bronchiectasis
hyperinflation
lobar collapse
pulmonary arterial enlargement due to pulmonary arterial hypertension in patients with long-standing disease
Scoring
Brasfield scoring system: can be used to score disease severity on plain film
Chrispin-Norman score: is used to provide a summative assessment of structural lung changes in patients
CT
HRCT
HRCT has become indispensable in the monitoring of CF patients and is used to guide therapy and assess response to treatment, as it not only correlates with lung function tests but in some cases pre-empts them. Typically scans are repeated every 6 to 18 months depending on the institution and clinical course.
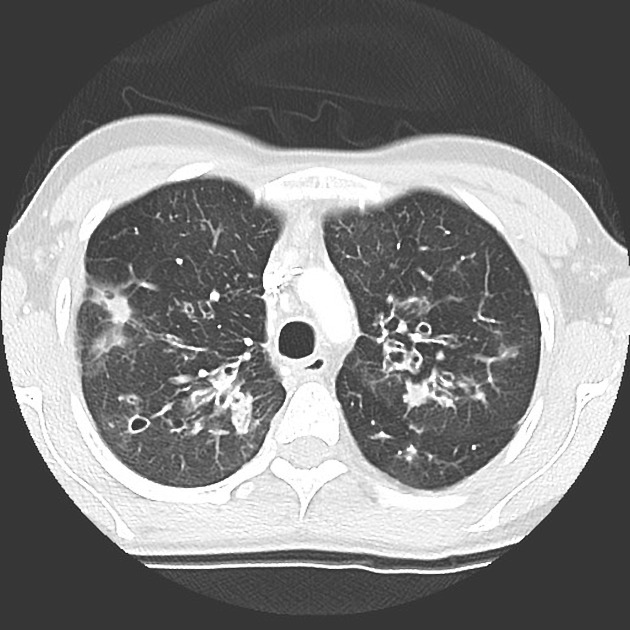
Mucous plugging is of particular importance as it is thought to precede infective exacerbations and thus identification of such plugging may be used to trigger changes in therapy 4.
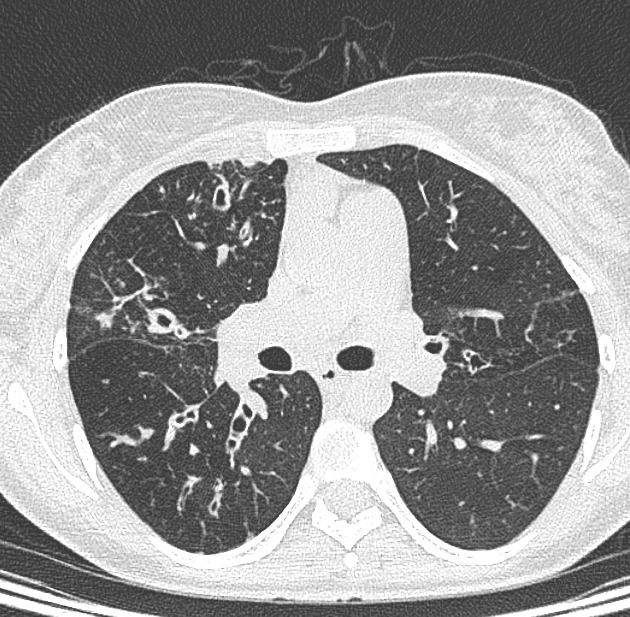
HRCT findings include:
-
bronchial wall and/or peribronchial interstitial thickening 6
especially early in the disease
-
acute infectious bronchiolitis 6
especially early in the disease
centrilobular nodular opacities
branching opacities
-
progresses with time from cylindrical to varicose to cystic
-
due to air trapping
best seen on expiratory scans
mucus plugging within bronchi: finger in glove appearance
CT angiography (thorax)
CT angiography is usually indicated in those patients presenting with hemoptysis and for whom endovascular intervention is being considered. Ideally, the scan should be triggered for maximum enhancement at the descending thoracic aorta 9.
-
it provides the delineation of the bronchial arteries anatomy and their recruited aortobronchial collaterals
contrast blush indicating active bleed is rare even for conventional angiography, and it is not the aim of the scan
Radiation exposure
As a result of repeated examinations, patients are exposed to significant radiation dose during childhood when sensitivity to radiation is much higher. The use of low-dose protocols can significantly reduce cumulative radiation dose 5.
Angiography (DSA)
Angiography is reserved for selective embolization of bronchial arteries in patients with airways bleeding, particularly when presenting unstable due to massive hemoptysis 9.
Treatment and prognosis
Transmembrane conductance regulator (CFTR) modulators correct the malfunctioning protein made by the CFTR gene for those with specific mutations. Further developments will address additional CF mutations. These drugs affect the movement of chloride and water across cell membranes and prevent build up of the sticky mucus that causes organ damage 10. Drugs are taken twice daily and those available are:
Trikafta® (elexacaftor/tezacaftor/ivacaftor)
Symdeko® (tezacaftor/ivacaftor)
Orkambi® (lumacaftor/ivacaftor)
Kalydeco® (ivacaftor)
It is too soon to assess the long-term outcome for patients. Until now, respiratory failure and pulmonary complications account for 95% of deaths in patients with cystic fibrosis 4.
bronchial arterial hypertrophy with pulmonary hemorrhage
allergic bronchopulmonary aspergillosis occurs in 5-10%
Differential diagnosis
Imaging differential considerations include:
-
allergic bronchopulmonary aspergillosis (ABPA)
may co-exist with CF in 5-10% of patients 3 (with serum precipitins against Aspergillus fumigatus detected ~50% of patients)
-
middle/lower lobe predilection, instead of central or upper lobe in cystic fibrosis
See also
cystic fibrosis (parent article)
conditions with upper lobe predominance (mnemonic)







 Unable to process the form. Check for errors and try again.
Unable to process the form. Check for errors and try again.